Trading syphilis for malaria: How doctors treated one deadly disease by infecting patients with another

In the 1920s, doctors induced a high fever in patients - so called "fever therapy" - as a way to help them recover from syphilis, though it involved ethical problems.
If you had lived one hundred years ago, syphilis – a bacterial infection spread by sexual contact – would likely have been one of your worst nightmares. Even though syphilis still exists, it can now be detected early and cured quickly with a course of antibiotics. Back then, however, before antibiotics and without an easy way to detect the disease, syphilis was very often a death sentence.
To understand how feared syphilis once was, it’s important to understand exactly what it does if it’s allowed to progress: the infections start off as small, painless sores or even a single sore near the vagina, penis, anus, or mouth. The sores disappear around three to six weeks after the initial infection – but untreated, syphilis moves into a secondary stage, often presenting as a mild rash in various areas of the body (such as the palms of a person’s hands) or through other minor symptoms. The disease progresses from there, often quietly and without noticeable symptoms, sometimes for decades before it reaches its final stages, where it can cause blindness, organ damage, and even dementia. Research indicates, in fact, that as much as 10 percent of psychiatric admissions in the early 20th century were due to dementia caused by syphilis, also known as neurosyphilis.
Like any bacterial disease, syphilis can affect kids, too. Though it’s spread primarily through sexual contact, it can also be transmitted from mother to child during birth, causing lifelong disability.
The poet-physician Aldabert Bettman, who wrote fictionalized poems based on his experiences as a doctor in the 1930s, described the effect syphilis could have on an infant in his poem Daniel Healy:
I always got away clean
when I went out
With the boys.
The night before
I was married
I went out,—But was not so fortunate;
And I infected
My bride.
When little Daniel
Was born
His eyes discharged;
And I dared not tell
That because
I had seen too much
Little Daniel sees not at all
Given the horrors of untreated syphilis, it’s maybe not surprising that people would go to extremes to try and treat it. One of the earliest remedies for syphilis, dating back to 15th century Naples, was using mercury – either rubbing it on the skin where blisters appeared, or breathing it in as a vapor. (Not surprisingly, many people who underwent this type of “treatment” died of mercury poisoning.)
Other primitive treatments included using tinctures made of a flowering plant called guaiacum, as well as inducing “sweat baths” to eliminate the syphilitic toxins. In 1910, an arsenic-based drug called Salvarsan hit the market and was hailed as a “magic bullet” for its ability to target and destroy the syphilis-causing bacteria without harming the patient. However, while Salvarsan was effective in treating early-stage syphilis, it was largely ineffective by the time the infection progressed beyond the second stage. Tens of thousands of people each year continued to die of syphilis or were otherwise shipped off to psychiatric wards due to neurosyphilis.
It was in one of these psychiatric units in the early 20th century that Dr. Julius Wagner-Juaregg got the idea for a potential cure.
Wagner-Juaregg was an Austrian-born physician trained in “experimental pathology” at the University of Vienna. Wagner-Juaregg started his medical career conducting lab experiments on animals and then moved on to work at different psychiatric clinics in Vienna, despite having no training in psychiatry or neurology.
Wagner-Juaregg’s work was controversial to say the least. At the time, medicine – particularly psychiatric medicine – did not have anywhere near the same rigorous ethical standards that doctors, researchers, and other scientists are bound to today. Wagner-Juaregg would devise wild theories about the cause of their psychiatric ailments and then perform experimental procedures in an attempt to cure them. (As just one example, Wagner-Juaregg would sterilize his adolescent male patients, thinking “excessive masturbation” was the cause of their schizophrenia.)
But sometimes these wild theories paid off. In 1883, during his residency, Wagner-Juaregg noted that a female patient with mental illness who had contracted a skin infection and suffered a high fever experienced a sudden (and seemingly miraculous) remission from her psychosis symptoms after the fever had cleared. Wagner-Juaregg theorized that inducing a high fever in his patients with neurosyphilis could help them recover as well.
Eventually, Wagner-Juaregg was able to put his theory to the test. Around 1890, Wagner-Juaregg got his hands on something called tuberculin, a therapeutic treatment created by the German microbiologist Robert Koch in order to cure tuberculosis. Tuberculin would later turn out to be completely ineffective for treating tuberculosis, often creating severe immune responses in patients – but for a short time, Wagner-Juaregg had some success in using tuberculin to help his dementia patients. Giving his patients tuberculin resulted in a high fever – and after completing the treatment, Wagner-Jauregg reported that his patient’s dementia was completely halted. The success was short-lived, however: Wagner-Juaregg eventually had to discontinue tuberculin as a treatment, as it began to be considered too toxic.
