What’s the Right Way to Regulate Gene-Edited Crops?

A cornfield in summer.
In the next few decades, humanity faces its biggest food crisis since the invention of the plow. The planet's population, currently 7.6 billion, is expected to reach 10 billion by 2050; to avoid mass famine, according to the World Resource Institute, we'll need to produce 70 percent more calories than we do today.
Imagine that a cheap, easy-to-use, and rapidly deployable technology could make crops more fertile and strengthen their resistance to threats.
Meanwhile, climate change will bring intensifying assaults by heat, drought, storms, pests, and weeds, depressing farm yields around the globe. Epidemics of plant disease—already laying waste to wheat, citrus, bananas, coffee, and cacao in many regions—will spread ever further through the vectors of modern trade and transportation.
So here's a thought experiment: Imagine that a cheap, easy-to-use, and rapidly deployable technology could make crops more fertile and strengthen their resistance to these looming threats. Imagine that it could also render them more nutritious and tastier, with longer shelf lives and less vulnerability to damage in shipping—adding enhancements to human health and enjoyment, as well as reduced food waste, to the possible benefits.
Finally, imagine that crops bred with the aid of this tool might carry dangers. Some could contain unsuspected allergens or toxins. Others might disrupt ecosystems, affecting the behavior or very survival of other species, or infecting wild relatives with their altered DNA.
Now ask yourself: If such a technology existed, should policymakers encourage its adoption, or ban it due to the risks? And if you chose the former alternative, how should crops developed by this method be regulated?
In fact, this technology does exist, though its use remains mostly experimental. It's called gene editing, and in the past five years it has emerged as a potentially revolutionary force in many areas—among them, treating cancer and genetic disorders; growing transplantable human organs in pigs; controlling malaria-spreading mosquitoes; and, yes, transforming agriculture. Several versions are currently available, the newest and nimblest of which goes by the acronym CRISPR.
Gene editing is far simpler and more efficient than older methods used to produce genetically modified organisms (GMOs). Unlike those methods, moreover, it can be used in ways that leave no foreign genes in the target organism—an advantage that proponents argue should comfort anyone leery of consuming so-called "Frankenfoods." But debate persists over what precautions must be taken before these crops come to market.
Recently, two of the world's most powerful regulatory bodies offered very different answers to that question. The United States Department of Agriculture (USDA) declared in March 2018 that it "does not currently regulate, or have any plans to regulate" plants that are developed through most existing methods of gene editing. The Court of Justice of the European Union (ECJ), by contrast, ruled in July that such crops should be governed by the same stringent regulations as conventional GMOs.
Some experts suggest that the broadly permissive American approach and the broadly restrictive EU policy are equally flawed.
Each announcement drew protests, for opposite reasons. Anti-GMO activists assailed the USDA's statement, arguing that all gene-edited crops should be tested and approved before marketing. "You don't know what those mutations or rearrangements might do in a plant," warned Michael Hansen, a senior scientist with the advocacy group Consumers Union. Biotech boosters griped that the ECJ's decision would stifle innovation and investment. "By any sensible standard, this judgment is illogical and absurd," wrote the British newspaper The Observer.
Yet some experts suggest that the broadly permissive American approach and the broadly restrictive EU policy are equally flawed. "What's behind these regulatory decisions is not science," says Jennifer Kuzma, co-director of the Genetic Engineering and Society Center at North Carolina State University, a former advisor to the World Economic Forum, who has researched and written extensively on governance issues in biotechnology. "It's politics, economics, and culture."
The U.S. Welcomes Gene-Edited Food
Humans have been modifying the genomes of plants and animals for 10,000 years, using selective breeding—a hit-or-miss method that can take decades or more to deliver rewards. In the mid-20th century, we learned to speed up the process by exposing organisms to radiation or mutagenic chemicals. But it wasn't until the 1980s that scientists began modifying plants by altering specific stretches of their DNA.
Today, about 90 percent of the corn, cotton and soybeans planted in the U.S. are GMOs; such crops cover nearly 4 million square miles (10 million square kilometers) of land in 29 countries. Most of these plants are transgenic, meaning they contain genes from an unrelated species—often as biologically alien as a virus or a fish. Their modifications are designed primarily to boost profit margins for mechanized agribusiness: allowing crops to withstand herbicides so that weeds can be controlled by mass spraying, for example, or to produce their own pesticides to lessen the need for chemical inputs.
In the early days, the majority of GM crops were created by extracting the gene for a desired trait from a donor organism, multiplying it, and attaching it to other snippets of DNA—usually from a microbe called an agrobacterium—that could help it infiltrate the cells of the target plant. Biotechnologists injected these particles into the target, hoping at least one would land in a place where it would perform its intended function; if not, they kept trying. The process was quicker than conventional breeding, but still complex, scattershot, and costly.
