Why Haven’t Researchers Developed an HIV Vaccine or Cure Yet?
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
A blood test for analysis of HIV.
Last week, top experts on HIV/AIDS convened in Amsterdam for the 22nd International AIDS conference, and the mood was not great. Even though remarkable advances in treating HIV have led to effective management for many people living with the disease, and its overall incidence has declined, there are signs that the virus could make a troubling comeback.
"In a perfect world, we'd get a vaccine like the HPV vaccine that was 100% effective and I think that's ultimately what we're going to strive for."
Growing resistance to current HIV drugs, a population boom in Sub-Saharan Africa, and insufficient public health resources are all poised to contribute to a second AIDS pandemic, according to published reports.
Already, the virus is nowhere near under control. Though the infection rate has declined 47 percent since its peak in 1996, last year 1.8 million people became newly infected with HIV around the world, and 37 million people are currently living with it. About 1 million people die of AIDS every year, making it the fourth biggest killer in low-income countries.
Leapsmag Editor-in-Chief Kira Peikoff reached out to Dr. Carl Dieffenbach, Director of the Division of AIDS at the National Institute of Allergy and Infectious Diseases, to find out what the U.S. government is doing to develop an HIV vaccine and cure. This interview has been edited and condensed for clarity.
What is the general trajectory of research in HIV/AIDS today?
We can break it down to two specific domains: focus on treatment and cure, and prevention.
Let's start with people living with HIV. This is the area where we've had the most success over the past 30 plus years, because we've taken a disease that was essentially a death sentence and converted it through the development of medications to a treatable chronic disease.
The second half of this equation is, can we cure or create a functional cure for people living with HIV? And the definition of functional cure would be the absence of circulating virus in the body in the absence of therapy. Essentially the human body would control the HIV infection within the individual. That is a much more, very early research stage of discovery. There are some interesting signals but it's still in need of innovation.
I'd like to make a contrast between what we are able to do with a virus called Hepatitis C and what we can do with the virus HIV. Hep C, with 12 weeks of highly active antiviral therapy, we can cure 95 to 100% of infections. With HIV, we cannot do that. The difference is the behavior of the virus. HIV integrates into the host's genome. Hep C is an RNA virus that stays in the cytoplasm of the cell and never gets into the DNA.
On the prevention side, we have two strategies: The first is pre-exposure prophylaxis. Then of course, we have the need for a safe, effective and durable HIV vaccine, which is a very active area of discovery. We've had some spectacular success with RV144, and we're following up on that success, and other vaccines are in the pipeline. Whether they are sufficient to provide the level of durability and activity is not yet clear, but progress has been made and there's still the need for innovation.
The most important breakthrough in the past 5 to 10 years has been the discovery of broad neutralizing monoclonal antibodies. They are proteins that the body makes, and not everybody who's HIV infected makes these antibodies, but we've been able to clone out these antibodies from certain individuals that are highly potent, and when used either singly or in combination, can truly neutralize the vast majority of HIV strains. Can those be used by themselves as treatment or as prevention? That is the question.
Can you explain more about RV144 and why you consider it a success?
Prior to RV144, we had run a number of vaccine studies and nothing had ever statistically shown to be protective. RV144 showed a level of efficacy of about 31 percent, which was statistically significant. Not enough to take forward into other studies, but it allowed us to generate some ideas about why this worked, go back to the drawing board, and redesign the immunogens to optimize and test the next generation for this vaccine. We just recently opened that new study, the follow-up to RV144, called HVTN702. That's up and enrolling and moving along quite nicely.

Carl Dieffenbach, Director of the Division of AIDS at the National Institute of Allergy and Infectious Diseases
(Courtesy)
Where is that enrolling?
Primarily in Sub-Saharan Africa and South Africa.
When will you expect to see signals from that?
Between 2020 and 2021. It's complicated because the signal also takes into account the durability. After a certain time of vaccination, we're going to count up endpoints.
How would you explain the main scientific obstacle in the way of creating a very efficacious HIV vaccine?
Simply put, it's the black box of the human immune system. HIV employs a shield technology, and the virus is constantly changing its shield to protect itself, but there are some key parts of the virus that it cannot shield, so that's the trick – to be able to target that.
So, you're trying to find the Achilles' Heel of the virus?
Exactly. To make a flu vaccine or a Zika vaccine or even an Ebola vaccine, the virus is a little bit more forthcoming with the target. In HIV, the virus does everything in its power to hide the target, so we're dealing with a well-adapted [adversary] that actively avoids neutralization. That's the scientific challenge we face.
What's next?
