Which Meds are Safe When You’re Pregnant? Science Wants to Find Out

A pregnant woman is uncertain which medicine is safe to take.
Sarah Mancoll was 22 years old when she noticed a bald spot on the back of her head. A dermatologist confirmed that it was alopecia aerata, an autoimmune disorder that causes hair loss.
Of 213 new drugs approved from 2003 to 2012, only five percent included any data from pregnant women.
She successfully treated the condition with corticosteroid shots for nearly 10 years. Then Mancoll and her husband began thinking about starting a family. Would the shots be safe for her while pregnant? For the fetus? What about breastfeeding?
Mancoll consulted her primary care physician, her dermatologist, even a pediatrician. Without clinical data, no one could give her a definitive answer, so she stopped treatment to be "on the safe side." By the time her son was born, she'd lost at least half her hair. She returned to her Washington, D.C., public policy job two months later entirely bald—and without either eyebrows or eyelashes.
After having two more children in quick succession, Mancoll recently resumed the shots but didn't forget her experience. Today, she is an advocate for including more pregnant and lactating women in clinical studies so they can have more information about therapies than she did.
"I live a very privileged life, and I'll do just fine with or without hair, but it's not just about me," Mancoll said. "It's about a huge population of women who are being disenfranchised…They're invisible."
About 4 million women give birth each year in the United States, and many face medical conditions, from hypertension and diabetes to psychiatric disorders. A 2011 study showed that most women reported taking at least one medication while pregnant between 1976 and 2008. But for decades, pregnant and lactating women have been largely excluded from clinical drug studies that rigorously test medications for safety and effectiveness.
An estimated 98 percent of government-approved drug treatments between 2000 and 2010 had insufficient data to determine risk to the fetus, and close to 75 percent had no human pregnancy data at all. All told, of 213 new pharmaceuticals approved from 2003 to 2012, only five percent included any data from pregnant women.
But recent developments suggest that could be changing. Amid widespread concerns about increased maternal mortality rates, women's health advocates, physicians, and researchers are sensing and encouraging a cultural shift toward protecting women through responsible research instead of from research.
"The question is not whether to do research with pregnant women, but how," Anne Drapkin Lyerly, professor and associate director of the Center for Bioethics at the University of North Carolina at Chapel Hill, wrote last year in an op-ed. "These advances are essential. It is well past time—and it is morally imperative—for research to benefit pregnant women."
"In excluding pregnant women from drug trials to protect them from experimentation, we subject them to uncontrolled experimentation."
To that end, the American College of Obstetricians and Gynecologists' Committee on Ethics acknowledged that research trials need to be better designed so they don't "inappropriately constrain the reproductive choices of study participants or unnecessarily exclude pregnant women." A federal task force also called for significantly expanded research and the removal of regulatory barriers that make it difficult for pregnant and lactating women to participate in research.
Several months ago, a government change to a regulation known as the Common Rule took effect, removing pregnant women as a "vulnerable population" in need of special protections -- a designation that had made it more difficult to enroll them in clinical drug studies. And just last week, the U.S. Food and Drug Administration (FDA) issued new draft guidances for industry on when and how to include pregnant and lactating women in clinical trials.
Inclusion is better than the absence of data on their treatment, said Catherine Spong, former chair of the federal task force.
"It's a paradox," said Spong, professor of obstetrics and gynecology and chief of maternal fetal medicine at University of Texas Southwestern Medical Center. "There is a desire to protect women and fetuses from harm, which is translated to a reluctance to include them in research. By excluding them, the evidence for their care is limited."
Jacqueline Wolf, a professor of the history of medicine at Ohio University, agreed.
"In excluding pregnant women from drug trials to protect them from experimentation, we subject them to uncontrolled experimentation," she said. "We give them the medication without doing any research, and that's dangerous."
Women, of course, don't stop getting sick or having chronic medical conditions just because they are pregnant or breastfeeding, and conditions during pregnancy can affect a baby's health later in life. Evidence-based data is important for other reasons, too.