Because agrobacteria can cause plant tumors, Kuzma explains, policymakers in the U.S. decided to regulate GMO crops under an existing law, the Plant Pest Act of 1957, which addressed dangers like imported trees infested with invasive bugs. Every GMO containing the DNA of agrobacterium or another plant pest had to be tested to see whether it behaved like a pest, and undergo a lengthy approval process. By 2010, however, new methods had been developed for creating GMOs without agrobacteria; such plants could typically be marketed without pre-approval.
Soon after that, the first gene-edited crops began appearing. If old-school genetic engineering was a shotgun, techniques like TALEN and CRISPR were a scalpel—or the search-and-replace function on a computer program. With CRISPR/Cas9, for example, an enzyme that bacteria use to recognize and chop up hostile viruses is reprogrammed to find and snip out a desired bit of a plant or other organism's DNA. The enzyme can also be used to insert a substitute gene. If a DNA sequence is simply removed, or the new gene comes from a similar species, the changes in the target plant's genotype and phenotype (its general characteristics) may be no different from those that could be produced through selective breeding. If a foreign gene is added, the plant becomes a transgenic GMO.
Companies are already teeing up gene-edited products for the U.S. market, like a cooking oil and waxy corn.
This development, along with the emergence of non-agrobacterium GMOs, eventually prompted the USDA to propose a tiered regulatory system for all genetically engineered crops, beginning with an initial screening for potentially hazardous metaboloids or ecological impacts. (The screening was intended, in part, to guard against the "off-target effects"—stray mutations—that occasionally appear in gene-edited organisms.) If no red flags appeared, the crop would be approved; otherwise, it would be subject to further review, and possible regulation.
The plan was unveiled in January 2017, during the last week of the Obama presidency. Then, under the Trump administration, it was shelved. Although the USDA continues to promise a new set of regulations, the only hint of what they might contain has been Secretary of Agriculture Sonny Perdue's statement last March that gene-edited plants would remain unregulated if they "could otherwise have been developed through traditional breeding techniques, as long as they are not plant pests or developed using plant pests."
Because transgenic plants could not be "developed through traditional breeding techniques," this statement could be taken to mean that gene editing in which foreign DNA is introduced might actually be regulated. But because the USDA regulates conventional transgenic GMOs only if they trigger the plant-pest stipulation, experts assume gene-edited crops will face similarly limited oversight.
Meanwhile, companies are already teeing up gene-edited products for the U.S. market. An herbicide-resistant oilseed rape, developed using a proprietary technique, has been available since 2016. A cooking oil made from TALEN-tweaked soybeans, designed to have a healthier fatty-acid profile, is slated for release within the next few months. A CRISPR-edited "waxy" corn, designed with a starch profile ideal for processed foods, should be ready by 2021.
In all likelihood, none of these products will have to be tested for safety.
In the E.U., Stricter Rules Apply
Now let's look at the European Union. Since the late 1990s, explains Gregory Jaffe, director of the Project on Biotechnology at the Center for Science in the Public Interest, the EU has had a "process-based trigger" for genetically engineered products: "If you use recombinant DNA, you are going to be regulated." All foods and animal feeds must be approved and labeled if they consist of or contain more than 0.9 percent GM ingredients. (In the U.S., "disclosure" of GM ingredients is mandatory, if someone asks, but labeling is not required.) The only GM crop that can be commercially grown in EU member nations is a type of insect-resistant corn, though some countries allow imports.
European scientists helped develop gene editing, and they—along with the continent's biotech entrepreneurs—have been busy developing applications for crops. But European farmers seem more divided over the technology than their American counterparts. The main French agricultural trades union, for example, supports research into non-transgenic gene editing and its exemption from GMO regulation. But it was the country's small-farmers' union, the Confédération Paysanne, along with several allied groups, that in 2015 submitted a complaint to the ECJ, asking that all plants produced via mutagenesis—including gene-editing—be regulated as GMOs.
At this point, it should be mentioned that in the past 30 years, large population studies have found no sign that consuming GM foods is harmful to human health. GMO critics can, however, point to evidence that herbicide-resistant crops have encouraged overuse of herbicides, giving rise to poison-proof "superweeds," polluting the environment with suspected carcinogens, and inadvertently killing beneficial plants. Those allegations were key to the French plaintiffs' argument that gene-edited crops might similarly do unexpected harm. (Disclosure: Leapsmag's parent company, Bayer, recently acquired Monsanto, a maker of herbicides and herbicide-resistant seeds. Also, Leaps by Bayer, an innovation initiative of Bayer and Leapsmag's direct founder, has funded a biotech startup called JoynBio that aims to reduce the amount of nitrogen fertilizer required to grow crops.)
The ruling was "scientifically nonsensical. It's because of things like this that I'll never go back to Europe."
In the end, the EU court found in the Confédération's favor on gene editing—though the court maintained the regulatory exemption for mutagenesis induced by chemicals or radiation, citing the 'long safety record' of those methods.