By 1917, Wagner-Juaregg’s theory about syphilis and fevers was becoming more credible – and one day a new opportunity presented itself when a wounded soldier, stricken with malaria and a related fever, was accidentally admitted to his psychiatric unit.
When his findings were published in 1918, Wagner-Juaregg’s so-called “fever therapy” swept the globe.
What Wagner-Juaregg did next was ethically deplorable by any standard: Before he allowed the soldier any quinine (the standard treatment for malaria at the time), Wagner-Juaregg took a small sample of the soldier’s blood and inoculated three syphilis patients with the sample, rubbing the blood on their open syphilitic blisters.
It’s unclear how well the malaria treatment worked for those three specific patients – but Wagner-Juaregg’s records show that in the span of one year, he inoculated a total of nine patients with malaria, for the sole purpose of inducing fevers, and six of them made a full recovery. Wagner-Juaregg’s treatment was so successful, in fact, that one of his inoculated patients, an actor who was unable to work due to his dementia, was eventually able to find work again and return to the stage. Two additional patients – a military officer and a clerk – recovered from their once-terminal illnesses and returned to their former careers as well.
When his findings were published in 1918, Wagner-Juaregg’s so-called “fever therapy” swept the globe. The treatment was hailed as a breakthrough – but it still had risks. Malaria itself had a mortality rate of about 15 percent at the time. Many people considered that to be a gamble worth taking, compared to dying a painful, protracted death from syphilis.
Malaria could also be effectively treated much of the time with quinine, whereas other fever-causing illnesses were not so easily treated. Triggering a fever by way of malaria specifically, therefore, became the standard of care.
Tens of thousands of people with syphilitic dementia would go on to be treated with fever therapy until the early 1940s, when a combination of Salvarsan and penicillin caused syphilis infections to decline. Eventually, neurosyphilis became rare, and then nearly unheard of.
Despite his contributions to medicine, it’s important to note that Wagner-Juaregg was most definitely not a person to idolize. In fact, he was an outspoken anti-Semite and proponent of eugenics, arguing that Jews were more prone to mental illness and that people who were mentally ill should be forcibly sterilized. (Wagner-Juaregg later became a Nazi sympathizer during Hitler’s rise to power even though, bizarrely, his first wife was Jewish.) Another problematic issue was that his fever therapy involved experimental treatments on many who, due to their cognitive issues, could not give informed consent.
Lack of consent was also a fundamental problem with the syphilis study at Tuskegee, appalling research that began just 14 years after Wagner-Juaregg published his “fever therapy” findings.
Still, despite his outrageous views, Wagner-Juaregg was awarded the Nobel Prize in Medicine or Physiology in 1927 – and despite some egregious human rights abuses, the miraculous “fever therapy” was partly responsible for taming one of the deadliest plagues in human history.
