Tiny, tough “water bears” may help bring new vaccines and medicines to sub-Saharan Africa
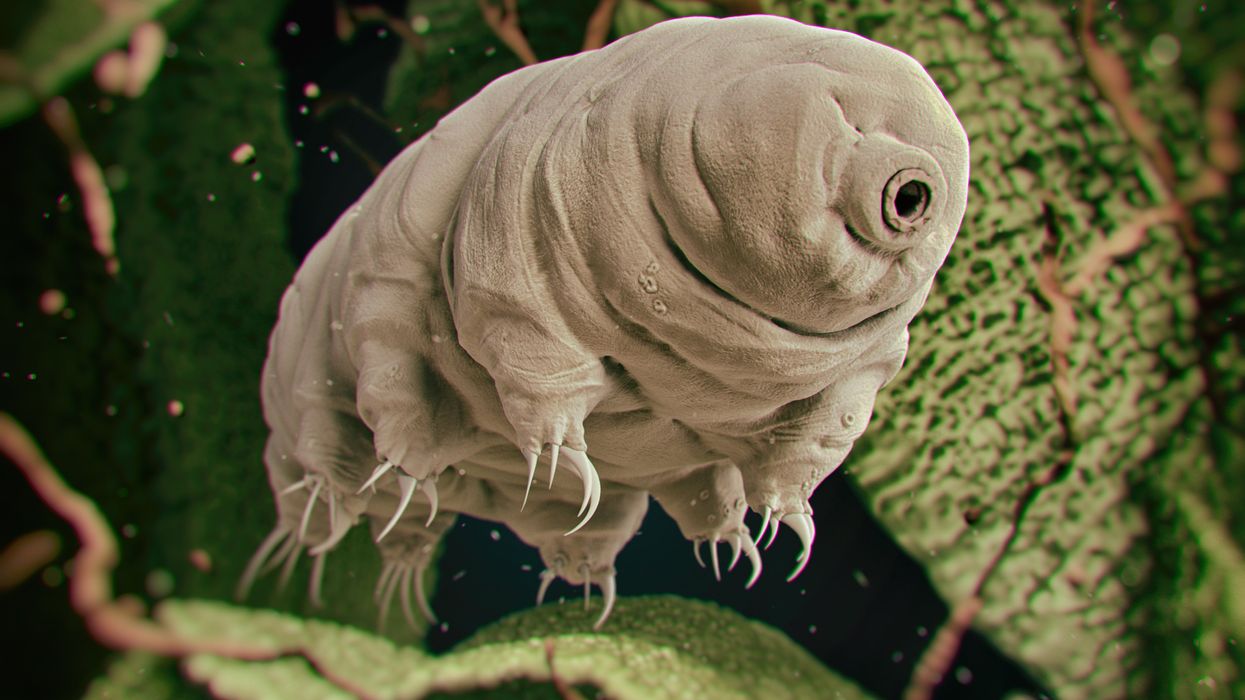
Tardigrades can completely dehydrate and later rehydrate themselves, a survival trick that scientists are harnessing to preserve medicines in hot temperatures.
Microscopic tardigrades, widely considered to be some of the toughest animals on earth, can survive for decades without oxygen or water and are thought to have lived through a crash-landing on the moon. Also known as water bears, they survive by fully dehydrating and later rehydrating themselves – a feat only a few animals can accomplish. Now scientists are harnessing tardigrades’ talents to make medicines that can be dried and stored at ambient temperatures and later rehydrated for use—instead of being kept refrigerated or frozen.
Many biologics—pharmaceutical products made by using living cells or synthesized from biological sources—require refrigeration, which isn’t always available in many remote locales or places with unreliable electricity. These products include mRNA and other vaccines, monoclonal antibodies and immuno-therapies for cancer, rheumatoid arthritis and other conditions. Cooling is also needed for medicines for blood clotting disorders like hemophilia and for trauma patients.
Formulating biologics to withstand drying and hot temperatures has been the holy grail for pharmaceutical researchers for decades. It’s a hard feat to manage. “Biologic pharmaceuticals are highly efficacious, but many are inherently unstable,” says Thomas Boothby, assistant professor of molecular biology at University of Wyoming. Therefore, during storage and shipping, they must be refrigerated at 2 to 8 degrees Celsius (35 to 46 degrees Fahrenheit). Some must be frozen, typically at -20 degrees Celsius, but sometimes as low -90 degrees Celsius as was the case with the Pfizer Covid vaccine.
