Tiny, tough “water bears” may help bring new vaccines and medicines to sub-Saharan Africa
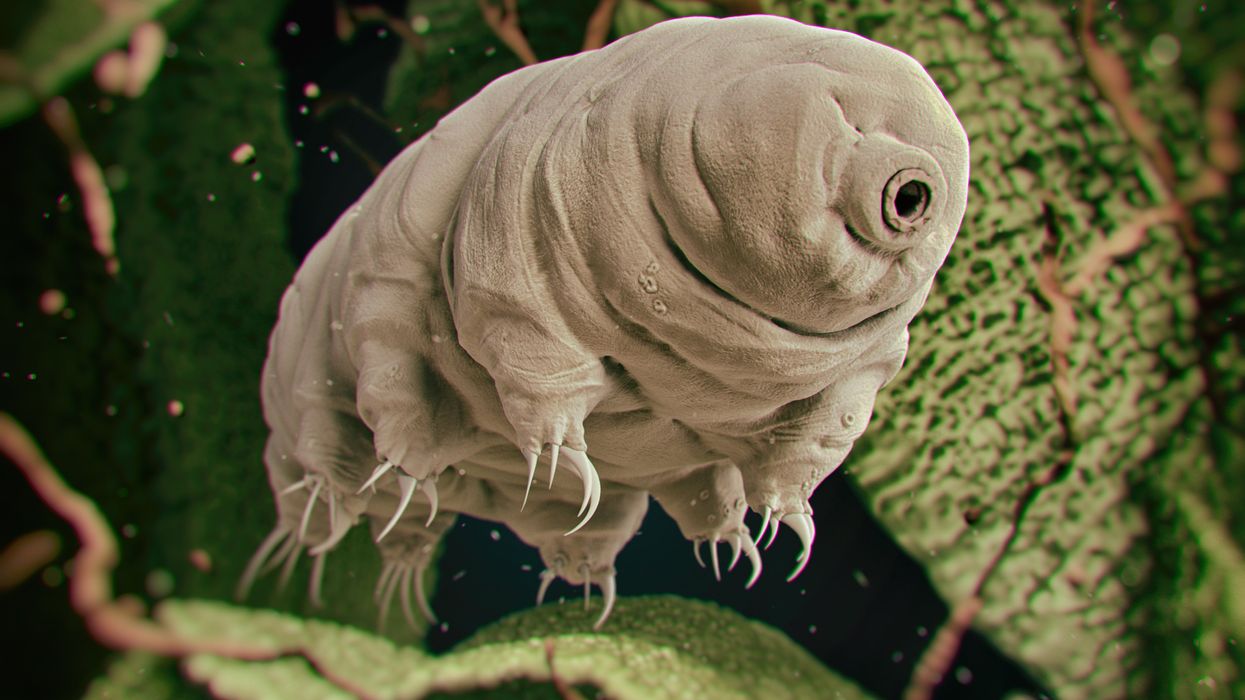
Tardigrades can completely dehydrate and later rehydrate themselves, a survival trick that scientists are harnessing to preserve medicines in hot temperatures.
Microscopic tardigrades, widely considered to be some of the toughest animals on earth, can survive for decades without oxygen or water and are thought to have lived through a crash-landing on the moon. Also known as water bears, they survive by fully dehydrating and later rehydrating themselves – a feat only a few animals can accomplish. Now scientists are harnessing tardigrades’ talents to make medicines that can be dried and stored at ambient temperatures and later rehydrated for use—instead of being kept refrigerated or frozen.
Many biologics—pharmaceutical products made by using living cells or synthesized from biological sources—require refrigeration, which isn’t always available in many remote locales or places with unreliable electricity. These products include mRNA and other vaccines, monoclonal antibodies and immuno-therapies for cancer, rheumatoid arthritis and other conditions. Cooling is also needed for medicines for blood clotting disorders like hemophilia and for trauma patients.