On the vaccine side, we are currently performing, in collaboration with partners, two vaccine trials – HVTN702, which we talked about, and another one called 705. If either of those are highly successful, they would both require an additional phase 3 clinical trial before they could be licensed. This is an important but not final step. Then we would move into scale up to global vaccination. Those conversations have begun but they are not very far along and need additional attention.
What percent of people in the current trials would need to be protected to move on to phase 3?
Between 50 and 60 percent. That comes with this question of durability: how long does the vaccine last?
It also includes, can we simplify the vaccine regimen? The vaccines we're testing right now are multiple shots over a period of time. Can we get more like the polio or smallpox vaccine, a shot with a booster down the road?
We're dealing with sovereign nations. We're doing this in partnership, not as helicopter-type researchers.
If these current trials pan out, do you think kids in the developed world will end up getting an HIV vaccine one day? Or just people in-at risk areas?
That's a good question. I don't have an answer to that. In a perfect world, we'd get a vaccine like the HPV vaccine that was 100% effective and I think that's ultimately what we're going to strive for. That's where that second or third generation of vaccines that trigger broad neutralizing antibodies come in.
With any luck at all, globally, the combination of antiretroviral treatment, pre-exposure prophylaxis and other prevention and treatment strategies will lower the incidence rate where the HIV pandemic continues to wane, and we will then be able to either target the vaccine or roll it out in a way that is both cost effective and destigmatizing.
And also, what does the country want? We're dealing with sovereign nations. We're doing this in partnership, not as helicopter-type researchers.
How close do you think we are globally to eradicating HIV infections?
Eradication's a big word. It means no new infections. We are nowhere close to eradicating HIV. Whether or not we can continue to bend the curve on the epidemic and have less infections so that the total number of people continues to decline over time, I think we can achieve that if we had the political will. And that's not just the U.S. political will. That's the will of the world. We have the tools, albeit they're not perfect. But that's where a vaccine that is efficacious and simple to deliver could be the gamechanger.
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
New elevators could lift up our access to space
A space elevator would be cheaper and cleaner than using rockets
Story by Big Think
When people first started exploring space in the 1960s, it cost upwards of $80,000 (adjusted for inflation) to put a single pound of payload into low-Earth orbit.
A major reason for this high cost was the need to build a new, expensive rocket for every launch. That really started to change when SpaceX began making cheap, reusable rockets, and today, the company is ferrying customer payloads to LEO at a price of just $1,300 per pound.
This is making space accessible to scientists, startups, and tourists who never could have afforded it previously, but the cheapest way to reach orbit might not be a rocket at all — it could be an elevator.
The space elevator
The seeds for a space elevator were first planted by Russian scientist Konstantin Tsiolkovsky in 1895, who, after visiting the 1,000-foot (305 m) Eiffel Tower, published a paper theorizing about the construction of a structure 22,000 miles (35,400 km) high.
This would provide access to geostationary orbit, an altitude where objects appear to remain fixed above Earth’s surface, but Tsiolkovsky conceded that no material could support the weight of such a tower.
We could then send electrically powered “climber” vehicles up and down the tether to deliver payloads to any Earth orbit.
In 1959, soon after Sputnik, Russian engineer Yuri N. Artsutanov proposed a way around this issue: instead of building a space elevator from the ground up, start at the top. More specifically, he suggested placing a satellite in geostationary orbit and dropping a tether from it down to Earth’s equator. As the tether descended, the satellite would ascend. Once attached to Earth’s surface, the tether would be kept taut, thanks to a combination of gravitational and centrifugal forces.
We could then send electrically powered “climber” vehicles up and down the tether to deliver payloads to any Earth orbit. According to physicist Bradley Edwards, who researched the concept for NASA about 20 years ago, it’d cost $10 billion and take 15 years to build a space elevator, but once operational, the cost of sending a payload to any Earth orbit could be as low as $100 per pound.
“Once you reduce the cost to almost a Fed-Ex kind of level, it opens the doors to lots of people, lots of countries, and lots of companies to get involved in space,” Edwards told Space.com in 2005.
In addition to the economic advantages, a space elevator would also be cleaner than using rockets — there’d be no burning of fuel, no harmful greenhouse emissions — and the new transport system wouldn’t contribute to the problem of space junk to the same degree that expendable rockets do.
So, why don’t we have one yet?
Tether troubles
Edwards wrote in his report for NASA that all of the technology needed to build a space elevator already existed except the material needed to build the tether, which needs to be light but also strong enough to withstand all the huge forces acting upon it.
The good news, according to the report, was that the perfect material — ultra-strong, ultra-tiny “nanotubes” of carbon — would be available in just two years.
