New tools could catch disease outbreaks earlier - or predict them

A team at the Catalan Institution for Research and Advanced Studies is utilizing drones and weather stations to collect data on how mosquito breeding patterns are changing in response to climate shifts.
Every year, the villages which lie in the so-called ‘Nipah belt’— which stretches along the western border between Bangladesh and India, brace themselves for the latest outbreak. For since 1998, when Nipah virus—a form of hemorrhagic fever most common in Bangladesh—first spilled over into humans, it has been a grim annual visitor to the people of this region.
With a 70 percent fatality rate, no vaccine, and no known treatments, Nipah virus has been dubbed in the Western world as ‘the worst disease no one has ever heard of.’ Currently, outbreaks tend to be relatively contained because it is not very transmissible. The virus circulates throughout Asia in fruit eating bats, and only tends to be passed on to people who consume contaminated date palm sap, a sweet drink which is harvested across Bangladesh.
But as SARS-CoV-2 has shown the world, this can quickly change.
“Nipah virus is among what virologists call ‘the Big 10,’ along with things like Lassa fever and Crimean Congo hemorrhagic fever,” says Noam Ross, a disease ecologist at New York-based non-profit EcoHealth Alliance. “These are pretty dangerous viruses from a lethality perspective, which don’t currently have the capacity to spread into broader human populations. But that can evolve, and you could very well see a variant emerge that has human-human transmission capability.”
That’s not an overstatement. Surveys suggest that mammals harbour about 40,000 viruses, with roughly a quarter capable of infecting humans. The vast majority never get a chance to do so because we don’t encounter them, but climate change can alter that. Recent studies have found that as animals relocate to new habitats due to shifting environmental conditions, the coming decades will bring around 300,000 first encounters between species which normally don’t interact, especially in tropical Africa and southeast Asia. All these interactions will make it far more likely for hitherto unknown viruses to cross paths with humans.
That’s why for the last 16 years, EcoHealth Alliance has been conducting ongoing viral surveillance projects across Bangladesh. The goal is to understand why Nipah is so much more prevalent in the western part of the country, compared to the east, and keep a watchful eye out for new Nipah strains as well as other dangerous pathogens like Ebola.
"There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them," says Cat Lippi, medical geography researcher at the University of Florida.
Until very recently this kind of work has been hampered by the limitations of viral surveillance technology. The PREDICT project, a $200 million initiative funded by the United States Agency for International Development, which conducted surveillance across the Amazon Basin, Congo Basin and extensive parts of South and Southeast Asia, relied upon so-called nucleic acid assays which enabled scientists to search for the genetic material of viruses in animal samples.
However, the project came under criticism for being highly inefficient. “That approach requires a big sampling effort, because of the rarity of individual infections,” says Ross. “Any particular animal may be infected for a couple of weeks, maybe once or twice in its lifetime. So if you sample thousands and thousands of animals, you'll eventually get one that has an Ebola virus infection right now.”
Ross explains that there is now far more interest in serological sampling—the scientific term for the process of drawing blood for antibody testing. By searching for the presence of antibodies in the blood of humans and animals, scientists have a greater chance of detecting viruses which started circulating recently.
Despite the controversy surrounding EcoHealth Alliance’s involvement in so-called gain of function research—experiments that study whether viruses might mutate into deadlier strains—the organization’s separate efforts to stay one step ahead of pathogen evolution are key to stopping the next pandemic.
“Having really cheap and fast surveillance is really important,” says Ross. “Particularly in a place where there's persistent, low level, moderate infections that potentially have the ability to develop into more epidemic or pandemic situations. It means there’s a pathway that something more dangerous can come through."