Formulating biologics to withstand drying and hot temperatures has been the holy grail for pharmaceutical researchers for decades. It’s a hard feat to manage. “Biologic pharmaceuticals are highly efficacious, but many are inherently unstable,” says Thomas Boothby, assistant professor of molecular biology at University of Wyoming. Therefore, during storage and shipping, they must be refrigerated at 2 to 8 degrees Celsius (35 to 46 degrees Fahrenheit). Some must be frozen, typically at -20 degrees Celsius, but sometimes as low -90 degrees Celsius as was the case with the Pfizer Covid vaccine.
For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
The costly cold chain
The logistics network that ensures those temperature requirements are met from production to administration is called the cold chain. This cold chain network is often unreliable or entirely lacking in remote, rural areas in developing nations that have malfunctioning electrical grids. “Almost all routine vaccines require a cold chain,” says Christopher Fox, senior vice president of formulations at the Access to Advanced Health Institute. But when the power goes out, so does refrigeration, putting refrigerated or frozen medical products at risk. Consequently, the mRNA vaccines developed for Covid-19 and other conditions, as well as more traditional vaccines for cholera, tetanus and other diseases, often can’t be delivered to the most remote parts of the world.
To understand the scope of the challenge, consider this: In the U.S., more than 984 million doses of Covid-19 vaccine have been distributed so far. Each one needed refrigeration that, even in the U.S., proved challenging. Now extrapolate to all vaccines and the entire world. For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
Globally, the cold chain packaging market is valued at over $15 billion and is expected to exceed $60 billion by 2033.
Adobe Stock
Freeze-drying, also called lyophilization, which is common for many vaccines, isn’t always an option. Many freeze-dried vaccines still need refrigeration, and even medicines approved for storage at ambient temperatures break down in the heat of sub-Saharan Africa. “Even in a freeze-dried state, biologics often will undergo partial rehydration and dehydration, which can be extremely damaging,” Boothby explains.
The cold chain is also very expensive to maintain. The global pharmaceutical cold chain packaging market is valued at more than $15 billion, and is expected to exceed $60 billion by 2033, according to a report by Future Market Insights. This cost is only expected to grow. According to the consulting company Accenture, the number of medicines that require the cold chain are expected to grow by 48 percent, compared to only 21 percent for non-cold-chain therapies.
Tardigrades to the rescue
Tardigrades are only about a millimeter long – with four legs and claws, and they lumber around like bears, thus their nickname – but could provide a big solution. “Tardigrades are unique in the animal kingdom, in that they’re able to survive a vast array of environmental insults,” says Boothby, the Wyoming professor. “They can be dried out, frozen, heated past the boiling point of water and irradiated at levels that are thousands of times more than you or I could survive.” So, his team is gradually unlocking tardigrades’ survival secrets and applying them to biologic pharmaceuticals to make them withstand both extreme heat and desiccation without losing efficacy.
Boothby’s team is focusing on blood clotting factor VIII, which, as the name implies, causes blood to clot. Currently, Boothby is concentrating on the so-called cytoplasmic abundant heat soluble (CAHS) protein family, which is found only in tardigrades, protecting them when they dry out. “We showed we can desiccate a biologic (blood clotting factor VIII, a key clotting component) in the presence of tardigrade proteins,” he says—without losing any of its effectiveness.
The researchers mixed the tardigrade protein with the blood clotting factor and then dried and rehydrated that substance six times without damaging the latter. This suggests that biologics protected with tardigrade proteins can withstand real-world fluctuations in humidity.
Furthermore, Boothby’s team found that when the blood clotting factor was dried and stabilized with tardigrade proteins, it retained its efficacy at temperatures as high as 95 degrees Celsius. That’s over 200 degrees Fahrenheit, much hotter than the 58 degrees Celsius that the World Meteorological Organization lists as the hottest recorded air temperature on earth. In contrast, without the protein, the blood clotting factor degraded significantly. The team published their findings in the journal Nature in March.
Although tardigrades rarely live more than 2.5 years, they have survived in a desiccated state for up to two decades, according to Animal Diversity Web. This suggests that tardigrades’ CAHS protein can protect biologic pharmaceuticals nearly indefinitely without refrigeration or freezing, which makes it significantly easier to deliver them in locations where refrigeration is unreliable or doesn’t exist.
