The Biggest Challenge for a COVID-19 Vaccine

As scientists race to develop a safe and effective vaccine, companies and governments must figure out how to distribute affordable doses all over the world as fast as possible.
Although no one has conducted a survey on the topic, it's safe to say that a single hope unites much of humanity at the present moment: the prospect of a vaccine for COVID-19, which has infected more than 9 million people worldwide, killed nearly 500,000, and sent the global economy into a tailspin since it first appeared in China last December.
"We've never delivered something to every corner of the world before."
Scientists are racing to make that vision a reality. As of this writing, 11 vaccine candidates are in clinical trials and over 100 others are in preclinical development, in a dozen countries. Pointing to new technology and compressed testing protocols, experts predict a winner could emerge in 12 to 18 months—a fraction of the four years it took to develop the previous record-holder, the mumps vaccine, in the 1960s. Teams at Oxford University and Boston-based Moderna Therapeutics say they could have a product ready even sooner, if the formulas they're testing prove safe and effective. A just-announced White House initiative, Operation Warp Speed, aims to fast-track multiple candidates, with the goal of delivering 100 million doses in November and another 200 million by January 2021.
These timetables could prove wildly over-optimistic. But even if the best-case scenario comes true, and a viable COVID-19 vaccine emerges this fall, a gargantuan challenge remains: getting the shot to everyone who needs it. Epidemiologists figure that at least 70 percent of Earth's population—or 5.6 billion people—would have to be inoculated to achieve "herd immunity," in which each person who catches the disease passes it to less than one other individual. "In order to stop the pandemic, we need to make the vaccine available to almost every person on the planet," Microsoft co-founder Bill Gates blogged in April, as his foundation pledged $300 million to the effort. "We've never delivered something to every corner of the world before."
The difficulties are partly logistical, partly political, and largely a combination of the two. Overcoming those obstacles will require unprecedented cooperation among national governments, international organizations, and profit-minded corporations—in an era when nationalist rivalries are rampant and global leadership is up for grabs.
That may be tougher than developing the vaccine itself.
Logistical Conundrums
Manufacturing and distributing billions of vaccine doses would be a daunting task even in the most harmonious of times. Take the packaging problem. The vaccines under development range from old-school (based on inactivated or weakened viruses) to cutting-edge (using snippets of RNA or DNA to train the immune system to attack the invader). Some may work better than others for different patient groups—the young versus the elderly, for example. All, however, must be stored in vials and administered with syringes.
Among the handful of U.S. companies that manufacture such products, many must import the special glass tubing for vials, as well as the polypropylene for syringe barrels and the rubber or silicone for stoppers and plungers. These materials are commonly sourced from China and India, where lockdowns and export bans restrict supply. Rick Bright, the ousted director of the federal Biomedical Advanced Research and Development Authority (BARDA), claims he was ignored when he warned the Trump Administration that a medical-glass shortage was looming before the coronavirus crisis hit; securing enough to vaccinate 300 million Americans, he told Congress in May, could take up to two years.
Getting the vaccine to poorer countries presents further hurdles. To begin with, there's refrigeration. Inactivated or live vaccines must be kept between 2 and 8 degrees Centigrade (or 35 to 46 degrees Fahrenheit); RNA vaccines typically require much colder temperatures—as low as -80 degrees. This makes storage and transport challenging in parts of the world that lack reliable electricity. DNA vaccines don't need cold storage, but (like RNA vaccines) they remain experimental. They've never been approved to treat any human disease.
Tracking vaccine distribution is another conundrum for low- to-middle-income countries. "Supply chain management is really about information," explains Rebecca Weintraub, assistant professor of global health and social medicine at Harvard Medical School and director of the Better Evidence project at Harvard's Ariadne Labs. "It's about leveraging data to determine demand, predict behavior, and understand the flow of the product itself." Systems for collecting and analyzing such data can be hard to find in poorer regions, she notes. What's more, many people in those areas lack any type of ID card, making it difficult to know who has or hasn't received a vaccine.