The ruling was "scientifically nonsensical," fumes Rodolphe Barrangou, a French food scientist who pioneered CRISPR while working for DuPont in Wisconsin and is now a professor at NC State. "It's because of things like this that I'll never go back to Europe."
Nonetheless, the decision was consistent with longstanding EU policy on crops made with recombinant DNA. Given the difficulty and expense of getting such products through the continent's regulatory system, many other European researchers may wind up following Barrangou to America.
Getting to the Root of the Cultural Divide
What explains the divergence between the American and European approaches to GMOs—and, by extension, gene-edited crops? In part, Jennifer Kuzma speculates, it's that Europeans have a different attitude toward eating. "They're generally more tied to where their food comes from, where it's produced," she notes. They may also share a mistrust of government assurances on food safety, borne of the region's Mad Cow scandals of the 1980s and '90s. In Catholic countries, consumers may have misgivings about tinkering with the machinery of life.
But the principal factor, Kuzma argues, is that European and American agriculture are structured differently. "GM's benefits have mostly been designed for large-scale industrial farming and commodity crops," she says. That kind of farming is dominant in the U.S., but not in Europe, leading to a different balance of political power. In the EU, there was less pressure on decisionmakers to approve GMOs or exempt gene-edited crops from regulation—and more pressure to adopt a GM-resistant stance.
Such dynamics may be operating in other regions as well. In China, for example, the government has long encouraged research in GMOs; a state-owned company recently acquired Syngenta, a Swiss-based multinational corporation that is a leading developer of GM and gene-edited crops. GM animal feed and cooking oil can be freely imported. Yet commercial cultivation of most GM plants remains forbidden, out of deference to popular suspicions of genetically altered food. "As a new item, society has debates and doubts on GMO techniques, which is normal," President Xi Jinping remarked in 2014. "We must be bold in studying it, [but] be cautious promoting it."
The proper balance between boldness and caution is still being worked out all over the world. Europe's process-based approach may prevent researchers from developing crops that, with a single DNA snip, could rescue millions from starvation. EU regulations will also make it harder for small entrepreneurs to challenge Big Ag with a technology that, as Barrangou puts it, "can be used affordably, quickly, scalably, by anyone, without even a graduate degree in genetics." America's product-based approach, conversely, may let crops with hidden genetic dangers escape detection. And by refusing to investigate such risks, regulators may wind up exacerbating consumers' doubts about GM and gene-edited products, rather than allaying them.
"Science...can't tell you what to regulate. That's a values-based decision."
Perhaps the solution lies in combining both approaches, and adding some flexibility and nuance to the mix. "I don't believe in regulation by the product or the process," says CSPI's Jaffe. "I think you need both." Deleting a DNA base pair to silence a gene, for example, might be less risky than inserting a foreign gene into a plant—unless the deletion enables the production of an allergen, and the transgene comes from spinach.
Kuzma calls for the creation of "cooperative governance networks" to oversee crop genome editing, similar to bodies that already help develop and enforce industry standards in fisheries, electronics, industrial cleaning products, and (not incidentally) organic agriculture. Such a network could include farmers, scientists, advocacy groups, private companies, and governmental agencies. "Safety isn't an all-or-nothing concept," Kuzma says. "Science can tell you what some of the issues are in terms of risk and benefit, but it can't tell you what to regulate. That's a values-based decision."
By drawing together a wide range of stakeholders to make such decisions, she adds, "we're more likely to anticipate future consequences, and to develop a robust approach—one that not only seems more legitimate to people, but is actually just plain old better."
Eight Big Medical and Science Trends to Watch in 2021
Promising developments underway include advancements in gene and cell therapy, better testing for COVID, and a renewed focus on climate change.
The world as we know it has forever changed. With a greater focus on science and technology than before, experts in the biotech and life sciences spaces are grappling with what comes next as SARS-CoV-2, the coronavirus that causes the COVID-19 illness, has spread and mutated across the world.
Even with vaccines being distributed, so much still remains unknown.
Jared Auclair, Technical Supervisor for the Northeastern University's Life Science Testing Center in Burlington, Massachusetts, guides a COVID testing lab that cranks out thousands of coronavirus test results per day. His lab is also focused on monitoring the quality of new cell and gene therapy products coming to the market.
Here are trends Auclair and other experts are watching in 2021.
Better Diagnostic Testing for COVID
Expect improvements in COVID diagnostic testing and the ability to test at home.
There are currently three types of coronavirus tests. The molecular test—also known as the RT-PCR test, detects the virus's genetic material, and is highly accurate, but it can take days to receive results. There are also antibody tests, done through a blood draw, designed to test whether you've had COVID in the past. Finally, there's the quick antigen test that isn't as accurate as the PCR test, but can identify if people are going to infect others.
