Tiny, tough “water bears” may help bring new vaccines and medicines to sub-Saharan Africa
Tardigrades can completely dehydrate and later rehydrate themselves, a survival trick that scientists are harnessing to preserve medicines in hot temperatures.
Microscopic tardigrades, widely considered to be some of the toughest animals on earth, can survive for decades without oxygen or water and are thought to have lived through a crash-landing on the moon. Also known as water bears, they survive by fully dehydrating and later rehydrating themselves – a feat only a few animals can accomplish. Now scientists are harnessing tardigrades’ talents to make medicines that can be dried and stored at ambient temperatures and later rehydrated for use—instead of being kept refrigerated or frozen.
Many biologics—pharmaceutical products made by using living cells or synthesized from biological sources—require refrigeration, which isn’t always available in many remote locales or places with unreliable electricity. These products include mRNA and other vaccines, monoclonal antibodies and immuno-therapies for cancer, rheumatoid arthritis and other conditions. Cooling is also needed for medicines for blood clotting disorders like hemophilia and for trauma patients.
Formulating biologics to withstand drying and hot temperatures has been the holy grail for pharmaceutical researchers for decades. It’s a hard feat to manage. “Biologic pharmaceuticals are highly efficacious, but many are inherently unstable,” says Thomas Boothby, assistant professor of molecular biology at University of Wyoming. Therefore, during storage and shipping, they must be refrigerated at 2 to 8 degrees Celsius (35 to 46 degrees Fahrenheit). Some must be frozen, typically at -20 degrees Celsius, but sometimes as low -90 degrees Celsius as was the case with the Pfizer Covid vaccine.
For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
The costly cold chain
The logistics network that ensures those temperature requirements are met from production to administration is called the cold chain. This cold chain network is often unreliable or entirely lacking in remote, rural areas in developing nations that have malfunctioning electrical grids. “Almost all routine vaccines require a cold chain,” says Christopher Fox, senior vice president of formulations at the Access to Advanced Health Institute. But when the power goes out, so does refrigeration, putting refrigerated or frozen medical products at risk. Consequently, the mRNA vaccines developed for Covid-19 and other conditions, as well as more traditional vaccines for cholera, tetanus and other diseases, often can’t be delivered to the most remote parts of the world.
To understand the scope of the challenge, consider this: In the U.S., more than 984 million doses of Covid-19 vaccine have been distributed so far. Each one needed refrigeration that, even in the U.S., proved challenging. Now extrapolate to all vaccines and the entire world. For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
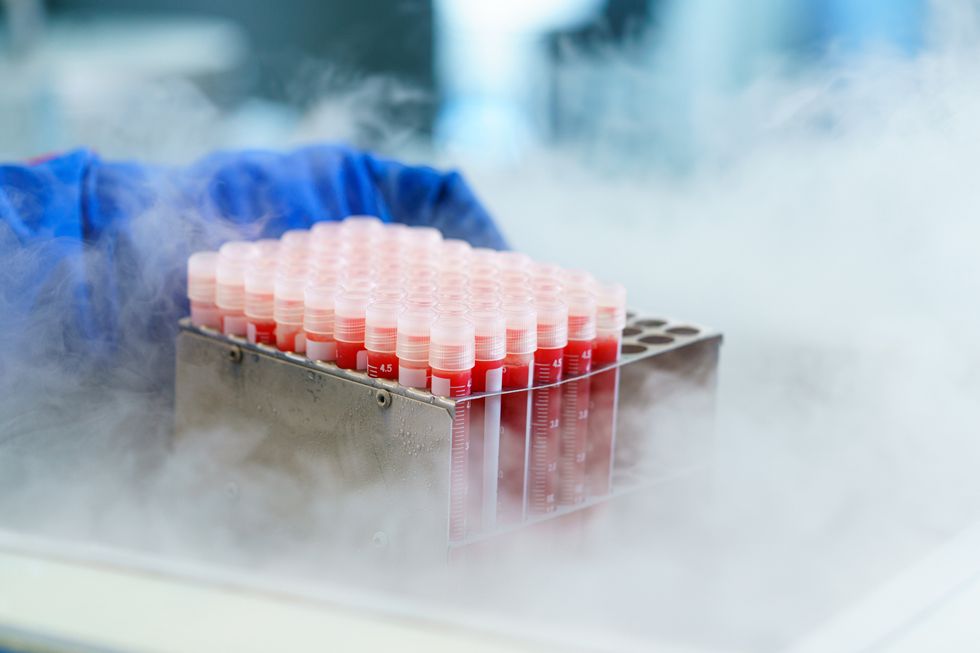
Globally, the cold chain packaging market is valued at over $15 billion and is expected to exceed $60 billion by 2033.