Pregnancy can dramatically change a woman's physiology, affecting how drugs act on her body and how her body acts or reacts to drugs. For instance, pregnant bodies can more quickly clear out medications such as glyburide, used during diabetes in pregnancy to stabilize high blood-sugar levels, which can be toxic to the fetus and harmful to women. That means a regular dose of the drug may not be enough to control blood sugar and prevent poor outcomes.
Pregnant patients also may be reluctant to take needed drugs for underlying conditions (and doctors may be hesitant to prescribe them), which in turn can cause more harm to the woman and fetus than had they been treated. For example, women who have severe asthma attacks while pregnant are at a higher risk of having low-birthweight babies, and pregnant women with uncontrolled diabetes in early pregnancy have more than four times the risk of birth defects.
Current clinical trials involving pregnant women are assessing treatments for obstructive sleep apnea, postpartum hemorrhage, lupus, and diabetes.
For Kate O'Brien, taking medication during her pregnancy was a matter of life and death. A freelance video producer who lives in New Jersey, O'Brien was diagnosed with tuberculosis in 2015 after she became pregnant with her second child, a boy. Even as she signed hospital consent forms, she had no idea if the treatment would harm him.
"It's a really awful experience," said O'Brien, who now is active with We are TB, an advocacy and support network. "All they had to tell me about the medication was just that women have been taking it for a really long time all over the world. That was the best they could do."
More and more doctors, researchers and women's health organizations and advocates are calling that unacceptable.
By indicating that filling current knowledge gaps is "a critical public health need," the FDA is signaling its support for advancing research with pregnant women, said Lyerly, also co-founder of the Second Wave Initiative, which promotes fair representation of the health interests of pregnant women in biomedical research and policies. "It's a very important shift."
Research with pregnant women can be done ethically, Lyerly said, whether by systematically collecting data from those already taking medications or enrolling pregnant women in studies of drugs or vaccines in development.
Current clinical trials involving pregnant women are assessing treatments for obstructive sleep apnea, postpartum hemorrhage, lupus, and diabetes. Notable trials in development target malaria and HIV prevention in pregnancy.
"It clearly is doable to do this research, and test trials are important to provide evidence for treatment," Spong said. "If we don't have that evidence, we aren't making the best educated decisions for women."
After spaceflight record, NASA looks to protect astronauts on even longer trips
NASA astronaut Frank Rubio floats by the International Space Station’s “window to the world.” Yesterday, he returned from the longest single spaceflight by a U.S. astronaut on record - over one year. Exploring deep space will require even longer missions.
At T-minus six seconds, the main engines of the Atlantis Space Shuttle ignited, rattling its capsule “like a skyscraper in an earthquake,” according to astronaut Tom Jones, describing the 1988 launch. As the rocket lifted off and accelerated to three times the force of Earth's gravity, “It felt as if two of my friends were standing on my chest and wouldn’t get off.” But when Atlantis reached orbit, the main engines cut off, and the astronauts were suddenly weightless.
Since 1961, NASA has sent hundreds of astronauts into space while working to making their voyages safer and smoother. Yet, challenges remain. Weightlessness may look amusing when watched from Earth, but it has myriad effects on cognition, movement and other functions. When missions to space stretch to six months or longer, microgravity can impact astronauts’ health and performance, making it more difficult to operate their spacecraft.
Yesterday, NASA astronaut Frank Rubio returned to Earth after over one year, the longest single spaceflight for a U.S. astronaut. But this is just the start; longer and more complex missions into deep space loom ahead, from returning to the moon in 2025 to eventually sending humans to Mars. To ensure that these missions succeed, NASA is increasing efforts to study the biological effects and prevent harm.
The dangers of microgravity are real
A NASA report published in 2016 details a long list of incidents and near-misses caused – at least partly – by space-induced changes in astronauts’ vision and coordination. These issues make it harder to move with precision and to judge distance and velocity.
According to the report, in 1997, a resupply ship collided with the Mir space station, possibly because a crew member bumped into the commander during the final docking maneuver. This mishap caused significant damage to the space station.