Last month, Lucira Health secured the U.S. FDA Emergency Use Authorization for the first prescription molecular diagnostic test for COVID-19 that can be performed at home. On December 15th, the Ellume Covid-19 Home Test received authorization as the first over-the-counter COVID-19 diagnostic antigen test that can be done at home without a prescription. The test uses a nasal swab that is connected to a smartphone app and returns results in 15-20 minutes. Similarly, the BinaxNOW COVID-19 Ag Card Home Test received authorization on Dec. 16 for its 15-minute antigen test that can be used within the first seven days of onset of COIVD-19 symptoms.
Home testing has the possibility to impact the pandemic pretty drastically, Auclair says, but there are other considerations: the type and timing of test that is administered, how expensive is the test (and if it is financially feasible for the general public) and the ability of a home test taker to accurately administer the test.
"The vaccine roll-out will not eliminate the need for testing until late 2021 or early 2022."
Ideally, everyone would frequently get tested, but that would mean the cost of a single home test—which is expected to be around $30 or more—would need to be much cheaper, more in the $5 range.
Auclair expects "innovations in the diagnostic space to explode" with the need for more accurate, inexpensive, quicker COVID tests. Auclair foresees innovations to be at first focused on COVID point-of-care testing, but he expects improvements within diagnostic testing for other types of viruses and diseases too.
"We still need more testing to get the pandemic under control, likely over the next 12 months," Auclair says. "The vaccine roll-out will not eliminate the need for testing until late 2021 or early 2022."
Rise of mRNA-based Vaccines and Therapies
A year ago, vaccines weren't being talked about like they are today.
"But clearly vaccines are the talk of the town," Auclair says. "The reason we got a vaccine so fast was there was so much money thrown at it."
A vaccine can take more than 10 years to fully develop, according to the World Economic Forum. Prior to the new COVID vaccines, which were remarkably developed and tested in under a year, the fastest vaccine ever made was for mumps -- and it took four years.
"Normally you have to produce a protein. This is typically done in eggs. It takes forever," says Catherine Dulac, a neuroscientist and developmental biologist at Harvard University who won the 2021 Breakthrough Prize in Life Sciences. "But an mRNA vaccine just enabled [us] to skip all sorts of steps [compared with burdensome conventional manufacturing] and go directly to a product that can be injected into people."
Non-traditional medicines based on genetic research are in their infancy. With mRNA-based vaccines hitting the market for the first time, look for more vaccines to be developed for whatever viruses we don't currently have vaccines for, like dengue virus and Ebola, Auclair says.
"There's a whole bunch of things that could be explored now that haven't been thought about in the past," Auclair says. "It could really be a game changer."
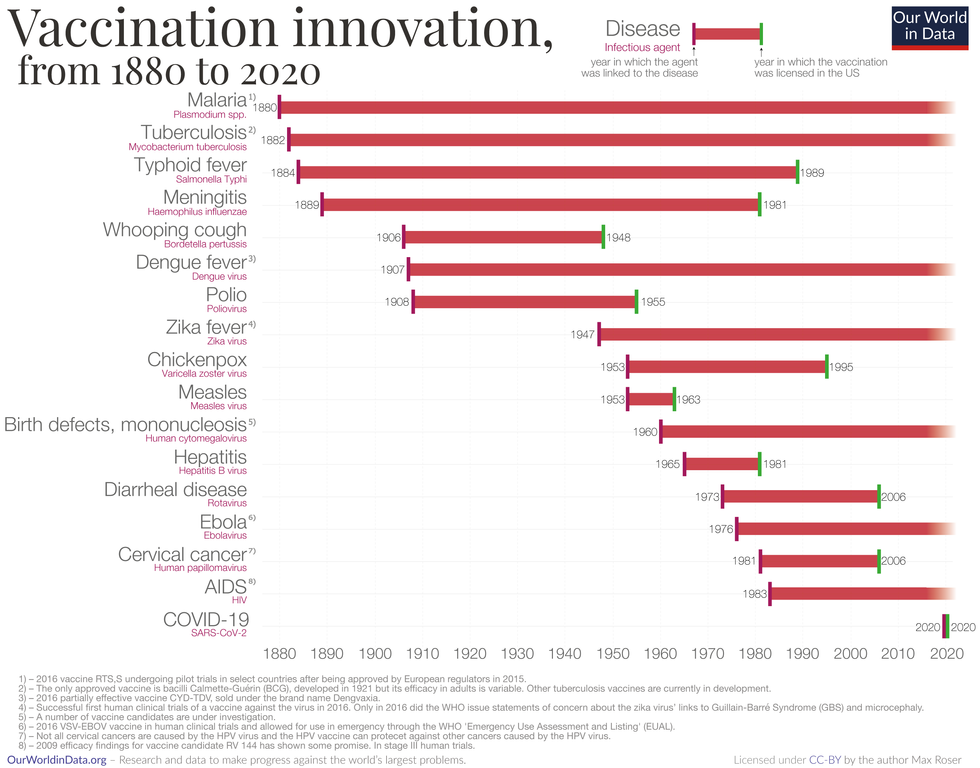
Vaccine Innovation over the last 140 years.