The tricks of the tardigrades
Besides the CAHS proteins, tardigrades rely on a type of sugar called trehalose and some other protectants. So, rather than drying up, their cells solidify into rigid, glass-like structures. As that happens, viscosity between cells increases, thereby slowing their biological functions so much that they all but stop.
Now Boothby is combining CAHS D, one of the proteins in the CAHS family, with trehalose. He found that CAHS D and trehalose each protected proteins through repeated drying and rehydrating cycles. They also work synergistically, which means that together they might stabilize biologics under a variety of dry storage conditions.
“We’re finding the protective effect is not just additive but actually is synergistic,” he says. “We’re keen to see if something like that also holds true with different protein combinations.” If so, combinations could possibly protect against a variety of conditions.
Commercialization outlook
Before any stabilization technology for biologics can be commercialized, it first must be approved by the appropriate regulators. In the U.S., that’s the U.S. Food and Drug Administration. Developing a new formulation would require clinical testing and vast numbers of participants. So existing vaccines and biologics likely won’t be re-formulated for dry storage. “Many were developed decades ago,” says Fox. “They‘re not going to be reformulated into thermo-stable vaccines overnight,” if ever, he predicts.
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits.
Instead, this technology is most likely to be used for the new products and formulations that are just being created. New and improved vaccines will be the first to benefit. Good candidates include the plethora of mRNA vaccines, as well as biologic pharmaceuticals for neglected diseases that affect parts of the world where reliable cold chain is difficult to maintain, Boothby says. Some examples include new, more effective vaccines for malaria and for pathogenic Escherichia coli, which causes diarrhea.
Tallying up the benefits
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits. For instance, MenAfriVac, a meningitis vaccine (without tardigrade proteins) developed for sub-Saharan Africa, can be stored at up to 40 degrees Celsius for four days before administration. “If you have a few days where you don’t need to maintain the cold chain, it’s easier to transport vaccines to remote areas,” Fox says, where refrigeration does not exist or is not reliable.
Better health is an obvious benefit. MenAfriVac reduced suspected meningitis cases by 57 percent in the overall population and more than 99 percent among vaccinated individuals.
Lower healthcare costs are another benefit. One study done in Togo found that the cold chain-related costs increased the per dose vaccine price up to 11-fold. The ability to ship the vaccines using the usual cold chain, but transporting them at ambient temperatures for the final few days cut the cost in half.
There are environmental benefits, too, such as reducing fuel consumption and greenhouse gas emissions. Cold chain transports consume 20 percent more fuel than non-cold chain shipping, due to refrigeration equipment, according to the International Trade Administration.
A study by researchers at Johns Hopkins University compared the greenhouse gas emissions of the new, oral Vaxart COVID-19 vaccine (which doesn’t require refrigeration) with four intramuscular vaccines (which require refrigeration or freezing). While the Vaxart vaccine is still in clinical trials, the study found that “up to 82.25 million kilograms of CO2 could be averted by using oral vaccines in the U.S. alone.” That is akin to taking 17,700 vehicles out of service for one year.
Although tardigrades’ protective proteins won’t be a component of biologic pharmaceutics for several years, scientists are proving that this approach is viable. They are hopeful that a day will come when vaccines and biologics can be delivered anywhere in the world without needing refrigerators or freezers en route.