For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
The costly cold chain
The logistics network that ensures those temperature requirements are met from production to administration is called the cold chain. This cold chain network is often unreliable or entirely lacking in remote, rural areas in developing nations that have malfunctioning electrical grids. “Almost all routine vaccines require a cold chain,” says Christopher Fox, senior vice president of formulations at the Access to Advanced Health Institute. But when the power goes out, so does refrigeration, putting refrigerated or frozen medical products at risk. Consequently, the mRNA vaccines developed for Covid-19 and other conditions, as well as more traditional vaccines for cholera, tetanus and other diseases, often can’t be delivered to the most remote parts of the world.
To understand the scope of the challenge, consider this: In the U.S., more than 984 million doses of Covid-19 vaccine have been distributed so far. Each one needed refrigeration that, even in the U.S., proved challenging. Now extrapolate to all vaccines and the entire world. For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
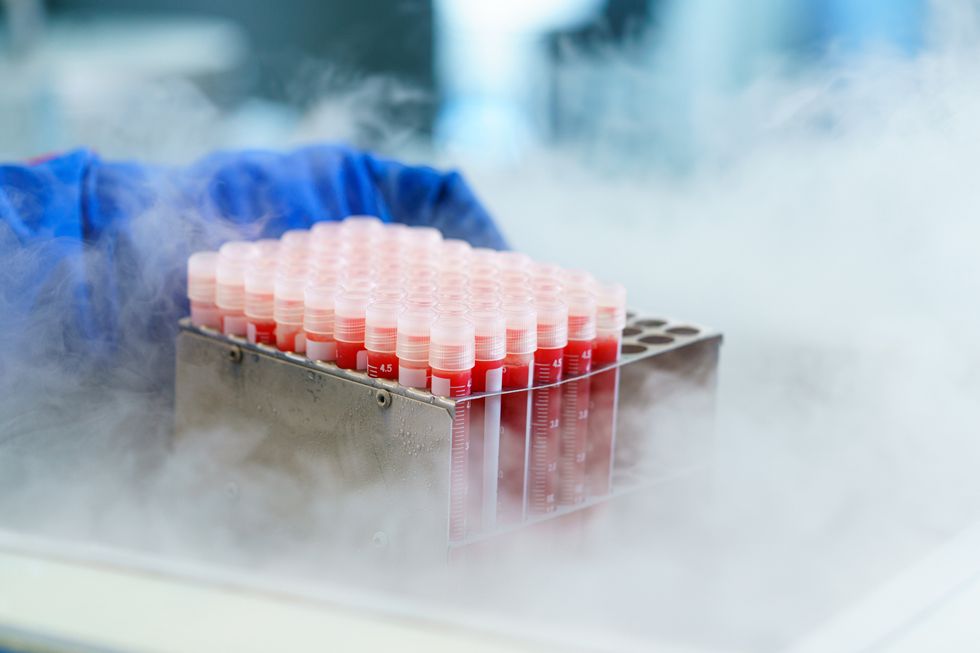
Globally, the cold chain packaging market is valued at over $15 billion and is expected to exceed $60 billion by 2033.
Adobe Stock
Freeze-drying, also called lyophilization, which is common for many vaccines, isn’t always an option. Many freeze-dried vaccines still need refrigeration, and even medicines approved for storage at ambient temperatures break down in the heat of sub-Saharan Africa. “Even in a freeze-dried state, biologics often will undergo partial rehydration and dehydration, which can be extremely damaging,” Boothby explains.
The cold chain is also very expensive to maintain. The global pharmaceutical cold chain packaging market is valued at more than $15 billion, and is expected to exceed $60 billion by 2033, according to a report by Future Market Insights. This cost is only expected to grow. According to the consulting company Accenture, the number of medicines that require the cold chain are expected to grow by 48 percent, compared to only 21 percent for non-cold-chain therapies.
Tardigrades to the rescue
Tardigrades are only about a millimeter long – with four legs and claws, and they lumber around like bears, thus their nickname – but could provide a big solution. “Tardigrades are unique in the animal kingdom, in that they’re able to survive a vast array of environmental insults,” says Boothby, the Wyoming professor. “They can be dried out, frozen, heated past the boiling point of water and irradiated at levels that are thousands of times more than you or I could survive.” So, his team is gradually unlocking tardigrades’ survival secrets and applying them to biologic pharmaceuticals to make them withstand both extreme heat and desiccation without losing efficacy.