Max Roser/Our World in Data (Creative Commons license)
Advancements in Cell and Gene Therapies
CRISPR, a type of gene editing, is going to be huge in 2021, especially after the Nobel Prize in Chemistry was awarded to Emmanuelle Charpentier and Jennifer Doudna in October for pioneering the technology.
Right now, CRISPR isn't completely precise and can cause deletions or rearrangements of DNA.
"It's definitely not there yet, but over the next year it's going to get a lot closer and you're going to have a lot of momentum in this space," Auclair says. "CRISPR is one of the technologies I'm most excited about and 2021 is the year for it."
Gene therapies are typically used on rare genetic diseases. They work by replacing the faulty dysfunctional genes with corrected DNA codes.
"Cell and gene therapies are really where the field is going," Auclair says. "There is so much opportunity....For the first time in our life, in our existence as a species, we may actually be able to cure disease by using [techniques] like gene editing, where you cut in and out of pieces of DNA that caused a disease and put in healthy DNA," Auclair says.
For example, Spinal Muscular Atrophy is a rare genetic disorder that leads to muscle weakness, paralysis and death in children by age two. As of last year, afflicted children can take a gene therapy drug called Zolgensma that targets the missing or nonworking SMN1 gene with a new copy.
Another recent breakthrough uses gene editing for sickle cell disease. Victoria Gray, a mom from Mississippi who was exclusively followed by NPR, was the first person in the United States to be successfully treated for the genetic disorder with the help of CRISPR. She has continued to improve since her landmark treatment on July 2, 2019 and her once-debilitating pain has greatly eased.
"This is really a life-changer for me," she told NPR. "It's magnificent."
"You are going to see bigger leaps in gene therapies."
Look out also for improvements in cell therapies, but on a much lesser scale.
Cell therapies remove immune cells from a person or use cells from a donor. The cells are modified or cultured in lab, multiplied by the millions and then injected back into patients. These include stem cell therapies as well as CAR-T cell therapies, which are typically therapies of last resort and used in cancers like leukemia, Auclair says.
"You are going to see bigger leaps in gene therapies," Auclair says. "It's being heavily researched and we understand more about how to do gene therapies. Cell therapies will lie behind it a bit because they are so much more difficult to work with right now."
More Monoclonal Antibody Therapies
Look for more customized drugs to personalize medicine even more in the biotechnology space.
In 2019, the FDA anticipated receiving more than 200 Investigational New Drug (IND) applications in 2020. But with COVID, the number of INDs skyrocketed to 6,954 applications for the 2020 fiscal year, which ended September 30, 2020, according to the FDA's online tracker. Look for antibody therapies to play a bigger role.
Monoclonal antibodies are lab-grown proteins that mimic or enhance the immune system's response to fight off pathogens, like viruses, and they've been used to treat cancer. Now they are being used to treat patients with COVID-19.
President Donald Trump received a monoclonal antibody cocktail, called REGEN-COV2, which later received FDA emergency use authorization.
A newer type of monoclonal antibody therapy is Antibody-Drug Conjugates, also called ADCs. It's something we're going to be hearing a lot about in 2021, Auclair says.
"Antibody-Drug Conjugates is a monoclonal antibody with a chemical, we consider it a chemical warhead on it," Auclair says. "The monoclonal antibody binds to a specific antigen in your body or protein and delivers a chemical to that location and kills the infected cell."
Moving Beyond Male-Centric Lab Testing
Scientific testing for biology has, until recently, focused on testing males. Dulac, a Howard Hughes Medical Investigator and professor of molecular and cellular biology at Harvard University, challenged that idea to find brain circuitry behind sex-specific behaviors.
"For the longest time, until now, all the model systems in biology, are male," Dulac says. "The idea is if you do testing on males, you don't need to do testing on females."
Clinical models are done in male animals, as well as fundamental research. Because biological research is always done on male models, Dulac says the outcomes and understanding in biology is geared towards understanding male biology.
"All the drugs currently on the market and diagnoses of diseases are biased towards the understanding of male biology," Dulac says. "The diagnostics of diseases is way weaker in women than men."
That means the treatment isn't necessarily as good for women as men, she says, including what is known and understood about pain medication.
"So pain medication doesn't work well in women," Dulac says. "It works way better in men. It's true for almost all diseases that I know. Why? because you have a science that is dominated by males."
Although some in the scientific community challenge that females are not interesting or too complicated with their hormonal variations, Dulac says that's simply not true.
"There's absolutely no reason to decide 50% of life forms are interesting and the other 50% are not interesting. What about looking at both?" says Dulac, who was awarded the $3 million Breakthrough Prize in Life Sciences in September for connecting specific neural mechanisms to male and female parenting behaviors.