Weintraub and two coauthors published an article in April in the Harvard Business Review, suggesting solutions to these and other developing-world problems: solar direct-drive refrigerators, app-based data-capture systems, biometric digital IDs. But such measures—not to mention purchasing adequate supplies of vaccine—would require massive funding.
And that's where the logistical begins to overlap with the political.
Global Access Versus "Vaccine Nationalism"
An array of institutions have already begun laying the groundwork for achieving worldwide, equitable access to COVID-19 vaccines. In February, the World Bank and the Norway-based Coalition for Epidemic Preparedness Innovations (CEPI) cohosted a global consultation on funding vaccine development and manufacturing. In late April, the World Health Organization (WHO), in collaboration with dozens of governments, nonprofits, and industry leaders, launched a program called the Access to COVID-19 Tools Accelerator to expedite such efforts.
Soon afterward, the European Union, along with six countries and the Bill and Melinda Gates Foundation, held a Coronavirus Global Response telethon that raised $8 billion to support Gavi, the Vaccine Alliance—a public-private partnership that subsidizes immunization in low-income countries. The United States and Russia, however, chose not to participate.
This snub by the world's remaining superpower and one of its principal challengers worried many observers. "I am concerned about what I call vaccine nationalism," CEPI executive director Richard Hatchett told the Los Angeles Times. "That's the tension between obligations elected leaders will feel to protect the lives of their citizens" versus the imperative for global sharing.
Some signs point to a possible rerun of the hoarding that accompanied the 2009 H1N1 influenza pandemic, when wealthy nations bought up virtually all vaccine supplies—denying them to poorer countries, and sometimes to one another. Operation Warp Speed has declared an "America First" policy for any vaccine arising from its efforts. Pharma giant Sanofi recently suggested that it would take a similar approach, since the U.S. was first to fund the company's COVID-19 research. (Sanofi's CEO backtracked after officials in France, where the firm is headquartered, protested.) The Oxford group, which is partnering with British-based drug maker AstraZeneca, intends to prioritize Great Britain.
Yet momentum is building for more generous strategies as well. In May, over 100 current and former world leaders, along with prominent economists and public health experts, issued an open letter calling for a "people's vaccine" for COVID-19, which would be patent-free, distributed globally, and available to all countries free of charge. At the WHO's annual World Health Assembly, all 194 member states accepted a resolution urging that vaccines for the disease be made available as a "global public good"—though the U.S. dissociated itself from a clause proposing a patent pool to keep costs down, which it argued might disincentivize "innovators who will be essential to the solutions the whole world needs."
Gavi, for its part, plans to launch a mechanism designed to encourage those innovators while promoting accessibility: an advance market commitment, in which countries pledge to purchase a vaccine, with no money down. Future contributions will be based on the value of the product to their health systems and their ability to pay.
"It's essential to realize that a threat anywhere is a threat everywhere."
A few private-sector players are stepping up, too. U.S.-based Johnson & Johnson, which has received nearly half a billion dollars from the federal government for COVID-19 vaccine research, has promised to provide up to 900 million doses on a not-for-profit basis, if its trials pan out. Other companies have agreed to produce vaccines on a "cost-plus" basis, with a smaller-than-usual profit margin.
How Sharing Can Pay Off
No one knows how all this will work out if and when a vaccine becomes available. (Another wild card: Trump has announced that he is cutting U.S. ties to the WHO over its alleged favoritism toward China, which could hobble the agency's ability to coordinate distribution -- though uncertainty remains about the process of withdrawal and reversing course may still be possible.) To public health experts, however, it's clear that ensuring accessibility is not just a matter of altruism.
"A historic example is smallpox," Rebecca Weintraub observes. "When it kept getting reintroduced into high-income countries from low-income countries, the rich countries realized it was worth investing in the vaccine for countries that couldn't afford it." After a two-decade campaign led by the WHO, the last case of this ancient scourge was diagnosed in 1977.
