Why you should (virtually) care

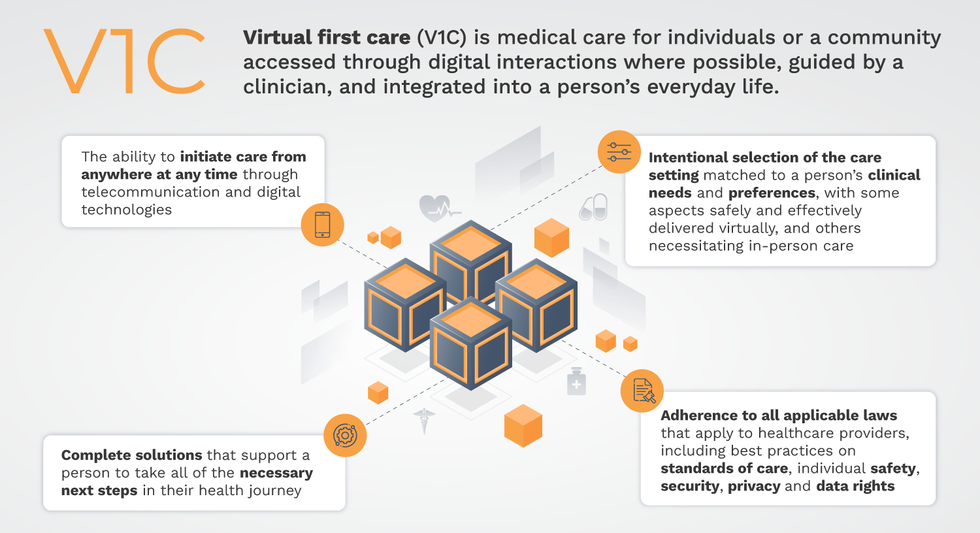
Virtual-first care, or V1C, could increase the quality of healthcare and make it more patient-centric by letting patients combine in-person visits with virtual options such as video for seeing their care providers.
As the pandemic turns endemic, healthcare providers have been eagerly urging patients to return to their offices to enjoy the benefits of in-person care.
But wait.
The last two years have forced all sorts of organizations to be nimble, adaptable and creative in how they work, and this includes healthcare providers’ efforts to maintain continuity of care under the most challenging of conditions. So before we go back to “business as usual,” don’t we owe it to those providers and ourselves to admit that business as usual did not work for most of the people the industry exists to help? If we’re going to embrace yet another period of change – periods that don’t happen often in our complex industry – shouldn’t we first stop and ask ourselves what we’re trying to achieve?
Certainly, COVID has shown that telehealth can be an invaluable tool, particularly for patients in rural and underserved communities that lack access to specialty care. It’s also become clear that many – though not all – healthcare encounters can be effectively conducted from afar. That said, the telehealth tactics that filled the gap during the pandemic were largely stitched together substitutes for existing visit-based workflows: with offices closed, patients scheduled video visits for help managing the side effects of their blood pressure medications or to see their endocrinologist for a quarterly check-in. Anyone whose children slogged through the last year or two of remote learning can tell you that simply virtualizing existing processes doesn’t necessarily improve the experience or the outcomes!
But what if our approach to post-pandemic healthcare came from a patient-driven perspective? We have a fleeting opportunity to advance a care model centered on convenient and equitable access that first prioritizes good outcomes, then selects approaches to care – and locations – tailored to each patient. Using the example of education, imagine how effective it would be if each student, regardless of their school district and aptitude, received such individualized attention.
That’s the idea behind virtual-first care (V1C), a new care model centered on convenient, customized, high-quality care that integrates a full suite of services tailored directly to patients’ clinical needs and preferences. This package includes asynchronous communication such as texting; video and other live virtual modes; and in-person options.
V1C goes beyond what you might think of as standard “telehealth” by using evidence-based protocols and tools that include traditional and digital therapeutics and testing, personalized care plans, dynamic patient monitoring, and team-based approaches to care. This could include spit kits mailed for laboratory tests and complementing clinical care with health coaching. V1C also replaces some in-person exams with ongoing monitoring, using sensors for more ‘whole person’ care.
Amidst all this momentum, we have the opportunity to rethink the goals of healthcare innovation, but that means bringing together key stakeholders to demonstrate that sustainable V1C can redefine healthcare.