Adobe Stock
Freeze-drying, also called lyophilization, which is common for many vaccines, isn’t always an option. Many freeze-dried vaccines still need refrigeration, and even medicines approved for storage at ambient temperatures break down in the heat of sub-Saharan Africa. “Even in a freeze-dried state, biologics often will undergo partial rehydration and dehydration, which can be extremely damaging,” Boothby explains.
The cold chain is also very expensive to maintain. The global pharmaceutical cold chain packaging market is valued at more than $15 billion, and is expected to exceed $60 billion by 2033, according to a report by Future Market Insights. This cost is only expected to grow. According to the consulting company Accenture, the number of medicines that require the cold chain are expected to grow by 48 percent, compared to only 21 percent for non-cold-chain therapies.
Tardigrades to the rescue
Tardigrades are only about a millimeter long – with four legs and claws, and they lumber around like bears, thus their nickname – but could provide a big solution. “Tardigrades are unique in the animal kingdom, in that they’re able to survive a vast array of environmental insults,” says Boothby, the Wyoming professor. “They can be dried out, frozen, heated past the boiling point of water and irradiated at levels that are thousands of times more than you or I could survive.” So, his team is gradually unlocking tardigrades’ survival secrets and applying them to biologic pharmaceuticals to make them withstand both extreme heat and desiccation without losing efficacy.
Boothby’s team is focusing on blood clotting factor VIII, which, as the name implies, causes blood to clot. Currently, Boothby is concentrating on the so-called cytoplasmic abundant heat soluble (CAHS) protein family, which is found only in tardigrades, protecting them when they dry out. “We showed we can desiccate a biologic (blood clotting factor VIII, a key clotting component) in the presence of tardigrade proteins,” he says—without losing any of its effectiveness.
The researchers mixed the tardigrade protein with the blood clotting factor and then dried and rehydrated that substance six times without damaging the latter. This suggests that biologics protected with tardigrade proteins can withstand real-world fluctuations in humidity.
Furthermore, Boothby’s team found that when the blood clotting factor was dried and stabilized with tardigrade proteins, it retained its efficacy at temperatures as high as 95 degrees Celsius. That’s over 200 degrees Fahrenheit, much hotter than the 58 degrees Celsius that the World Meteorological Organization lists as the hottest recorded air temperature on earth. In contrast, without the protein, the blood clotting factor degraded significantly. The team published their findings in the journal Nature in March.
Although tardigrades rarely live more than 2.5 years, they have survived in a desiccated state for up to two decades, according to Animal Diversity Web. This suggests that tardigrades’ CAHS protein can protect biologic pharmaceuticals nearly indefinitely without refrigeration or freezing, which makes it significantly easier to deliver them in locations where refrigeration is unreliable or doesn’t exist.
The tricks of the tardigrades
Besides the CAHS proteins, tardigrades rely on a type of sugar called trehalose and some other protectants. So, rather than drying up, their cells solidify into rigid, glass-like structures. As that happens, viscosity between cells increases, thereby slowing their biological functions so much that they all but stop.
Now Boothby is combining CAHS D, one of the proteins in the CAHS family, with trehalose. He found that CAHS D and trehalose each protected proteins through repeated drying and rehydrating cycles. They also work synergistically, which means that together they might stabilize biologics under a variety of dry storage conditions.
“We’re finding the protective effect is not just additive but actually is synergistic,” he says. “We’re keen to see if something like that also holds true with different protein combinations.” If so, combinations could possibly protect against a variety of conditions.