“[S]teel is not strong enough, neither is Kevlar, carbon fiber, spider silk, or any other material other than carbon nanotubes,” wrote Edwards. “Fortunately for us, carbon nanotube research is extremely hot right now, and it is progressing quickly to commercial production.”Unfortunately, he misjudged how hard it would be to synthesize carbon nanotubes — to date, no one has been able to grow one longer than 21 inches (53 cm).
Further research into the material revealed that it tends to fray under extreme stress, too, meaning even if we could manufacture carbon nanotubes at the lengths needed, they’d be at risk of snapping, not only destroying the space elevator, but threatening lives on Earth.
Looking ahead
Carbon nanotubes might have been the early frontrunner as the tether material for space elevators, but there are other options, including graphene, an essentially two-dimensional form of carbon that is already easier to scale up than nanotubes (though still not easy).
Contrary to Edwards’ report, Johns Hopkins University researchers Sean Sun and Dan Popescu say Kevlar fibers could work — we would just need to constantly repair the tether, the same way the human body constantly repairs its tendons.
“Using sensors and artificially intelligent software, it would be possible to model the whole tether mathematically so as to predict when, where, and how the fibers would break,” the researchers wrote in Aeon in 2018.
“When they did, speedy robotic climbers patrolling up and down the tether would replace them, adjusting the rate of maintenance and repair as needed — mimicking the sensitivity of biological processes,” they continued.Astronomers from the University of Cambridge and Columbia University also think Kevlar could work for a space elevator — if we built it from the moon, rather than Earth.
They call their concept the Spaceline, and the idea is that a tether attached to the moon’s surface could extend toward Earth’s geostationary orbit, held taut by the pull of our planet’s gravity. We could then use rockets to deliver payloads — and potentially people — to solar-powered climber robots positioned at the end of this 200,000+ mile long tether. The bots could then travel up the line to the moon’s surface.
This wouldn’t eliminate the need for rockets to get into Earth’s orbit, but it would be a cheaper way to get to the moon. The forces acting on a lunar space elevator wouldn’t be as strong as one extending from Earth’s surface, either, according to the researchers, opening up more options for tether materials.
“[T]he necessary strength of the material is much lower than an Earth-based elevator — and thus it could be built from fibers that are already mass-produced … and relatively affordable,” they wrote in a paper shared on the preprint server arXiv.
After riding up the Earth-based space elevator, a capsule would fly to a space station attached to the tether of the moon-based one.
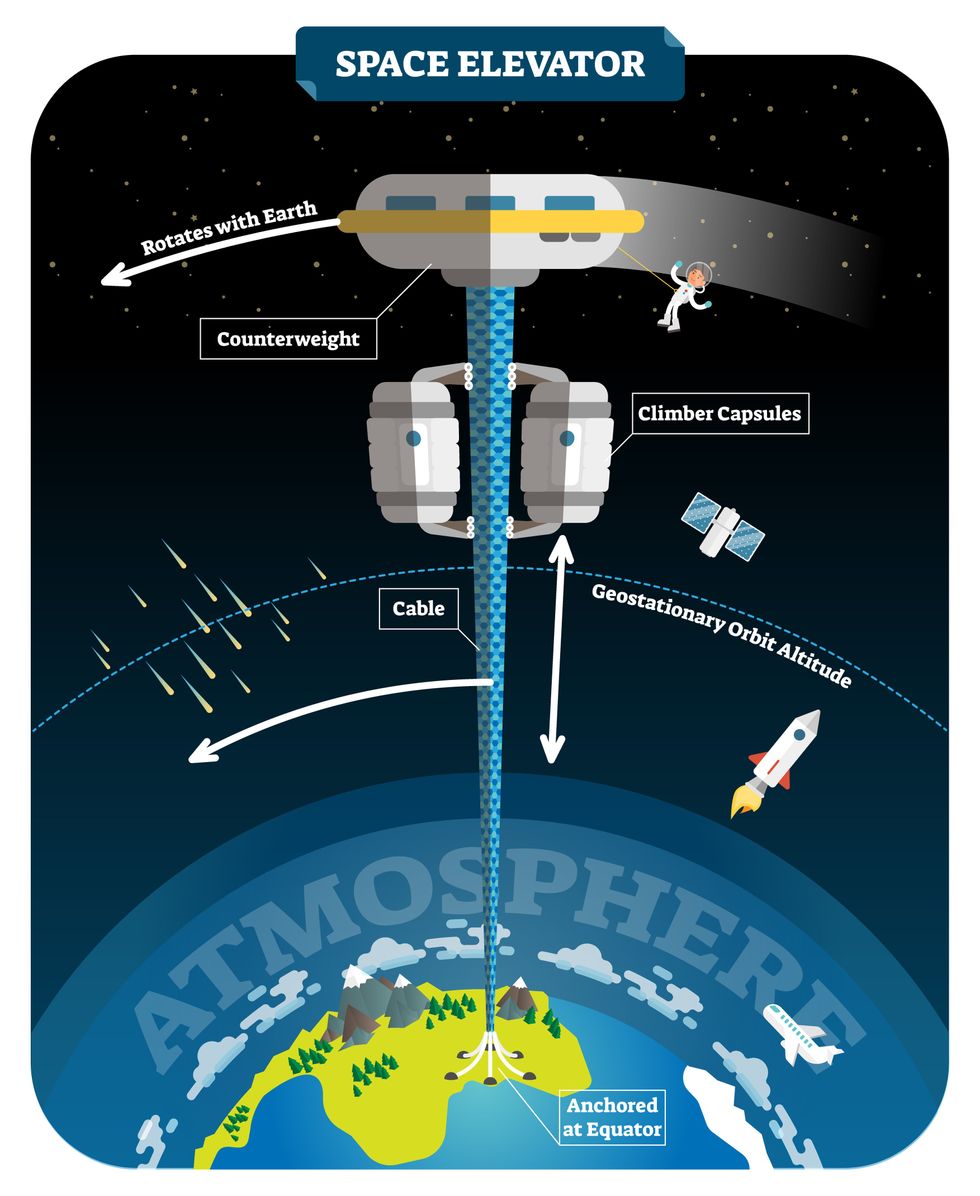
Electrically powered climber capsules could go up down the tether to deliver payloads to any Earth orbit.
Adobe Stock
Some Chinese researchers, meanwhile, aren’t giving up on the idea of using carbon nanotubes for a space elevator — in 2018, a team from Tsinghua University revealed that they’d developed nanotubes that they say are strong enough for a tether.