Established V1C healthcare providers such as Omada, Headspace, and Heartbeat Health, as well as emerging market entrants like Oshi, Visana, and Wellinks, work with a variety of patients who have complicated long-term conditions such as diabetes, heart failure, gastrointestinal illness, endometriosis, and COPD. V1C is comprehensive in ways that are lacking in digital health and its other predecessors: it has the potential to integrate multiple data streams, incorporate more frequent touches and check-ins over time, and manage a much wider range of chronic health conditions, improving lives and reducing disease burden now and in the future.
Recognizing the pandemic-driven interest in virtual care, significant energy and resources are already flowing fast toward V1C. Some of the world’s largest innovators jumped into V1C early on: Verily, Alphabet’s Life Sciences Company, launched Onduo in 2016 to disrupt the diabetes healthcare market, and is now well positioned to scale its solutions. Major insurers like Aetna and United now offer virtual-first plans to members, responding as organizations expand virtual options for employees. Amidst all this momentum, we have the opportunity to rethink the goals of healthcare innovation, but that means bringing together key stakeholders to demonstrate that sustainable V1C can redefine healthcare.
That was the immediate impetus for IMPACT, a consortium of V1C companies, investors, payers and patients founded last year to ensure access to high-quality, evidence-based V1C. Developed by our team at the Digital Medicine Society (DiMe) in collaboration with the American Telemedicine Association (ATA), IMPACT has begun to explore key issues that include giving patients more integrated experiences when accessing both virtual and brick-and-mortar care.
Digital Medicine Society
V1C is not, nor should it be, virtual-only care. In this new era of hybrid healthcare, success will be defined by how well providers help patients navigate the transitions. How do we smoothly hand a patient off from an onsite primary care physician to, say, a virtual cardiologist? How do we get information from a brick-and-mortar to a digital portal? How do you manage dataflow while still staying HIPAA compliant? There are many complex regulatory implications for these new models, as well as an evolving landscape in terms of privacy, security and interoperability. It will be no small task for groups like IMPACT to determine the best path forward.
None of these factors matter unless the industry can recruit and retain clinicians. Our field is facing an unprecedented workforce crisis. Traditional healthcare is making clinicians miserable, and COVID has only accelerated the trend of overworked, disenchanted healthcare workers leaving in droves. Clinicians want more interactions with patients, and fewer with computer screens – call it “More face time, less FaceTime.” No new model will succeed unless the industry can more efficiently deploy its talent – arguably its most scarce and precious resource. V1C can help with alleviating the increasing burden and frustration borne by individual physicians in today’s status quo.
In healthcare, new technological approaches inevitably provoke no shortage of skepticism. Past lessons from Silicon Valley-driven fixes have led to understandable cynicism. But V1C is a different breed of animal. By building healthcare around the patient, not the clinic, V1C can make healthcare work better for patients, payers and providers. We’re at a fork in the road: we can revert back to a broken sick-care system, or dig in and do the hard work of figuring out how this future-forward healthcare system gets financed, organized and executed. As a field, we must find the courage and summon the energy to embrace this moment, and make it a moment of change.
Chicken that is grown entirely in a laboratory, without harming a single bird, could be sold in supermarkets in the coming months. But critics say the doubts about lab-grown meat have not been appropriately explored.
Last November, when the U.S. Food and Drug Administration disclosed that chicken from a California firm called UPSIDE Foods did not raise safety concerns, it drily upended how humans have obtained animal protein for thousands of generations.
“The FDA is ready to work with additional firms developing cultured animal cell food and production processes to ensure their food is safe and lawful,” the agency said in a statement at the time.
Assuming UPSIDE obtains clearances from the U.S. Department of Agriculture, its chicken – grown entirely in a laboratory without harming a single bird – could be sold in supermarkets in the coming months.
“Ultimately, we want our products to be available everywhere meat is sold, including retail and food service channels,” a company spokesperson said. The upscale French restaurant Atelier Crenn in San Francisco will have UPSIDE chicken on its menu once it is approved, she added.
Known as lab-grown or cultured meat, a product such as UPSIDE’s is created using stem cells and other tissue obtained from a chicken, cow or other livestock. Those cells are then multiplied in a nutrient-dense environment, usually in conjunction with a “scaffold” of plant-based materials or gelatin to give them a familiar form, such as a chicken breast or a ribeye steak. A Dutch company called Mosa Meat claims it can produce 80,000 hamburgers derived from a cluster of tissue the size of a sesame seed.