Disease Research on Single Cells
To better understand how diseases manifest in the body's cell and tissues, many researchers are looking at single-cell biology. Cells are the most fundamental building blocks of life. Much still needs to be learned.
"A remarkable development this year is the massive use of analysis of gene expression and chromosomal regulation at the single-cell level," Dulac says.
Much is focused on the Human Cell Atlas (HCA), a global initiative to map all cells in healthy humans and to better identify which genes associated with diseases are active in a person's body. Most estimates put the number of cells around 30 trillion.
Dulac points to work being conducted by the Cell Census Network (BICCN) Brain Initiative, an initiative by the National Institutes of Health to come up with an atlas of cell types in mouse, human and non-human primate brains, and the Chan Zuckerberg Initiative's funding of single-cell biology projects, including those focused on single-cell analysis of inflammation.
"Our body and our brain are made of a large number of cell types," Dulac says. "The ability to explore and identify differences in gene expression and regulation in massively multiplex ways by analyzing millions of cells is extraordinarily important."
Converting Plastics into Food
Yep, you heard it right, plastics may eventually be turned into food. The Defense Advanced Research Projects Agency, better known as DARPA, is funding a project—formally titled "Production of Macronutrients from Thermally Oxo-Degraded Wastes"—and asking researchers how to do this.
"When I first heard about this challenge, I thought it was absolutely absurd," says Dr. Robert Brown, director of the Bioeconomy Institute at Iowa State University and the project's principal investigator, who is working with other research partners at the University of Delaware, Sandia National Laboratories, and the American Institute of Chemical Engineering (AIChE)/RAPID Institute.
But then Brown realized plastics will slowly start oxidizing—taking in oxygen—and microorganisms can then consume it. The oxidation process at room temperature is extremely slow, however, which makes plastics essentially not biodegradable, Brown says.
That changes when heat is applied at brick pizza oven-like temperatures around 900-degrees Fahrenheit. The high temperatures get compounds to oxidize rapidly. Plastics are synthetic polymers made from petroleum—large molecules formed by linking many molecules together in a chain. Heated, these polymers will melt and crack into smaller molecules, causing them to vaporize in a process called devolatilization. Air is then used to cause oxidation in plastics and produce oxygenated compounds—fatty acids and alcohols—that microorganisms will eat and grow into single-cell proteins that can be used as an ingredient or substitute in protein-rich foods.
"The caveat is the microorganisms must be food-safe, something that we can consume," Brown says. "Like supplemental or nutritional yeast, like we use to brew beer and to make bread or is used in Australia to make Vegemite."
What do the microorganisms look like? For any home beer brewers, it's the "gunky looking stuff you'd find at the bottom after the fermentation process," Brown says. "That's cellular biomass. Like corn grown in the field, yeast or other microorganisms like bacteria can be harvested as macro-nutrients."
Brown says DARPA's ReSource program has challenged all the project researchers to find ways for microorganisms to consume any plastics found in the waste stream coming out of a military expeditionary force, including all the packaging of food and supplies. Then the researchers aim to remake the plastic waste into products soldiers can use, including food. The project is in the first of three phases.
"We are talking about polyethylene, polypropylene, like PET plastics used in water bottles and converting that into macronutrients that are food," says Brown.
Renewed Focus on Climate Change
The Union of Concerned Scientists say carbon dioxide levels are higher today than any point in at least 800,000 years.
"Climate science is so important for all of humankind. It is critical because the quality of life of humans on the planet depends on it."
Look for technology to help locate large-scale emitters of carbon dioxide, including sensors on satellites and artificial intelligence to optimize energy usage, especially in data centers.
Other technologies focus on alleviating the root cause of climate change: emissions of heat-trapping gasses that mainly come from burning fossil fuels.
Direct air carbon capture, an emerging effort to capture carbon dioxide directly from ambient air, could play a role.
The technology is in the early stages of development and still highly uncertain, says Peter Frumhoff, director of science and policy at Union of Concerned Scientists. "There are a lot of questions about how to do that at sufficiently low costs...and how to scale it up so you can get carbon dioxide stored in the right way," he says, and it can be very energy intensive.
One of the oldest solutions is planting new forests, or restoring old ones, which can help convert carbon dioxide into oxygen through photosynthesis. Hence the Trillion Trees Initiative launched by the World Economic Forum. Trees are only part of the solution, because planting trees isn't enough on its own, Frumhoff says. That's especially true, since 2020 was the year that human-made, artificial stuff now outweighs all life on earth.
More research is also going into artificial photosynthesis for solar fuels. The U.S. Department of Energy awarded $100 million in 2020 to two entities that are conducting research. Look also for improvements in battery storage capacity to help electric vehicles, as well as back-up power sources for solar and wind power, Frumhoff says.