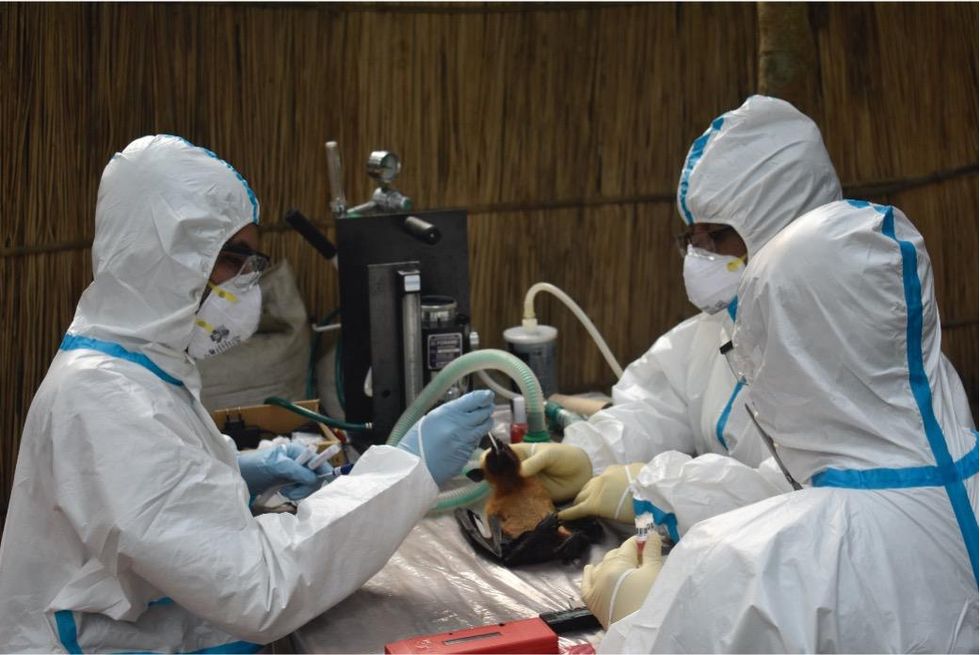
Scientists are searching for the presence of antibodies in the blood of humans and animals in hopes to detect viruses that recently started circulating.
EcoHealth Alliance
In Bangladesh, EcoHealth Alliance is attempting to do this using a newer serological technology known as a multiplex Luminex assay, which tests samples against a panel of known antibodies against many different viruses. It collects what Ross describes as a ‘footprint of information,’ which allows scientists to tell whether the sample contains the presence of a known pathogen or something completely different and needs to be investigated further.
By using this technology to sample human and animal populations across the country, they hope to gain an idea of whether there are any novel Nipah virus variants or strains from the same family, as well as other deadly viral families like Ebola.
This is just one of several novel tools being used for viral discovery in surveillance projects around the globe. Multiple research groups are taking PREDICT’s approach of looking for novel viruses in animals in various hotspots. They collect environmental DNA—mucus, faeces or shed skin left behind in soil, sediment or water—which can then be genetically sequenced.
Five years ago, this would have been a painstaking work requiring bringing collected samples back to labs. Today, thanks to the vast amounts of money spent on new technologies during COVID-19, researchers now have portable sequencing tools they can take out into the field.
Christopher Jerde, a researcher at the UC Santa Barbara Marine Science Institute, points to the Oxford Nanopore MinION sequencer as one example. “I tried one of the early versions of it four years ago, and it was miserable,” he says. “But they’ve really improved, and what we’re going to be able to do in the next five to ten years will be amazing. Instead of having to carefully transport samples back to the lab, we're going to have cigar box-shaped sequencers that we take into the field, plug into a laptop, and do the whole sequencing of an organism.”
In the past, viral surveillance has had to be very targeted and focused on known families of viruses, potentially missing new, previously unknown zoonotic pathogens. Jerde says that the rise of portable sequencers will lead to what he describes as “true surveillance.”
“Before, this was just too complex,” he says. “It had to be very focused, for example, looking for SARS-type viruses. Now we’re able to say, ‘Tell us all the viruses that are here?’ And this will give us true surveillance – we’ll be able to see the diversity of all the pathogens which are in these spots and have an understanding of which ones are coming into the population and causing damage.”
But being able to discover more viruses also comes with certain challenges. Some scientists fear that the speed of viral discovery will soon outpace the human capacity to analyze them all and assess the threat that they pose to us.
“I think we're already there,” says Jason Ladner, assistant professor at Northern Arizona University’s Pathogen and Microbiome Institute. “If you look at all the papers on the expanding RNA virus sphere, there are all of these deposited partial or complete viral sequences in groups that we just don't know anything really about yet.” Bats, for example, carry a myriad of viruses, whose ability to infect human cells we understand very poorly.