Critics say the doubts about lab-grown meat and the possibility it could merge “Brave New World” with “The Jungle” and “Soylent Green” have not been appropriately explored.
That’s a far cry from when it took months of work to create the first lab-grown hamburger a decade ago. That minuscule patty – which did not contain any fat and was literally plucked from a Petri dish to go into a frying pan – cost about $325,000 to produce.
Just a decade later, an Israeli company called Future Meat said it can produce lab-grown meat for about $1.70 per pound. It plans to open a production facility in the U.S. sometime in 2023 and distribute its products under the brand name “Believer.”
Costs for production have sunk so low that researchers at Carnegie Mellon University in Pittsburgh expect sometime in early 2024 to produce lab-grown Wagyu steak to showcase the viability of growing high-end cuts of beef cheaply. The Carnegie Mellon team is producing its Wagyu using a consumer 3-D printer bought secondhand on eBay and modified to print the highly marbled flesh using a method developed by the university. The device costs $200 – about the same as a pound of Wagyu in the U.S. The initiative’s modest five-figure budget was successfully crowdfunded last year.
“The big cost is going to be the cells (which are being extracted by a cow somewhere in Pennsylvania), but otherwise printing doesn’t add much to the process,” said Rosalyn Abbott, a Carnegie Mellon assistant professor of bioengineering who is co-leader on the project. “But it adds value, unlike doing this with ground meat.”
Lab-Grown Meat’s Promise
Proponents of lab-grown meat say it will cut down on traditional agriculture, which has been a leading contributor to deforestation, water shortages and contaminated waterways from animal waste, as well as climate change.
An Oxford University study from 2011 concludes lab-grown meat could have greenhouse emissions 96 percent lower compared to traditionally raised livestock. Moreover, proponents of lab-grown meat claim that the suffering of animals would decline dramatically, as they would no longer need to be warehoused and slaughtered. A recently opened 26-story high-rise in China dedicated to the raising and slaughtering of pigs illustrates the current plight of livestock in stark terms.
Scientists may even learn how to tweak lab-grown meat to make it more nutritious. Natural red meat is high in saturated fat and, if it’s eaten too often, can lead to chronic diseases. In lab versions, the saturated fat could be swapped for healthier, omega-3 fatty acids.
But critics say the doubts about lab-grown meat and the possibility it could merge “Brave New World” with “The Jungle” and “Soylent Green” have not been appropriately explored.
A Slippery Slope?
Some academics who have studied the moral and ethical issues surrounding lab-grown meat believe it will have a tough path ahead gaining acceptance by consumers. Should it actually succeed in gaining acceptance, many ethical questions must be answered.
“People might be interested” in lab-grown meat, perhaps as a curiosity, said Carlos Alvaro, an associate professor of philosophy at the New York City College of Technology, part of the City University of New York. But the allure of traditionally sourced meat has been baked – or perhaps grilled – into people’s minds for so long that they may not want to make the switch. Plant-based meat provides a recent example of the uphill battle involved in changing old food habits, with Beyond Meat’s stock prices dipping nearly 80 percent in 2022.
"There are many studies showing that people don’t really care about the environment (to that extent)," Alvaro said. "So I don’t know how you would convince people to do this because of the environment.”
“From my research, I understand that the taste (of lab-grown meat) is not quite there,” Alvaro said, noting that the amino acids, sugars and other nutrients required to grow cultivated meat do not mimic what livestock are fed. He also observed that the multiplication of cells as part of the process “really mimic cancer cells” in the way they grow, another off-putting thought for would-be consumers of the product.
Alvaro is also convinced the public will not buy into any argument that lab-grown meat is more environmentally friendly.
“If people care about the environment, they either try and consume considerably less meat and other animal products, or they go vegan or vegetarian,” he said. “But there are many studies showing that people don’t really care about the environment (to that extent). So I don’t know how you would convince people to do this because of the environment.”
Ben Bramble, a professor at Australian National University who previously held posts at Princeton and Trinity College in Ireland, takes a slightly different tack. He noted that “if lab-grown meat becomes cheaper, healthier, or tastier than regular meat, there will be a large market for it. If it becomes all of these things, it will dominate the market.”
However, Bramble has misgivings about that occurring. He believes a smooth transition from traditionally sourced meat to a lab-grown version would allow humans to elide over the decades of animal cruelty perpetrated by large-scale agriculture, without fully reckoning with and learning from this injustice.