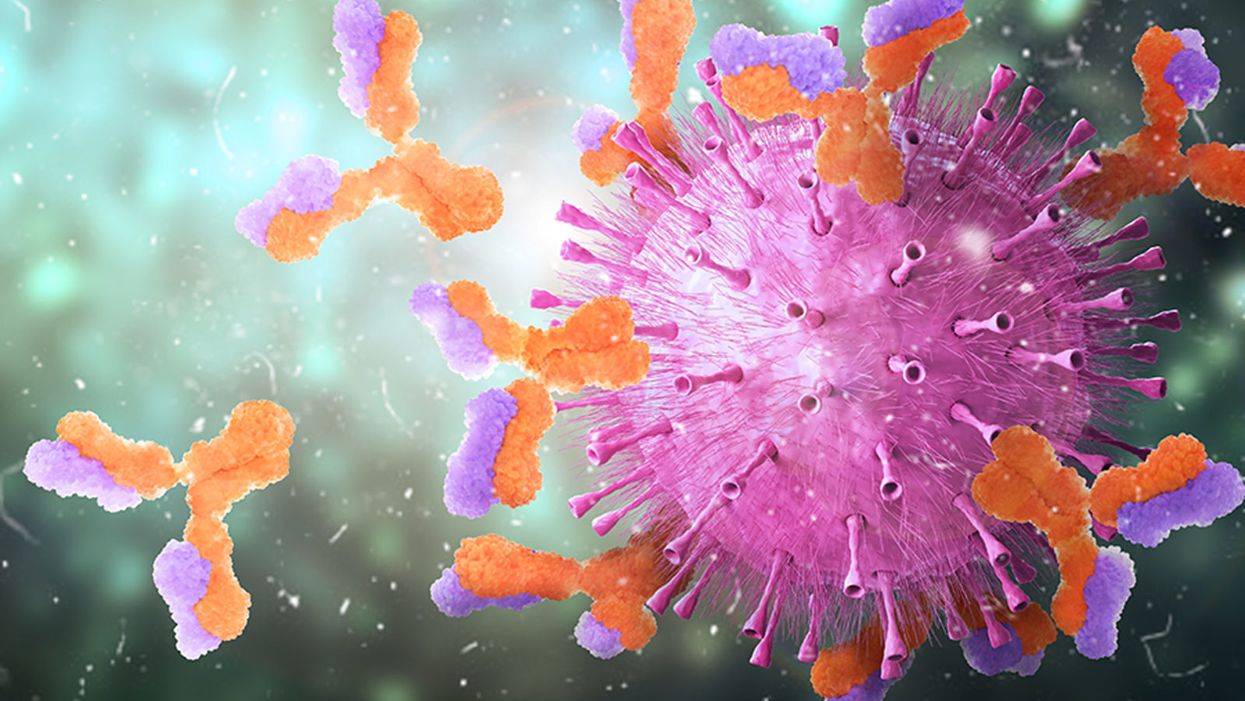
Harvard Researchers Are Using a Breakthrough Tool to Find the Antibodies That Best Knock Out the Coronavirus
Knowing which antibodies bind best to the coronavirus can help guide new medicines and vaccine development.
To find a cure for a deadly infectious disease in the 1995 medical thriller Outbreak, scientists extract the virus's antibodies from its original host—an African monkey.
"When a person is infected, the immune system makes antibodies kind of blindly."
The antibodies prevent the monkeys from getting sick, so doctors use these antibodies to make the therapeutic serum for humans. With SARS-CoV-2, the original hosts might be bats or pangolins, but scientists don't have access to either, so they are turning to the humans who beat the virus.
Patients who recovered from COVID-19 are valuable reservoirs of viral antibodies and may help scientists develop efficient therapeutics, says Stephen J. Elledge, professor of genetics and medicine at Harvard Medical School in Boston. Studying the structure of the antibodies floating in their blood can help understand what their immune systems did right to kill the pathogen.
When viruses invade the body, the immune system builds antibodies against them. The antibodies work like Velcro strips—they use special spots on their surface called paratopes to cling to the specific spots on the viral shell called epitopes. Once the antibodies circulating in the blood find their "match," they cling on to the virus and deactivate it.
But that process is far from simple. The epitopes and paratopes are built of various peptides that have complex shapes, are folded in specific ways, and may carry an electrical charge that repels certain molecules. Only when all of these parameters match, an antibody can get close enough to a viral particle—and shut it out.
So the immune system forges many different antibodies with varied parameters in hopes that some will work. "When a person is infected, the immune system makes antibodies kind of blindly," Elledge says. "It's doing a shotgun approach. It's not sure which ones will work, but it knows once it's made a good one that works."
Elledge and his team want to take the guessing out of the process. They are using their home-built tool VirScan to comb through the blood samples of the recovered COVID-19 patients to see what parameters the efficient antibodies should have. First developed in 2015, the VirScan has a library of epitopes found on the shells of viruses known to afflict humans, akin to a database of criminals' mug shots maintained by the police.