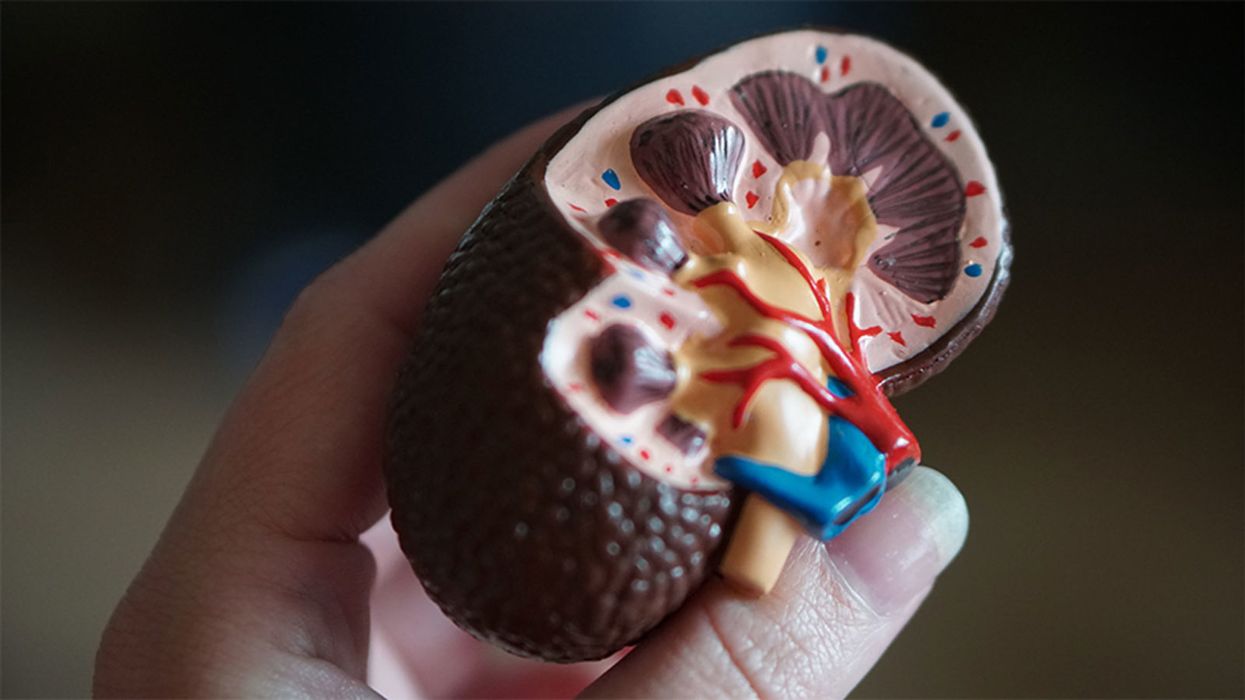
A model of a human kidney.
Science's dream of creating perfect custom organs on demand as soon as a patient needs one is still a long way off. But tiny versions are already serving as useful research tools and stepping stones toward full-fledged replacements.
Although organoids cannot yet replace kidneys, they are invaluable tools for research.
The Lowdown
Australian researchers have grown hundreds of mini human kidneys in the past few years. Known as organoids, they function much like their full-grown counterparts, minus a few features due to a lack of blood supply.
Cultivated in a petri dish, these kidneys are still a shadow of their human counterparts. They grow no larger than one-sixth of an inch in diameter; fully developed organs are up to five inches in length. They contain no more than a few dozen nephrons, the kidney's individual blood-filtering unit, whereas a fully-grown kidney has about 1 million nephrons. And the dish variety live for just a few weeks.
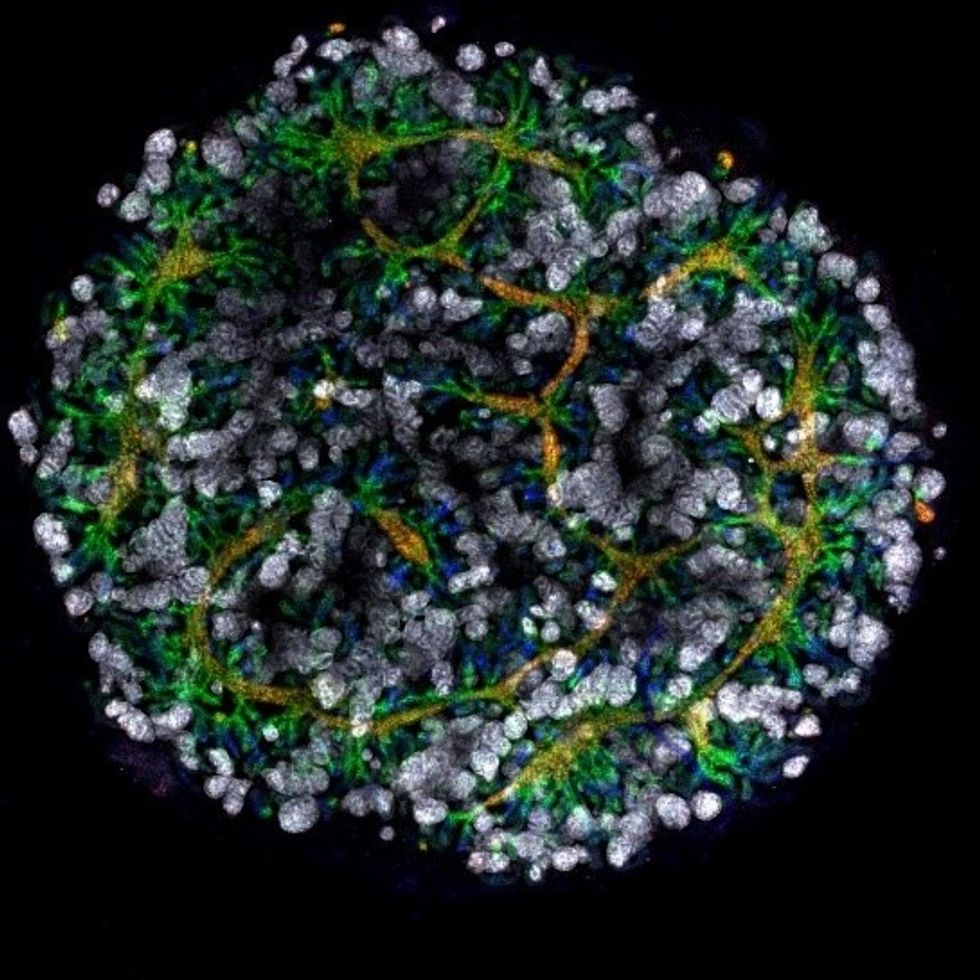
An organoid kidney created by the Murdoch Children's Institute in Melbourne, Australia.