Returns to Earth suffered from problems, too. The same report notes that touchdown speeds during the first 100 space shuttle landings were “outside acceptable limits. The fastest landing on record – 224 knots (258 miles) per hour – was linked to the commander’s momentary spatial disorientation.” Earlier, each of the six Apollo crews that landed on the moon had difficulty recognizing moon landmarks and estimating distances. For example, Apollo 15 landed in an unplanned area, ultimately straddling the rim of a five-foot deep crater on the moon, harming one of its engines.
Spaceflight causes unique stresses on astronauts’ brains and central nervous systems. NASA is working to reduce these harmful effects.
NASA
Space messes up your brain
In space, astronauts face the challenges of microgravity, ionizing radiation, social isolation, high workloads, altered circadian rhythms, monotony, confined living quarters and a high-risk environment. Among these issues, microgravity is one of the most consequential in terms of physiological changes. It changes the brain’s structure and its functioning, which can hurt astronauts’ performance.
The brain shifts upwards within the skull, displacing the cerebrospinal fluid, which reduces the brain’s cushioning. Essentially, the brain becomes crowded inside the skull like a pair of too-tight shoes.
That’s partly because of how being in space alters blood flow. On Earth, gravity pulls our blood and other internal fluids toward our feet, but our circulatory valves ensure that the fluids are evenly distributed throughout the body. In space, there’s not enough gravity to pull the fluids down, and they shift up, says Rachael D. Seidler, a physiologist specializing in spaceflight at the University of Florida and principal investigator on many space-related studies. The head swells and legs appear thinner, causing what astronauts call “puffy face chicken legs.”
“The brain changes at the structural and functional level,” says Steven Jillings, equilibrium and aerospace researcher at the University of Antwerp in Belgium. “The brain shifts upwards within the skull,” displacing the cerebrospinal fluid, which reduces the brain’s cushioning. Essentially, the brain becomes crowded inside the skull like a pair of too-tight shoes. Some of the displaced cerebrospinal fluid goes into cavities within the brain, called ventricles, enlarging them. “The remaining fluids pool near the chest and heart,” explains Jillings. After 12 consecutive months in space, one astronaut had a ventricle that was 25 percent larger than before the mission.
Some changes reverse themselves while others persist for a while. An example of a longer-lasting problem is spaceflight-induced neuro-ocular syndrome, which results in near-sightedness and pressure inside the skull. A study of approximately 300 astronauts shows near-sightedness affects about 60 percent of astronauts after long missions on the International Space Station (ISS) and more than 25 percent after spaceflights of only a few weeks.
Another long-term change could be the decreased ability of cerebrospinal fluid to clear waste products from the brain, Seidler says. That’s because compressing the brain also compresses its waste-removing glymphatic pathways, resulting in inflammation, vulnerability to injuries and worsening its overall health.
The effects of long space missions were best demonstrated on astronaut twins Scott and Mark Kelly. This NASA Twins Study showed multiple, perhaps permanent, changes in Scott after his 340-day mission aboard the ISS, compared to Mark, who remained on Earth. The differences included declines in Scott’s speed, accuracy and cognitive abilities that persisted longer than six months after returning to Earth in March 2016.
By the end of 2020, Scott’s cognitive abilities improved, but structural and physiological changes to his eyes still remained, he said in a BBC interview.
“It seems clear that the upward shift of the brain and compression of the surrounding tissues with ventricular expansion might not be a good thing,” Seidler says. “But, at this point, the long-term consequences to brain health and human performance are not really known.”