Boothby’s team is focusing on blood clotting factor VIII, which, as the name implies, causes blood to clot. Currently, Boothby is concentrating on the so-called cytoplasmic abundant heat soluble (CAHS) protein family, which is found only in tardigrades, protecting them when they dry out. “We showed we can desiccate a biologic (blood clotting factor VIII, a key clotting component) in the presence of tardigrade proteins,” he says—without losing any of its effectiveness.
The researchers mixed the tardigrade protein with the blood clotting factor and then dried and rehydrated that substance six times without damaging the latter. This suggests that biologics protected with tardigrade proteins can withstand real-world fluctuations in humidity.
Furthermore, Boothby’s team found that when the blood clotting factor was dried and stabilized with tardigrade proteins, it retained its efficacy at temperatures as high as 95 degrees Celsius. That’s over 200 degrees Fahrenheit, much hotter than the 58 degrees Celsius that the World Meteorological Organization lists as the hottest recorded air temperature on earth. In contrast, without the protein, the blood clotting factor degraded significantly. The team published their findings in the journal Nature in March.
Although tardigrades rarely live more than 2.5 years, they have survived in a desiccated state for up to two decades, according to Animal Diversity Web. This suggests that tardigrades’ CAHS protein can protect biologic pharmaceuticals nearly indefinitely without refrigeration or freezing, which makes it significantly easier to deliver them in locations where refrigeration is unreliable or doesn’t exist.
The tricks of the tardigrades
Besides the CAHS proteins, tardigrades rely on a type of sugar called trehalose and some other protectants. So, rather than drying up, their cells solidify into rigid, glass-like structures. As that happens, viscosity between cells increases, thereby slowing their biological functions so much that they all but stop.
Now Boothby is combining CAHS D, one of the proteins in the CAHS family, with trehalose. He found that CAHS D and trehalose each protected proteins through repeated drying and rehydrating cycles. They also work synergistically, which means that together they might stabilize biologics under a variety of dry storage conditions.
“We’re finding the protective effect is not just additive but actually is synergistic,” he says. “We’re keen to see if something like that also holds true with different protein combinations.” If so, combinations could possibly protect against a variety of conditions.
Commercialization outlook
Before any stabilization technology for biologics can be commercialized, it first must be approved by the appropriate regulators. In the U.S., that’s the U.S. Food and Drug Administration. Developing a new formulation would require clinical testing and vast numbers of participants. So existing vaccines and biologics likely won’t be re-formulated for dry storage. “Many were developed decades ago,” says Fox. “They‘re not going to be reformulated into thermo-stable vaccines overnight,” if ever, he predicts.
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits.
Instead, this technology is most likely to be used for the new products and formulations that are just being created. New and improved vaccines will be the first to benefit. Good candidates include the plethora of mRNA vaccines, as well as biologic pharmaceuticals for neglected diseases that affect parts of the world where reliable cold chain is difficult to maintain, Boothby says. Some examples include new, more effective vaccines for malaria and for pathogenic Escherichia coli, which causes diarrhea.
Tallying up the benefits
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits. For instance, MenAfriVac, a meningitis vaccine (without tardigrade proteins) developed for sub-Saharan Africa, can be stored at up to 40 degrees Celsius for four days before administration. “If you have a few days where you don’t need to maintain the cold chain, it’s easier to transport vaccines to remote areas,” Fox says, where refrigeration does not exist or is not reliable.
Better health is an obvious benefit. MenAfriVac reduced suspected meningitis cases by 57 percent in the overall population and more than 99 percent among vaccinated individuals.
Lower healthcare costs are another benefit. One study done in Togo found that the cold chain-related costs increased the per dose vaccine price up to 11-fold. The ability to ship the vaccines using the usual cold chain, but transporting them at ambient temperatures for the final few days cut the cost in half.
There are environmental benefits, too, such as reducing fuel consumption and greenhouse gas emissions. Cold chain transports consume 20 percent more fuel than non-cold chain shipping, due to refrigeration equipment, according to the International Trade Administration.
A study by researchers at Johns Hopkins University compared the greenhouse gas emissions of the new, oral Vaxart COVID-19 vaccine (which doesn’t require refrigeration) with four intramuscular vaccines (which require refrigeration or freezing). While the Vaxart vaccine is still in clinical trials, the study found that “up to 82.25 million kilograms of CO2 could be averted by using oral vaccines in the U.S. alone.” That is akin to taking 17,700 vehicles out of service for one year.
Although tardigrades’ protective proteins won’t be a component of biologic pharmaceutics for several years, scientists are proving that this approach is viable. They are hopeful that a day will come when vaccines and biologics can be delivered anywhere in the world without needing refrigerators or freezers en route.