Another method to combat climate change is solar geoengineering, also called solar radiation management, which reflects sunlight back to space. The idea stems from a volcanic eruption in 1991 that released a tremendous amount of sulfate aerosol particles into the stratosphere, reflecting the sunlight away from Earth. The planet cooled by a half degree for nearly a year, Frumhoff says. However, he acknowledges, "there's a lot of things we don't know about the potential impacts and risks" involved in this controversial approach.
Whatever the approach, scientific solutions to climate change are attracting renewed attention. Under President Trump, the White House Office of Science and Technology Policy didn't have an acting director for almost two years. Expect that to change when President-elect Joe Biden takes office.
"Climate science is so important for all of humankind," Dulac says. "It is critical because the quality of life of humans on the planet depends on it."
Vaccines Are the Safest Medical Procedure We Have. Make Your Wager Wisely.
Frontline infectious disease physician Amesh Adalja received his COVID-19 vaccine on December 18th, 2020 in Butler, PA.
In the late 1650's the French polymath and renowned scientist Blaise Pascal, having undergone a religious experience that transformed him into something of a zealot, suggested the following logical strategy regarding belief in God: If there is a God, then believing in him will ensure you an eternity of bliss, while not believing in him could earn you an eternal sentence to misery.
On the other hand, if there is no God, believing in him anyway will cost you very little, and not believing in him will mean nothing in the non-existent after life. Therefore, the only sensible bet is to believe in God. This has come to be known as Pascal's wager.
It has a surprising number of applications beyond concerns for a comfortable afterlife. There are many things for which the value of believing something or not can be seen as a cost vs. likely benefit wager, often without regard to the actual truth of the matter. Since science does not profess to have a final truth, and in many areas freely admits its incomplete knowledge, Pascal's wager can provide a useful method of deciding between two alternatives.
For example, it seems that a significant percentage of the population is suspicious of science, or so we are told. We often hear that some large number, approaching or exceeding half of Americans, do not believe in evolution. This seems remarkable on the face of it because there is no viable scientific opposition to evolution and it is widely accepted by biologists and other life-scientists as being fundamental to understanding biology – from genetics to medicine.
What we are not often told is that most of those who answer negatively about believing in evolution nonetheless understand evolution – or at least the basics of it. They are not stupid, ignorant or uninformed. They have simply made a Pascalian wager. What benefit we might ask is derived from believing in evolution rather than a divine creation? Unless you are a professional biologist it is hard to see how this would affect your everyday life. On the other hand professing a belief in Darwinian evolution over the biblical narrative will likely ostracize you from family, friends, co-workers, your church community - in short most of your social infrastructure. Place your bets.
Can we apply any of this to decisions over the current controversy surrounding vaccination – and in particular the newly arrived Covid-19 vaccine?
While it is true that for entirely economic reasons, this is the first vaccine to be produced in this way, the method is not really new and the science that makes it possible has been developing over the last 40 years.
Common Concerns
There are certainly reasons to be concerned about being vaccinated and it would be a gross over-simplification to consider anyone who expresses reticence about taking a vaccine, this new vaccine in particular, as being just plain dumb or scientifically illiterate or gullible. They need be none of these things and still may be suspicious of the vaccine.
One issue is safety. The vaccine, any vaccine, is designed to mobilize your immune system, essentially to fool it into believing that there is an invading virus present and to mount an immune response. That way it will be ready when the real invasion comes, if it comes. This seems pretty sensible and preferable to going to war with an opponent you know nothing about. But still, it is fooling around with Mother Nature and some people are uneasy about that. Although it must be pointed out that the virus is not at all shy about fooling around with your immune system and many other parts of you, so letting it have its way is not good policy either.
What about a vaccine made of genes? This vaccine is being produced by what is being touted as a new method using RNA – genes. While it is true that for entirely economic reasons, this is the first vaccine to be produced in this way, the method is not really new and the science that makes it possible has been developing over the last 40 years. So it's not so radical as the press makes it seem.
But it is true that this method uses RNA, genetic material, to make the vaccine. We hear a lot about gene modification and the potential dangers associated with it. Why then am I going to allow RNA, genes, to be injected into me? The first thing to realize is that this is exactly what the virus does – so whether you get a vaccine or an infection, you are getting genes injected into you. The virus RNA encodes around 12 functional genes (by comparison humans and other mammals have around 25,000 genes). The virus only contains the genes to make a new virus – it does not have any of the capabilities of a normal cell to actually turn those genes into the proteins that make up the complete virus. It hijacks your cells to do this – and that's how it sickens you, by forcing your cells to make new viruses instead of what they should be doing.
Now the new vaccines have taken just one of those genes – the one that directs the production of the now infamous spike protein that appears on the surface of a normal virus – and injects just that one gene into your muscle cells, which then make that one single protein. Your immune system comes along and sees that weird protein and makes antibodies to it. These same antibodies will now recognize the spike protein on the surface of any viral particles that invade your body. We have effectively turned the virus into its own enemy.