Commercialization outlook
Before any stabilization technology for biologics can be commercialized, it first must be approved by the appropriate regulators. In the U.S., that’s the U.S. Food and Drug Administration. Developing a new formulation would require clinical testing and vast numbers of participants. So existing vaccines and biologics likely won’t be re-formulated for dry storage. “Many were developed decades ago,” says Fox. “They‘re not going to be reformulated into thermo-stable vaccines overnight,” if ever, he predicts.
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits.
Instead, this technology is most likely to be used for the new products and formulations that are just being created. New and improved vaccines will be the first to benefit. Good candidates include the plethora of mRNA vaccines, as well as biologic pharmaceuticals for neglected diseases that affect parts of the world where reliable cold chain is difficult to maintain, Boothby says. Some examples include new, more effective vaccines for malaria and for pathogenic Escherichia coli, which causes diarrhea.
Tallying up the benefits
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits. For instance, MenAfriVac, a meningitis vaccine (without tardigrade proteins) developed for sub-Saharan Africa, can be stored at up to 40 degrees Celsius for four days before administration. “If you have a few days where you don’t need to maintain the cold chain, it’s easier to transport vaccines to remote areas,” Fox says, where refrigeration does not exist or is not reliable.
Better health is an obvious benefit. MenAfriVac reduced suspected meningitis cases by 57 percent in the overall population and more than 99 percent among vaccinated individuals.
Lower healthcare costs are another benefit. One study done in Togo found that the cold chain-related costs increased the per dose vaccine price up to 11-fold. The ability to ship the vaccines using the usual cold chain, but transporting them at ambient temperatures for the final few days cut the cost in half.
There are environmental benefits, too, such as reducing fuel consumption and greenhouse gas emissions. Cold chain transports consume 20 percent more fuel than non-cold chain shipping, due to refrigeration equipment, according to the International Trade Administration.
A study by researchers at Johns Hopkins University compared the greenhouse gas emissions of the new, oral Vaxart COVID-19 vaccine (which doesn’t require refrigeration) with four intramuscular vaccines (which require refrigeration or freezing). While the Vaxart vaccine is still in clinical trials, the study found that “up to 82.25 million kilograms of CO2 could be averted by using oral vaccines in the U.S. alone.” That is akin to taking 17,700 vehicles out of service for one year.
Although tardigrades’ protective proteins won’t be a component of biologic pharmaceutics for several years, scientists are proving that this approach is viable. They are hopeful that a day will come when vaccines and biologics can be delivered anywhere in the world without needing refrigerators or freezers en route.
Sept. 13th Event: Delta, Vaccines, and Breakthrough Infections
A visualization of the virus that causes COVID-19.
This virtual event will convene leading scientific and medical experts to address the public's questions and concerns about COVID-19 vaccines, Delta, and breakthrough infections. Audience Q&A will follow the panel discussion. Your questions can be submitted in advance at the registration link.
DATE:
Monday, September 13th, 2021
12:30 p.m. - 1:45 p.m. EDT
REGISTER:

Dr. Amesh Adalja, M.D., FIDSA, Senior Scholar, Johns Hopkins Center for Health Security; Adjunct Assistant Professor, Johns Hopkins Bloomberg School of Public Health; Affiliate of the Johns Hopkins Center for Global Health. His work is focused on emerging infectious disease, pandemic preparedness, and biosecurity.

Dr. Nahid Bhadelia, M.D., MALD, Founding Director, Boston University Center for Emerging Infectious Diseases Policy and Research (CEID); Associate Director, National Emerging Infectious Diseases Laboratories (NEIDL), Boston University; Associate Professor, Section of Infectious Diseases, Boston University School of Medicine. She is an internationally recognized leader in highly communicable and emerging infectious diseases (EIDs) with clinical, field, academic, and policy experience in pandemic preparedness.

Dr. Akiko Iwasaki, Ph.D., Waldemar Von Zedtwitz Professor of Immunobiology and Molecular, Cellular and Developmental Biology and Professor of Epidemiology (Microbial Diseases), Yale School of Medicine; Investigator, Howard Hughes Medical Institute. Her laboratory researches how innate recognition of viral infections lead to the generation of adaptive immunity, and how adaptive immunity mediates protection against subsequent viral challenge.