Cultivating these viruses under laboratory conditions and testing them on organoids— miniature, simplified versions of organs created from stem cells—can help with these assessments, but it is a slow and painstaking work. One hope is that in the future, machine learning could help automate this process. The new SpillOver Viral Risk Ranking platform aims to assess the risk level of a given virus based on 31 different metrics, while other computer models have tried to do the same based on the similarity of a virus’s genomic sequence to known zoonotic threats.
However, Ladner says that these types of comparisons are still overly simplistic. For one thing, scientists are still only aware of a few hundred zoonotic viruses, which is a very limited data sample for accurately assessing a novel pathogen. Instead, he says that there is a need for virologists to develop models which can determine viral compatibility with human cells, based on genomic data.
“One thing which is really useful, but can be challenging to do, is understand the cell surface receptors that a given virus might use,” he says. “Understanding whether a virus is likely to be able to use proteins on the surface of human cells to gain entry can be very informative.”
As the Earth’s climate heats up, scientists also need to better model the so-called vector borne diseases such as dengue, Zika, chikungunya and yellow fever. Transmitted by the Aedes mosquito residing in humid climates, these blights currently disproportionally affect people in low-income nations. But predictions suggest that as the planet warms and the pests find new homes, an estimated one billion people who currently don’t encounter them might be threatened by their bites by 2080. “When it comes to mosquito-borne diseases we have to worry about shifts in suitable habitat,” says Cat Lippi, a medical geography researcher at the University of Florida. “As climate patterns change on these big scales, we expect to see shifts in where people will be at risk for contracting these diseases.”
Public health practitioners and government decision-makers need tools to make climate-informed decisions about the evolving threat of different infectious diseases. Some projects are already underway. An ongoing collaboration between the Catalan Institution for Research and Advanced Studies and researchers in Brazil and Peru is utilizing drones and weather stations to collect data on how mosquitoes change their breeding patterns in response to climate shifts. This information will then be fed into computer algorithms to predict the impact of mosquito-borne illnesses on different regions.

The team at the Catalan Institution for Research and Advanced Studies is using drones and weather stations to collect data on how mosquito breeding patterns change due to climate shifts.
Gabriel Carrasco
Lippi says that similar models are urgently needed to predict how changing climate patterns affect respiratory, foodborne, waterborne and soilborne illnesses. The UK-based Wellcome Trust has allocated significant assets to fund such projects, which should allow scientists to monitor the impact of climate on a much broader range of infections. “There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them,” she says.
COVID-19’s havoc boosted funding for infectious disease research, but as its threats begin to fade from policymakers’ focus, the money may dry up. Meanwhile, scientists warn that another major infectious disease outbreak is inevitable, potentially within the next decade, so combing the planet for pathogens is vital. “Surveillance is ultimately a really boring thing that a lot of people don't want to put money into, until we have a wide scale pandemic,” Jerde says, but that vigilance is key to thwarting the next deadly horror. “It takes a lot of patience and perseverance to keep looking.”
This article originally appeared in One Health/One Planet, a single-issue magazine that explores how climate change and other environmental shifts are increasing vulnerabilities to infectious diseases by land and by sea. The magazine probes how scientists are making progress with leaders in other fields toward solutions that embrace diverse perspectives and the interconnectedness of all lifeforms and the planet.
Podcast: The Friday Five - your health research roundup
The Friday Five is a new series in which Leaps.org covers five breakthroughs in research over the previous week that you may have missed.
The Friday Five is a new podcast series in which Leaps.org covers five breakthroughs in research over the previous week that you may have missed. There are plenty of controversies and ethical issues in science – and we get into many of them in our online magazine – but there’s also plenty to be excited about, and this news roundup is focused on inspiring scientific work to give you some momentum headed into the weekend.
Covered in this week's Friday Five:
- Puffer fish chemical for treating chronic pain
- Sleep study on the health benefits of waking up multiples times per night
- Best exercise regimens for reducing the risk of mortality aka living longer
- AI breakthrough in mapping protein structures with DeepMind
- Ultrasound stickers to see inside your body
CandyCodes could provide sweet justice against fake pills
A bioengineer at the University of California, Riverside, may have found a way to prevent counterfeit medications: pill coatings inspired by the sprinkles on baked goods and candies.