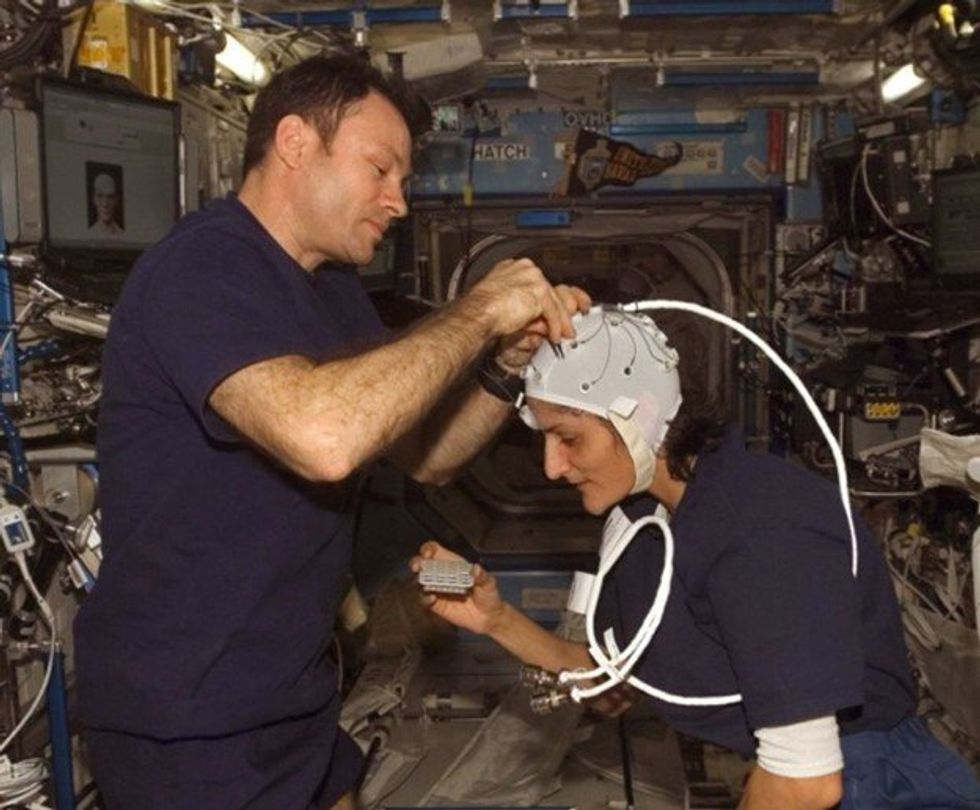
NASA astronaut Kate Rubins conducts a session for the Neuromapping investigation.
NASA
Staying sharp in space
To investigate how prolonged space travel affects the brain, NASA launched a new initiative called the Complement of Integrated Protocols for Human Exploration Research (CIPHER). “CIPHER investigates how long-duration spaceflight affects both brain structure and function,” says neurobehavioral scientist Mathias Basner at the University of Pennsylvania, a principal investigator for several NASA studies. “Through it, we can find out how the brain adapts to the spaceflight environment and how certain brain regions (behave) differently after – relative to before – the mission.”
To do this, he says, “Astronauts will perform NASA’s cognition test battery before, during and after six- to 12-month missions, and will also perform the same test battery in an MRI scanner before and after the mission. We have to make sure we better understand the functional consequences of spaceflight on the human brain before we can send humans safely to the moon and, especially, to Mars.”
As we go deeper into space, astronauts cognitive and physical functions will be even more important. “A trip to Mars will take about one year…and will introduce long communication delays,” Seidler says. “If you are on that mission and have a problem, it may take eight to 10 minutes for your message to reach mission control, and another eight to 10 minutes for the response to get back to you.” In an emergency situation, that may be too late for the response to matter.
“On a mission to Mars, astronauts will be exposed to stressors for unprecedented amounts of time,” Basner says. To counter them, NASA is considering the continuous use of artificial gravity during the journey, and Seidler is studying whether artificial gravity can reduce the harmful effects of microgravity. Some scientists are looking at precision brain stimulation as a way to improve memory and reduce anxiety due to prolonged exposure to radiation in space.
Other scientists are exploring how to protect neural stem cells (which create brain cells) from radiation damage, developing drugs to repair damaged brain cells and protect cells from radiation.
To boldly go where no astronauts have gone before, they must have optimal reflexes, vision and decision-making. In the era of deep space exploration, the brain—without a doubt—is the final frontier.
Additionally, NASA is scrutinizing each aspect of the mission, including astronaut exercise, nutrition and intellectual engagement. “We need to give astronauts meaningful work. We need to stimulate their sensory, cognitive and other systems appropriately,” Basner says, especially given their extreme confinement and isolation. The scientific experiments performed on the ISS – like studying how microgravity affects the ability of tissue to regenerate is a good example.