The viral RNA that you are getting will decompose over a few days because RNA is not a stable molecule (that, by the way, is why the vaccine needs to be kept frozen) and it will no longer exist in your body. It could only become a permanent part of your genome if it were a DNA molecule instead of an RNA molecule – and even the chances of that happening would be chemically remote. So regardless of how it sounds, this may actually be the safest sort of vaccine to use. In the future it is likely that all vaccines will be made this way.
Then, of course, there is the issue of who is running this whole vaccine program – the government and the pharmaceutical industry. These are the guys who brought you opioid addiction, death by Vioxx, soaring drug prices, the worst health care system in the developed world, regulations where you don't need them and none where you do – am I really going to trust this cast of so-called "inept villains," as some believe, to dictate my personal health choices? Do we know for sure that the claims of efficacy are real or just made up to sell some worthless procedure? It would not be the first time. (I would not, on the other hand, worry about Bill Gates having a chip inserted into you along with the vaccine – if you use any social media, navigational tools, or purchase anything online, then Bill Gates already knows more about you than he will get from any injectable chip. So that train has left the station.)
The main upside to vaccines is that because they use your already existing defense system, they are surprisingly safe.
The Vaccine Wager
All this and a few lesser issues are worth a pause for sure. But we must also look on the positive side of the ledger. Why trust science? Modern medicine and the science behind it has eliminated or dramatically lessened such scourges as smallpox, polio, cholera, chicken pox, measles, rabies and dozens of other killer pathogens that had previously wiped out enormous numbers of people, in some cases significant parts of entire generations. Don't we depend on science for much of the comfort and safety of our everyday lives? Isn't science the way we heat our homes, drive to work, fly around the world, have dependable food? Yes, there is the bomb – but there is also anesthesia.
When it comes to viruses, the only tool we have to fight them is vaccination. The only tool. Antibiotics are for bacteria, a completely different sort of creature. Sanitation beyond personal hand washing is ineffective. Vaccines trick the immune system into recognizing the virus earlier than it would otherwise and protect normal cells from invasion by the virus. Tricking the immune system is understandably problematic for people who believe that their body knows best if it's just kept healthy. This virus, as we have seen from the array of infected people that includes apparently healthy folks, unfortunately does not subscribe to that belief.
By a similar sort of reasoning, some people make the plausible error of calculating that the vaccine is 95% effective but the survival rate is 99%, so why not just let my natural resistance take care of this? Indeed, that might not be unreasonable thinking if we were talking about the common cold, but this virus has shown itself to be a tricky character and we are not yet able to predict who gets a serious case and who a mild one. With those sorts of stakes, you shouldn't wager on either of those numbers because they have nothing to do with you as an individual. Like flipping a coin, there is only a 1% chance of it coming up heads 6 times in a row. But if it has come up heads 5 times in a row the probability of it coming up heads on the next flip is … still 50/50.
An even larger unknown is whether there may be long-term effects associated with SARS-Cov-2, as is the case for many viruses. The 1918 influenza virus has been linked to a subsequent 2-3 fold increase in Parkinson's disease by a mechanism we still don't understand. The virus that gives children chicken pox will hide out in a person's body for 40 years or more and then emerge as a painful, sometimes debilitating, case of shingles. The 99% survivability rate of this virus is meaningless if 20 years from now it causes some devastating pulmonary or brain disease.
The main upside to vaccines is that because they use your already existing defense system, they are surprisingly safe. Safer than antibiotics which have numerous side effects because they are not part of our normal make up and are cell killers – mostly bacterial cells, but they are not so perfectly targeted that they don't leave some collateral damage in their wake. All drugs and treatments have side effects, but vaccines in general have the fewest. This vaccine in particular has undergone many more than the usual safety measures - multiple independent review boards, massive press and public attention, governmental and non-governmental oversight, the most diverse trial cohorts ever assembled. Nothing here was rushed, no shortcuts were taken.
So here's the vaccine wager. Vaccines are the safest medical procedure we have. They are also among the most effective, but that's curiously not important for the bet. My claim about their safety is because vaccines are in a special class of medical tools. They are the only medical procedure or drug that is given to healthy people. Every other treatment we use medically is aimed at some existing pathology - from a cold to cancer.
Vaccines therefore have to reach a higher standard of safety than any other medical treatment. You can't take healthy people and make them sick. Vaccines have fewer side effects than virtually any other drug you wouldn't even think twice about taking – aspirin, for instance, which can cause internal bleeding, gastric ulcers, stroke. But since you are sick when you take those drugs you are willing to make the bet that the benefits will outweigh the possible side effects.
With vaccines the wager is much simpler – it is indeed more like Pascal's original wager. It may or may not be highly effective (some vaccines are only 60% effective) but they are so safe that taking them poses little risk, whereas not taking them subjects you (and others) to considerable risk, i.e., getting the virus. Like believing or not in an afterlife, the smart money is with Pascal, who I think would have reasoned himself right to the head of the vaccination line.