Conversely, vaccine nationalism doesn't just hurt poor countries. During the H1N1 pandemic, which killed an estimated 284,000 people worldwide, production problems led to shortages in the United States. But Australia stopped a domestic manufacturer from exporting doses to the U.S until all Aussies had been immunized.
Such considerations, Weintraub believes, might help convince even the most reluctant rich-country leaders that an accessible vaccine—if deployed in an epidemiologically targeted way—would serve both the greater good and the national interest. "I suspect the pressures put on our politicians to act globally will be significant," she says.
Other analysts share her guarded optimism. Kelly Moore, who teaches health policy at Vanderbilt University Medical Center, oversaw Tennessee's immunization programs for more than a decade, and later became a member of the Sabin-Aspen Vaccine Science & Policy Group—a panel of international experts that in 2019 released a report titled "Accelerating the Development of a Universal Influenza Vaccine." The 117-page document provided a road map toward a long-sought goal: creating a flu shot that doesn't need to be reformulated each year to target changing viral strains.
"One lesson we learned was that it's crucial to deploy financial resources in a systematic way to support coordination among laboratories that would typically be competitors," Moore says. And that, she adds, is happening with COVID-19, despite nationalist frictions: scientists from Sanofi joining forces with those at rival GSK; researchers at other companies allying with teams at government laboratories; university labs worldwide sharing data across borders. "I have been greatly encouraged to see the amount of global collaboration involved in this enterprise. Partners are working together who would normally never be partners."
For Moore, whose 77-year-old mother survived a bout with the disease, the current pandemic has hit close to home. "It's essential to realize that a threat anywhere is a threat everywhere," she says. "Morally and ethically, we have a tremendous obligation to ensure that the most vulnerable have access to an affordable vaccine, irrespective of where they live."
[Editor's Note: This article was originally published on June 8th, 2020 as part of a standalone magazine called GOOD10: The Pandemic Issue. Produced as a partnership among LeapsMag, The Aspen Institute, and GOOD, the magazine is available for free online. For this reprinting of the article, we have updated the latest statistics on COVID-19 and related global news.]
CORRECTION: A sentence about DNA vaccines incorrectly stated that they require cold storage, like RNA vaccines. The error has been fixed.
Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
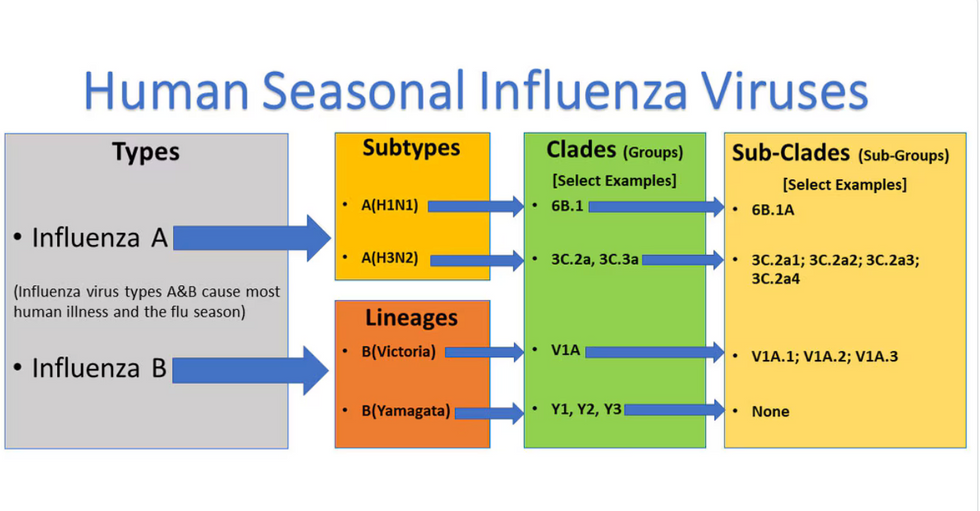
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.