“We need to keep them engaged socially, too,” he continues. The ISS crew, for example, regularly broadcasts from space and answers prerecorded questions from students on Earth, and can engage with social media in real time. And, despite tight quarters, NASA is ensuring the crew capsule and living quarters on the moon or Mars include private space, which is critical for good mental health.
Exploring deep space builds on a foundation that began when astronauts first left the planet. With each mission, scientists learn more about spaceflight effects on astronauts’ bodies. NASA will be using these lessons to succeed with its plans to build science stations on the moon and, eventually, Mars.
“Through internally and externally led research, investigations implemented in space and in spaceflight simulations on Earth, we are striving to reduce the likelihood and potential impacts of neurostructural changes in future, extended spaceflight,” summarizes NASA scientist Alexandra Whitmire. To boldly go where no astronauts have gone before, they must have optimal reflexes, vision and decision-making. In the era of deep space exploration, the brain—without a doubt—is the final frontier.
A newly discovered brain cell may lead to better treatments for cognitive disorders
Swiss researchers have found a type of brain cell that appears to be a hybrid of the two other main types — and it could lead to new treatments for brain disorders.
Swiss researchers have discovered a third type of brain cell that appears to be a hybrid of the two other primary types — and it could lead to new treatments for many brain disorders.
The challenge: Most of the cells in the brain are either neurons or glial cells. While neurons use electrical and chemical signals to send messages to one another across small gaps called synapses, glial cells exist to support and protect neurons.
Astrocytes are a type of glial cell found near synapses. This close proximity to the place where brain signals are sent and received has led researchers to suspect that astrocytes might play an active role in the transmission of information inside the brain — a.k.a. “neurotransmission” — but no one has been able to prove the theory.
A new brain cell: Researchers at the Wyss Center for Bio and Neuroengineering and the University of Lausanne believe they’ve definitively proven that some astrocytes do actively participate in neurotransmission, making them a sort of hybrid of neurons and glial cells.
According to the researchers, this third type of brain cell, which they call a “glutamatergic astrocyte,” could offer a way to treat Alzheimer’s, Parkinson’s, and other disorders of the nervous system.
“Its discovery opens up immense research prospects,” said study co-director Andrea Volterra.
The study: Neurotransmission starts with a neuron releasing a chemical called a neurotransmitter, so the first thing the researchers did in their study was look at whether astrocytes can release the main neurotransmitter used by neurons: glutamate.
By analyzing astrocytes taken from the brains of mice, they discovered that certain astrocytes in the brain’s hippocampus did include the “molecular machinery” needed to excrete glutamate. They found evidence of the same machinery when they looked at datasets of human glial cells.
Finally, to demonstrate that these hybrid cells are actually playing a role in brain signaling, the researchers suppressed their ability to secrete glutamate in the brains of mice. This caused the rodents to experience memory problems.
“Our next studies will explore the potential protective role of this type of cell against memory impairment in Alzheimer’s disease, as well as its role in other regions and pathologies than those explored here,” said Andrea Volterra, University of Lausanne.
But why? The researchers aren’t sure why the brain needs glutamatergic astrocytes when it already has neurons, but Volterra suspects the hybrid brain cells may help with the distribution of signals — a single astrocyte can be in contact with thousands of synapses.
“Often, we have neuronal information that needs to spread to larger ensembles, and neurons are not very good for the coordination of this,” researcher Ludovic Telley told New Scientist.
Looking ahead: More research is needed to see how the new brain cell functions in people, but the discovery that it plays a role in memory in mice suggests it might be a worthwhile target for Alzheimer’s disease treatments.
The researchers also found evidence during their study that the cell might play a role in brain circuits linked to seizures and voluntary movements, meaning it’s also a new lead in the hunt for better epilepsy and Parkinson’s treatments.
“Our next studies will explore the potential protective role of this type of cell against memory impairment in Alzheimer’s disease, as well as its role in other regions and pathologies than those explored here,” said Volterra.