Originally, VirScan was meant to reveal which pathogens a person overcame throughout a lifetime, and could identify over 1,000 different strains of viruses and bacteria. When the team ran blood samples against the VirScan's library, the tool would pick out all the "usual suspects." And unlike traditional blood tests called ELISA, which can only detect one pathogen at a time, VirScan can detect all of them at once. Now, the team has updated VirScan with the SARS-CoV-2 "mug shot" and is beginning to test which antibodies from the recovered patients' blood will bind to them.
Knowing which antibodies bind best can also help fine-tune vaccines.
Obtaining blood samples was a challenge that caused some delays. "So far most of the recovered patients have been in China and those samples are hard to get," Elledge says. It also takes a person five to 10 days to develop antibodies, so the blood must be drawn at the right time during the illness. If a person is asymptomatic, it's hard to pinpoint the right moment. "We just got a couple of blood samples so we are testing now," he said. The team hopes to get some results very soon.
Elucidating the structure of efficient antibodies can help create therapeutics for COVID-19. "VirScan is a powerful technology to study antibody responses," says Harvard Medical School professor Dan Barouch, who also directs the Center for Virology and Vaccine Research. "A detailed understanding of the antibody responses to COVID-19 will help guide the design of next-generation vaccines and therapeutics."
For example, scientists can synthesize antibodies to specs and give them to patients as medicine. Once vaccines are designed, medics can use VirScan to see if those vaccinated again COVID-19 generate the necessary antibodies.
Knowing which antibodies bind best can also help fine-tune vaccines. Sometimes, viruses cause the immune system to generate antibodies that don't deactivate it. "We think the virus is trying to confuse the immune system; it is its business plan," Elledge says—so those unhelpful antibodies shouldn't be included in vaccines.
More importantly, VirScan can also tell which people have developed immunity to SARS-CoV-2 and can return to their workplaces and businesses, which is crucial to restoring the economy. Knowing one's immunity status is especially important for doctors working on the frontlines, Elledge notes. "The resistant ones can intubate the sick."
Lina Zeldovich has written about science, medicine and technology for Popular Science, Smithsonian, National Geographic, Scientific American, Reader’s Digest, the New York Times and other major national and international publications. A Columbia J-School alumna, she has won several awards for her stories, including the ASJA Crisis Coverage Award for Covid reporting, and has been a contributing editor at Nautilus Magazine. In 2021, Zeldovich released her first book, The Other Dark Matter, published by the University of Chicago Press, about the science and business of turning waste into wealth and health. You can find her on http://linazeldovich.com/ and @linazeldovich.
Defenders of civil liberties fear that the erosion of privacy will become a long-term consequence of the pandemic.
As countries around the world combat the coronavirus outbreak, governments that already operated sophisticated surveillance programs are ramping up the tracking of their citizens.
"The potential for invasions of privacy, abuse, and stigmatization is enormous."
Countries like China, South Korea, Israel, Singapore and others are closely monitoring citizens to track the spread of the virus and prevent further infections, and policymakers in the United States have proposed similar steps. These shifts in policy have civil liberties defenders alarmed, as history has shown increases in surveillance tend to stick around after an emergency is over.
In China, where the virus originated and surveillance is already ubiquitous, the government has taken measures like having people scan a QR code and answer questions about their health and travel history to enter their apartment building. The country has also increased the tracking of cell phones, encouraged citizens to report people who appear to be sick, utilized surveillance drones, and developed facial recognition that can identify someone even if they're wearing a mask.
In Israel, the government has begun tracking people's cell phones without a court order under a program that was initially meant to counter terrorism. Singapore has also been closely tracking people's movements using cell phone data. In South Korea, the government has been monitoring citizens' credit card and cell phone data and has heavily utilized facial recognition to combat the spread of the coronavirus.
Here at home, the United States government and state governments have been using cell phone data to determine where people are congregating. White House senior adviser Jared Kushner's task force to combat the coronavirus outbreak has proposed using cell phone data to track coronavirus patients. Cities around the nation are also using surveillance drones to maintain social distancing orders. Companies like Apple and Google that work closely with the federal government are currently developing systems to track Americans' cell phones.