Here's how one doctor overcame extraordinary odds to help create the birth control pill
Dr. Percy Julian had so many personal and professional obstacles throughout his life, it’s amazing he was able to accomplish anything at all. But this hidden figure not only overcame these incredible obstacles, he also laid the foundation for the creation of the birth control pill.
Julian’s first obstacle was growing up in the Jim Crow-era south in the early part of the twentieth century, where racial segregation kept many African-Americans out of schools, libraries, parks, restaurants, and more. Despite limited opportunities and education, Julian was accepted to DePauw University in Indiana, where he majored in chemistry. But in college, Julian encountered another obstacle: he wasn’t allowed to stay in DePauw’s student housing because of segregation. Julian found lodging in an off-campus boarding house that refused to serve him meals. To pay for his room, board, and food, Julian waited tables and fired furnaces while he studied chemistry full-time. Incredibly, he graduated in 1920 as valedictorian of his class.
After graduation, Julian landed a fellowship at Harvard University to study chemistry—but here, Julian ran into yet another obstacle. Harvard thought that white students would resent being taught by Julian, an African-American man, so they withdrew his teaching assistantship. Julian instead decided to complete his PhD at the University of Vienna in Austria. When he did, he became one of the first African Americans to ever receive a PhD in chemistry.
Julian received offers for professorships, fellowships, and jobs throughout the 1930s, due to his impressive qualifications—but these offers were almost always revoked when schools or potential employers found out Julian was black. In one instance, Julian was offered a job at the Institute of Paper Chemistory in Appleton, Wisconsin—but Appleton, like many cities in the United States at the time, was known as a “sundown town,” which meant that black people weren’t allowed to be there after dark. As a result, Julian lost the job.
During this time, Julian became an expert at synthesis, which is the process of turning one substance into another through a series of planned chemical reactions. Julian synthesized a plant compound called physostigmine, which would later become a treatment for an eye disease called glaucoma.
In 1936, Julian was finally able to land—and keep—a job at Glidden, and there he found a way to extract soybean protein. This was used to produce a fire-retardant foam used in fire extinguishers to smother oil and gasoline fires aboard ships and aircraft carriers, and it ended up saving the lives of thousands of soldiers during World War II.
At Glidden, Julian found a way to synthesize human sex hormones such as progesterone, estrogen, and testosterone, from plants. This was a hugely profitable discovery for his company—but it also meant that clinicians now had huge quantities of these hormones, making hormone therapy cheaper and easier to come by. His work also laid the foundation for the creation of hormonal birth control: Without the ability to synthesize these hormones, hormonal birth control would not exist.
Julian left Glidden in the 1950s and formed his own company, called Julian Laboratories, outside of Chicago, where he manufactured steroids and conducted his own research. The company turned profitable within a year, but even so Julian’s obstacles weren’t over. In 1950 and 1951, Julian’s home was firebombed and attacked with dynamite, with his family inside. Julian often had to sit out on the front porch of his home with a shotgun to protect his family from violence.
But despite years of racism and violence, Julian’s story has a happy ending. Julian’s family was eventually welcomed into the neighborhood and protected from future attacks (Julian’s daughter lives there to this day). Julian then became one of the country’s first black millionaires when he sold his company in the 1960s.
When Julian passed away at the age of 76, he had more than 130 chemical patents to his name and left behind a body of work that benefits people to this day.
Therapies for Healthy Aging with Dr. Alexandra Bause
My guest today is Dr. Alexandra Bause, a biologist who has dedicated her career to advancing health, medicine and healthier human lifespans. Dr. Bause co-founded a company called Apollo Health Ventures in 2017. Currently a venture partner at Apollo, she's immersed in the discoveries underway in Apollo’s Venture Lab while the company focuses on assembling a team of investors to support progress. Dr. Bause and Apollo Health Ventures say that biotech is at “an inflection point” and is set to become a driver of important change and economic value.
Previously, Dr. Bause worked at the Boston Consulting Group in its healthcare practice specializing in biopharma strategy, among other priorities
She did her PhD studies at Harvard Medical School focusing on molecular mechanisms that contribute to cellular aging, and she’s also a trained pharmacist
In the episode, we talk about the present and future of therapeutics that could increase people’s spans of health, the benefits of certain lifestyle practice, the best use of electronic wearables for these purposes, and much more.
Dr. Bause is at the forefront of developing interventions that target the aging process with the aim of ensuring that all of us can have healthier, more productive lifespans.