When we swallow a pill, we hope it will work without side effects. Few of us know to worry about a growing issue facing the pharmaceutical industry: counterfeit medications. These pills, patches, and other medical products might look just like the real thing. But they’re often stuffed with fillers that dilute the medication’s potency or they’re simply substituted for lookalikes that contain none of the prescribed medication at all.
Now, bioengineer William Grover at the University of California, Riverside, may have a solution. Inspired by the tiny, multi-colored sprinkles called nonpareils that decorate baked goods and candies, Grover created CandyCodes pill coatings to prevent counterfeits.
The idea was borne out of pandemic boredom. Confined to his home, Grover was struck by the patterns of nonpareils he saw on candies, and found himself counting the number of little balls on each one. “It’s random, how they’re applied,” he says. “I wondered if it ever repeats itself or if each of these candies is unique in the entire world.” He suspected the latter, and some quick math proved his hypothesis: Given dozens of nonpareils per candy in a handful of different colors, it’s highly unlikely that the sprinklings on any two candies would be identical.
He quickly realized his finding could have practical applications: pills or capsules could be coated with similar “sprinkles,” with the manufacturer photographing each pill or capsule before selling its products. Consumers looking to weed out fakes could potentially take a photo with their cell phones and go online to compare images of their own pills to the manufacturer’s database, with the help of an algorithm that would determine their authenticity. Or, a computer could generate another type of unique identifier, such as a text-based code, tracking to the color and location of the sprinkles. This would allow for a speedier validation than a photo-based comparison, Grover says. “It could be done very quickly, in a fraction of a second.”
Researchers and manufacturers have already developed some anti-counterfeit tools, including built-in identifiers like edible papers with scannable QR codes. But such methods, while functional, can be costly to implement, Grover says.
It wouldn’t be paranoid to take such precautions. Counterfeits are a growing problem, according to Young Kim, a biomedical engineer at Purdue University who was not involved in the CandyCodes study. “There are approximately 40,000 online pharmacies that one can access via the Internet,” he says. “Only three to four percent of them are operated legally.” Purchases from online pharmacies rose dramatically during the pandemic, and Kim expects a boom in counterfeit medical products alongside it.
The FDA warns that U.S. consumers can be exposed to counterfeits through online purchases, in particular. The problem is magnified in low- to middle-income nations, where one in 10 medical products are counterfeit, according to a World Health Organization estimate. Cost doesn’t seem to be a factor, either; antimalarials and antibiotics are most often reported as counterfeits or fakes, and generic medications are swapped as often as brand-name drugs, according to the same WHO report.
Counterfeits weren’t tracked globally until 2013; since then, there have been 1,500 reports to the WHO, with actual incidences of counterfeiting likely much higher. Fake medicines have been estimated to result in costs of $200 billion each year, and are blamed for more than 72,000 pneumonia- and 116,000 malaria-related deaths.
Researchers and manufacturers have already developed some anti-counterfeit tools, including built-in identifiers like edible papers with scannable QR codes or barcodes that are stamped onto or otherwise incorporated into pills and other medical products. But such methods, while functional, can be costly to implement, Grover says.

CandyCodes could provide unique identifiers for at least 41 million pills for every person on the planet.
William Grover
“Putting universal codes on each pill and each dosage is attractive,” he says. “The challenge is, how can we do it in a way that requires as little modification to the existing manufacturing process as possible? That's where I hope CandyCodes have an edge. It's not zero modification, but I hope it is as minor a modification of the manufacturing process as possible.”
Kim calls the concept “a clever idea to introduce entropy for high-level security” even if it may not be as close to market as other emerging technologies, including some edible watermarks he’s helped develop. He points out that CandyCodes still needs to be tested for reproducibility and readability.
The possibilities are already intriguing, though. Grover’s recent research, published in Scientific Reports, predicts that unique codes could be used for at least 41 million pills for every person on the planet.
Sadly, CandyCodes’ multicolored bits probably won’t taste like candy. They must be made of non-caloric ingredients to meet the international regulatory standards that govern food dyes and colorants. But Grover hopes CandyCodes represent a simple, accessible solution to a heart-wrenching issue. “This feels like trying to track down and go after bad guys,” he says. “Someone who would pass off a medicine intended for a child or a sick person and pass it off as something effective, I can't imagine anything much more evil than that. It's fun and, and a little fulfilling to try to develop technologies that chip away at that.”