All of this might sound acceptable if you're worried about containing the outbreak and getting back to normal life, but as we saw when the Patriot Act was passed in 2001 in the wake of the 9/11 terrorist attacks, expansions of the surveillance state can persist long after the emergency that seemed to justify them.
Jay Stanley, senior policy analyst with the ACLU Speech, Privacy, and Technology Project, says that this public health emergency requires bold action, but he worries that actions may be taken that will infringe on our privacy rights.
"This is an extraordinary crisis that justifies things that would not be justified in ordinary times, but we, of course, worry that any such things would be made permanent," Stanley says.
Stanley notes that the 9/11 situation was different from this current situation because we still face the threat of terrorism today, and we always will. The Patriot Act was a response to that threat, even if it was an extreme response. With this pandemic, it's quite possible we won't face something like this again for some time.
"We know that for the last seven or eight decades, we haven't seen a microbe this dangerous become a pandemic, and it's reasonable to expect it's not going to be happening for a while afterward," Stanley says. "We do know that when a vaccine is produced and is produced widely enough, the COVID crisis will be over. This does, unlike 9/11, have a definitive ending."
The ACLU released a white paper last week outlining the problems with using location data from cell phones and how policymakers should proceed when they discuss the usage of surveillance to combat the outbreak.
"Location data contains an enormously invasive and personal set of information about each of us, with the potential to reveal such things as people's social, sexual, religious, and political associations," they wrote. "The potential for invasions of privacy, abuse, and stigmatization is enormous. Any uses of such data should be temporary, restricted to public health agencies and purposes, and should make the greatest possible use of available techniques that allow for privacy and anonymity to be protected, even as the data is used."
"The first thing you need to combat pervasive surveillance is to know that it's occurring."
Sara Collins, policy counsel at the digital rights organization Public Knowledge, says that one of the problems with the current administration is that there's not much transparency, so she worries surveillance could be increased without the public realizing it.
"You'll often see the White House come out with something—that they're going to take this action or an agency just says they're going to take this action—and there's no congressional authorization," Collins says. "There's no regulation. There's nothing there for the public discourse."
Collins says it's almost impossible to protect against infringements on people's privacy rights if you don't actually know what kind of surveillance is being done and at what scale.
"I think that's very concerning when there's no accountability and no way to understand what's actually happening," Collins says. "The first thing you need to combat pervasive surveillance is to know that it's occurring."
We should also be worried about corporate surveillance, Collins says, because the tech companies that keep track of our data work closely with the government and do not have a good track record when it comes to protecting people's privacy. She suspects these companies could use the coronavirus outbreak to defend the kind of data collection they've been engaging in for years.
Collins stresses that any increase in surveillance should be transparent and short-lived, and that there should be a limit on how long people's data can be kept. Otherwise, she says, we're risking an indefinite infringement on privacy rights. Her organization will be keeping tabs as the crisis progresses.
It's not that we shouldn't avail ourselves of modern technology to fight the pandemic. Indeed, once lockdown restrictions are gradually lifted, public health officials must increase their ability to isolate new cases and trace, test, and quarantine contacts.
But tracking the entire populace "Big Brother"-style is not the ideal way out of the crisis. Last week, for instance, a group of policy experts -- including former FDA Commissioner Scott Gottlieb -- published recommendations for how to achieve containment. They emphasized the need for widespread diagnostic and serologic testing as well as rapid case-based interventions, among other measures -- and they, too, were wary of pervasive measures to follow citizens.
The group wrote: "Improved capacity [for timely contact tracing] will be most effective if coordinated with health care providers, health systems, and health plans and supported by timely electronic data sharing. Cell phone-based apps recording proximity events between individuals are unlikely to have adequate discriminating ability or adoption to achieve public health utility, while introducing serious privacy, security, and logistical concerns."
The bottom line: Any broad increases in surveillance should be carefully considered before we go along with them out of fear. The Founders knew that privacy is integral to freedom; that's why they wrote the Fourth Amendment to protect it, and that right shouldn't be thrown away because we're in an emergency. Once you lose a right, you don't tend to get it back.