 Dr. Marion Pepper, Ph.D., Associate Professor, Department of Immunology, University of Washington. Her lab studies how cells of the adaptive immune system, called CD4+ T cells and B cells, form immunological memory by visualizing their differentiation, retention, and function.
Dr. Marion Pepper, Ph.D., Associate Professor, Department of Immunology, University of Washington. Her lab studies how cells of the adaptive immune system, called CD4+ T cells and B cells, form immunological memory by visualizing their differentiation, retention, and function.
This event is the third of a four-part series co-hosted by Leaps.org, the Aspen Institute Science & Society Program, and the Sabin–Aspen Vaccine Science & Policy Group, with generous support from the Gordon and Betty Moore Foundation and the Howard Hughes Medical Institute.
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
Don't Panic Over Waning Antibodies. Here's Why.
A health care worker places a bandaid on the arm of a man who has just been vaccinated.
Since the Delta variant became predominant in the United States on July 7, both scientists and the media alike have been full of mixed messages ("breakthrough infections rare"; "breakthrough infections common"; "vaccines still work"; "vaccines losing their effectiveness") but – if we remember our infectious diseases history- one thing remains clear: immunity is the only way to get through a pandemic.
What Happened in the Past
The 1918 influenza pandemic was far the deadliest respiratory virus pandemic recorded in recent human history with over 50 million deaths (maybe even 100 million deaths, or 3% of the world's population) worldwide. Although they used some of the same measures we are using now (masks, distancing, event closures, as neither testing nor a vaccine existed back then), the deaths slowed only after enough of the population had either acquired immunity through natural infection or died. Indeed, the first influenza vaccine was not developed until 1942, more than 20 years later. As judged by the amount of suffering and death from 1918 influenza (and the deadly Delta surge in India in spring 2021), natural immunity is obviously a terrible way to get through a pandemic.
Similarly, measles was a highly transmissible respiratory virus that led to high levels of immunity among adults who were invariably exposed as children. However, measles led to deaths each year among the nonimmune until a vaccine was developed in 1963, largely restricting current measles outbreaks in the U.S. now to populations who decline to vaccinate. Smallpox also led to high levels of immunity through natural infection, which was often fatal. That's why unleashing smallpox on a largely nonimmune population in the New World was so deadly. Only an effective vaccine – and its administration worldwide, including among populations who declined smallpox vaccine at first via mandates – could control and then eventually eradicate smallpox from Earth.
Fully vaccinated people are already now able to generate some antibodies against all the variants we know of to date, thanks to their bank of memory B cells.
The Delta variant is extremely transmissible, making it unlikely we will ever eliminate or eradicate SARS-CoV-2. Even Australia, which had tried to maintain a COVID-zero nation with masks, distancing, lockdowns, testing and contact tracing before and during the vaccines, ended a strategy aimed at eliminating COVID-19 this week. But, luckily, since highly effective and safe vaccines were developed for COVID-19 less than a year after its advent on a nonimmune population and since vaccines are retaining their effectiveness against severe disease, we have a safe way out of the misery of this pandemic: more and more immunity. "Defanging" SARS-CoV-2 and stripping it of its ability to cause severe disease through immunity will relegate it to the fate of the other four circulating cold-causing coronaviruses, an inconvenience but not a world-stopper.
Immunity Is More Than Antibodies
When we say immunity, we have to be clear that we are talking about cellular immunity and immune memory, not only antibodies. This is a key point. Neutralizing antibodies, which prevent the virus from entering our cells, are generated by the vaccines. But those antibodies will necessarily wane over time since we cannot keep antibodies from every infection and vaccine we have ever seen in the bloodstream (or our blood would be thick as paste!). Vaccines with shorter intervals between doses (like Pfizer vaccines given 3 weeks apart) are likely to have their antibodies wane sooner than vaccines with longer intervals between doses (like Moderna), making mild symptomatic breakthroughs less likely with the Moderna vaccine than the Pfizer during our Delta surge, as a recent Mayo Clinic study showed.