Photo Credit: Shahnaz Khan.
But Melissa Little, head of the kidney research laboratory at the Murdoch Children's Institute in Melbourne, says these organoids are invaluable tools for research. Although renal failure is rare in children, more than half of those who suffer from such a disorder inherited it.
The mini kidneys enable scientists to better understand the progression of such disorders because they can be grown with a patient's specific genetic condition.
Mature stem cells can be extracted from a patient's blood sample and then reprogrammed to become like embryonic cells, able to turn into any type of cell in the body. It's akin to walking back the clock so that the cells regain unlimited potential for development. (The Japanese scientist who pioneered this technique was awarded the Nobel Prize in 2012.) These "induced pluripotent stem cells" can then be chemically coaxed to grow into mini kidneys that have the patient's genetic disorder.
"The (genetic) defects are quite clear in the organoids, and they can be monitored in the dish," Little says. To date, her research team has created organoids from 20 different stem cell lines.
Medication regimens can also be tested on the organoids, allowing specific tailoring for each patient. For now, such testing remains restricted to mice, but Little says it eventually will be done on human organoids so that the results can more accurately reflect how a given patient will respond to particular drugs.
Next Steps
Although these organoids cannot yet replace kidneys, Little says they may plug a huge gap in renal care by assisting in developing new treatments for chronic conditions. Currently, most patients with a serious kidney disorder see their options narrow to dialysis or organ transplantation. The former not only requires multiple sessions a week, but takes a huge toll on patient health.
Ten percent of older patients on dialysis die every year in the U.S. Aside from the physical trauma of organ transplantation, finding a suitable donor outside of a family member can be difficult.
"This is just another great example of the potential of pluripotent stem cells."
Meanwhile, the ongoing creation of organoids is supplying Little and her colleagues with enough information to create larger and more functional organs in the future. According to Little, researchers in the Netherlands, for example, have found that implanting organoids in mice leads to the creation of vascular growth, a potential pathway toward creating bigger and better kidneys.
And while Little acknowledges that creating a fully-formed custom organ is the ultimate goal, the mini organs are an important bridge step.
"This is just another great example of the potential of pluripotent stem cells, and I am just passionate to see it do some good."
Phil Gutis, an Alzheimer's patient who participated in a failed clinical trial, poses with his dog Abe.
Phil Gutis never had a stellar memory, but when he reached his early 50s, it became a problem he could no longer ignore. He had trouble calculating how much to tip after a meal, finding things he had just put on his desk, and understanding simple driving directions.
From 1998-2017, industry sources reported 146 failed attempts at developing Alzheimer's drugs.
So three years ago, at age 54, he answered an ad for a drug trial seeking people experiencing memory issues. He scored so low in the memory testing he was told something was wrong. M.R.I.s and PET scans confirmed that he had early-onset Alzheimer's disease.
Gutis, who is a former New York Times reporter and American Civil Liberties Union spokesman, felt fortunate to get into an advanced clinical trial of a new treatment for Alzheimer's disease. The drug, called aducanumab, had shown promising results in earlier studies.