“My fear is that if we all switch over to lab-grown meat because it has become cheaper, healthier, or tastier than regular meat, we might never come to realize what we have done, and the terrible things we are capable of,” he said. “This would be a catastrophe.”
Bramble’s writings about cultured meat also raise some serious moral conundrums. If, for example, animal meat may be cultivated without killing animals, why not create products from human protein?
Actually, that’s already happened.
It occurred in 2019, when Orkan Telhan, a professor of fine arts at the University of Pennsylvania, collaborated with two scientists to create an art exhibit at the Philadelphia Museum of Art on the future of foodstuffs.
Although the exhibit included bioengineered bread and genetically modified salmon, it was an installation called “Ouroboros Steak” that drew the most attention. That was comprised of pieces of human flesh grown in a lab from cultivated cells and expired blood products obtained from online sources.
The exhibit was presented as four tiny morsels of red meat – shaped in patterns suggesting an ouroboros, a dragon eating its own tail. They were placed in tiny individual saucers atop a larger plate and placemat with a calico pattern, suggesting an item to order in a diner. The artwork drew international headlines – as well as condemnation for Telhan’s vision.
Telhan’s artwork is intended to critique the overarching assumption that lab-grown meat will eventually replace more traditional production methods, as well as the lack of transparency surrounding many processed foodstuffs. “They think that this problem (from industrial-scale agriculture) is going be solved by this new technology,” Telhan said. “I am critical (of) that perspective.”
Unlike Bramble, Telhan is not against lab-grown meat, so long as its producers are transparent about the sourcing of materials and its cultivation. But he believes that large-scale agricultural meat production – which dates back centuries – is not going to be replaced so quickly.
“We see this again and again with different industries, like algae-based fuels. A lot of companies were excited about this, and promoted it,” Telhan said. “And years later, we know these fuels work. But to be able to displace the oil industry means building the infrastructure to scale takes billions of dollars, and nobody has the patience or money to do it.”
Alvaro concurred on this point, which he believes is already weakened because a large swath of consumers aren’t concerned about environmental degradation.
“They’re going to have to sell this big, but in order to convince people to do so, they have to convince them to eat this product instead of regular meat,” Alvaro said.
Hidden Tweaks?
Moreover, if lab-based meat does obtain a significant market share, Telhan suggested companies may do things to the product – such as to genetically modify it to become more profitable – and never notify consumers. That is a particular concern in the U.S., where regulations regarding such modifications are vastly more relaxed than in the European Union.
“I think that they have really good objectives, and they aspire to good objectives,” Telhan said. “But the system itself doesn't really allow for that much transparency.”
No matter what the future holds, sometime next year Carnegie Mellon is expected to hold a press conference announcing it has produced a cut of the world’s most expensive beef with the help of a modified piece of consumer electronics. It will likely take place at around the same time UPSIDE chicken will be available for purchase in supermarkets and restaurants, pending the USDA’s approvals.
Abbott, the Carnegie Mellon professor, suggested the future event will be both informative and celebratory.
“I think Carnegie Mellon would have someone potentially cook it for us,” she said. “Like have a really good chef in New York City do it.”
In this week's Friday Five, breathing this way may cut down on anxiety, a fasting regimen that could make you sick, this type of job makes men more virile, 3D printed hearts could save your life, and the role of metformin in preventing dementia.
The Friday Five covers five stories in research that you may have missed this week. There are plenty of controversies and troubling ethical issues in science – and we get into many of them in our online magazine – but this news roundup focuses on scientific creativity and progress to give you a therapeutic dose of inspiration headed into the weekend.
Here are the promising studies covered in this week's Friday Five, featuring interviews with Dr. David Spiegel, associate chair of psychiatry and behavioral sciences at Stanford, and Dr. Filip Swirski, professor of medicine and cardiology at the Icahn School of Medicine at Mount Sinai.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
Here are the promising studies covered in this week's Friday Five, featuring interviews with Dr. David Spiegel, associate chair of psychiatry and behavioral sciences at Stanford, and Dr. Filip Swirski, professor of medicine and cardiology at the Icahn School of Medicine at Mount Sinai.
- Breathing this way cuts down on anxiety*
- Could your fasting regimen make you sick?
- This type of job makes men more virile
- 3D printed hearts could save your life
- Yet another potential benefit of metformin
* This video with Dr. Andrew Huberman of Stanford shows exactly how to do the breathing practice.