Luckily, the vaccines generate B cells that get relegated to our memory banks and these memory B cells are able to produce high levels of antibodies to fight the virus if they see it again. Amazingly, these memory B cells will actually produce antibodies adapted against the COVID variants if they see a variant in the future, rather than the original antibodies directed against the ancestral strain. This is because memory B cells serve as a blueprint to make antibodies, like the blueprint of a house. If a house needs an extra column (or antibodies need to evolve to work against variants), the blueprint will oblige just as memory B cells will!
One problem with giving a 3rd dose of our current vaccines is that those antibodies won't be adapted towards the variants. Fully vaccinated people are already now able to generate some antibodies against all the variants we know of to date, thanks to their bank of memory B cells. In other words, no variant has evolved to date that completely evades our vaccines. Memory B cells, once generated by either natural infection or vaccination, should be long-lasting.
If memory B cells are formed by a vaccine, they should be as long-lasting as those from natural infection per various human studies. A 2008 Nature study found that survivors of the 1918 influenza pandemic were able to produce antibodies from memory B cells when exposed to the same influenza strain nine decades later. Of note, mild infections (such as the common cold from the cold-causing coronaviruses called 229E, NL63, OC43, and HKU1) may not reliably produce memory B cell immunity like more severe infections caused by SARS-CoV-2.
Right about now, you may be worrying about a super-variant that might yet emerge to evade all our hard-won immune responses. But most immunologists do not think this is very realistic because of T cells. How are T cells different from B Cells? While B cells are like the memory banks to produce antibodies when needed (helped by T cells), T cells will specifically amplify in response to a piece of the virus and help recruit cells to attack the pathogen directly. We likely have T cells to thank for the vaccine's incredible durability in protecting us against severe disease. Data from La Jolla Immunology Institute and UCSF show that the T cell response from the Pfizer vaccine is strong across all the variants.
Think of your spike protein as being comprised of 100 houses with a T cell there to cover each house (to protect you against severe disease). The variants have around 13 mutations along the spike protein so 13 of those T cells won't work, but there are over 80 T cells remaining to protect your "houses" or your body against severe disease.
Although these are theoretical numbers and we don't know exactly the number of T cell antigens (or "epitopes") across the spike protein, one review showed 1400 across the whole virus, with many of those in the spike protein. Another study showed that there were 87 epitopes across the spike protein to which T cells respond, and mutations in one of the variants (beta) took those down to 75. The virus cannot mutate indefinitely in its spike protein and still retain function. This is why it is unlikely we will get a variant that will evade the in-breadth, robust response of our T cells.
Where We Go From Here
So, what does this mean for getting through this pandemic? Immunity and more immunity. For those of us who are vaccinated, if we get exposed to the Delta variant, it will boost our immune response although the memory B cells might take 3-5 days to make new antibodies, which can leave us susceptible to a mild breakthrough infection. That's part of the reason the CDC put back masks for the vaccinated in late July. For those who are unvaccinated, immunity will be gained from Delta but often through terrible ways like severe disease.
The way for the U.S. to determine the need for 3rd shots among those who are not obviously immunocompromised, given the amazing immune memory generated by the vaccines among immunocompetent individuals, is to analyze the cases of the ~6000 individuals who have had severe breakthrough infections among the 171 million Americans fully vaccinated. Define their co-morbidities and age ranges, and boost those susceptible to severe infections (examples could include older people, those with co-morbidities, health care workers, and residents of long-term care facilities). This is an approach likely to be taken by the CDC's Advisory Committee on Immunization Practices.
If immunity is the only way to get through the pandemic and if variants are caused mostly by large populations being unvaccinated, there is not only a moral and ethical imperative but a practical imperative to vaccinate the world in order to keep us all safe. Immunocompetent Americans can boost their antibodies, which may enhance their ability to avoid mild breakthrough infections, but the initial shots still work well against the most important outcomes: hospitalizations and deaths.
There has been no randomized, controlled trial to assess whether three shots vs. two shots meaningfully improve those outcomes. While we ought to trust immune memory to get the immunocompetent in the United States through, we can hasten the end of this pandemic by providing surplus vaccines to poor countries to combat severe disease. Doing so would not only revitalize the role of the U.S. as a global health leader – it would save countless lives.