Four years of data had found that the drug effectively reduced the burden of protein fragments called beta-amyloids, which destroy connections between nerve cells. Amyloid plaques are found in the brains of patients with Alzheimer's disease and are associated with impairments in thinking and memory.
Gutis eagerly participated in the clinical trial and received 35 monthly infusions. "For the first 20 infusions, I did not know whether I was receiving the drug or the placebo," he says. "During the last 15 months, I received aducanumab. But it really didn't matter if I was receiving the drug or the placebo because on March 21, the trial was stopped because [the drug company] Biogen found that the treatments were ineffective."
The news was devastating to the trial participants, but also to the Alzheimer's research community. Earlier this year, another pharmaceutical company, Roche, announced it was discontinuing two of its Alzheimer's clinical trials. From 1998-2017, industry sources reported 146 failed attempts at developing Alzheimer's drugs. There are five prescription drugs approved to treat its symptoms, but a cure remains elusive. The latest failures have left researchers scratching their heads about how to approach attacking the disease.
The failure of aducanumab was also another setback for the estimated 5.8 million people who have Alzheimer's in the United States. Of these, around 5.6 million are older than 65 and 200,000 suffer from the younger-onset form, including Gutis.
Gutis is understandably distraught about the cancellation of the trial. "I really had hopes it would work. So did all the patients."
While drug companies have failed so far, another group is stepping up to expedite the development of a cure: venture philanthropists.
For now, he is exercising every day to keep his blood flowing, which is supposed to delay the progression of the disease, and trying to eat a low-fat diet. "But I know that none of it will make a difference. Alzheimer's is a progressive disease. There are no treatments to delay it, let alone cure it."
But while drug companies have failed so far, another group is stepping up to expedite the development of a cure: venture philanthropists. These are successful titans of industry and dedicated foundations who are donating large sums of money to fill a much-needed void – funding research to look for new biomarkers.
Biomarkers are neurochemical indicators that can be used to detect the presence of a disease and objectively measure its progression. There are currently no validated biomarkers for Alzheimer's, but researchers are actively studying promising candidates. The hope is that they will find a reliable way to identify the disease even before the symptoms of mental decline show up, so that treatments can be directed at a very early stage.
Howard Fillit, Founding Executive Director and Chief Science Officer of the Alzheimer's Drug Discovery Foundation, says, "We need novel biomarkers to diagnose Alzheimer's disease and related dementias. But pharmaceutical companies don't put money into biomarkers research."
One of the venture philanthropists who has recently stepped up to the task is Bill Gates. In January 2018, he announced his father had Alzheimer's disease in an interview on the Today Show with Maria Shriver, whose father Sargent Shriver, died of Alzheimer's disease in 2011. Gates told Ms. Shriver that he had invested $100 million into Alzheimer's research, with $50 million of his donation going to Dementia Discovery Fund, which looks for new cures and treatments.
That August, Gates joined other investors in a new fund called Diagnostics Accelerator. The project aims to supports researchers looking to speed up new ideas for earlier and better diagnosis of the disease.
Gates and other donors committed more than $35 million to help launch it, and this April, Jeff and Mackenzie Bezos joined the coalition, bringing the current program funding to nearly $50 million.
"It makes sense that a challenge this significant would draw the attention of some of the world's leading thinkers."
None of these funders stand to make a profit on their donation, unlike traditional research investments by drug companies. The standard alternatives to such funding have upsides -- and downsides.
As Bill Gates wrote on his blog, "Investments from governments or charitable organizations are fantastic at generating new ideas and cutting-edge research -- but they're not always great at creating usable products, since no one stands to make a profit at the end of the day.
"Venture capital, on the other end of the spectrum, is more likely to develop a test that will reach patients, but its financial model favors projects that will earn big returns for investors. Venture philanthropy splits the difference. It incentivizes a bold, risk-taking approach to research with an end goal of a real product for real patients. If any of the projects backed by Diagnostics Accelerator succeed, our share of the financial windfall goes right back into the fund."
Gutis said he is thankful for any attention given to finding a cure for Alzheimer's.
"Most doctors and scientists will tell you that we're still in the dark ages when it comes to fully understanding how the brain works, let alone figuring out the cause or treatment for Alzheimer's.
"It makes sense that a challenge this significant would draw the attention of some of the world's leading thinkers. I only hope they can be more successful with their entrepreneurial approach to finding a cure than the drug companies have been with their more traditional paths."