The researchers are still working on the issue of scaling up production, but in 2021, state-owned news outlet Xinhua released a video depicting an in-development concept, called “Sky Ladder,” that would consist of space elevators above Earth and the moon.
After riding up the Earth-based space elevator, a capsule would fly to a space station attached to the tether of the moon-based one. If the project could be pulled off — a huge if — China predicts Sky Ladder could cut the cost of sending people and goods to the moon by 96 percent.
The bottom line
In the 120 years since Tsiolkovsky looked at the Eiffel Tower and thought way bigger, tremendous progress has been made developing materials with the properties needed for a space elevator. At this point, it seems likely we could one day have a material that can be manufactured at the scale needed for a tether — but by the time that happens, the need for a space elevator may have evaporated.
Several aerospace companies are making progress with their own reusable rockets, and as those join the market with SpaceX, competition could cause launch prices to fall further.
California startup SpinLaunch, meanwhile, is developing a massive centrifuge to fling payloads into space, where much smaller rockets can propel them into orbit. If the company succeeds (another one of those big ifs), it says the system would slash the amount of fuel needed to reach orbit by 70 percent.
Even if SpinLaunch doesn’t get off the ground, several groups are developing environmentally friendly rocket fuels that produce far fewer (or no) harmful emissions. More work is needed to efficiently scale up their production, but overcoming that hurdle will likely be far easier than building a 22,000-mile (35,400-km) elevator to space.
This article originally appeared on Big Think, home of the brightest minds and biggest ideas of all time.

New tech aims to make the ocean healthier for marine life
Overabundance of dissolved carbon dioxide poses a threat to marine life. A new system detects elevated levels of the greenhouse gases and mitigates them on the spot.
A defunct drydock basin arched by a rusting 19th century steel bridge seems an incongruous place to conduct state-of-the-art climate science. But this placid and protected sliver of water connecting Brooklyn’s Navy Yard to the East River was just right for Garrett Boudinot to float a small dock topped with water carbon-sensing gear. And while his system right now looks like a trio of plastic boxes wired up together, it aims to mediate the growing ocean acidification problem, caused by overabundance of dissolved carbon dioxide.
Boudinot, a biogeochemist and founder of a carbon-management startup called Vycarb, is honing his method for measuring CO2 levels in water, as well as (at least temporarily) correcting their negative effects. It’s a challenge that’s been occupying numerous climate scientists as the ocean heats up, and as states like New York recognize that reducing emissions won’t be enough to reach their climate goals; they’ll have to figure out how to remove carbon, too.
To date, though, methods for measuring CO2 in water at scale have been either intensely expensive, requiring fancy sensors that pump CO2 through membranes; or prohibitively complicated, involving a series of lab-based analyses. And that’s led to a bottleneck in efforts to remove carbon as well.
But recently, Boudinot cracked part of the code for measurement and mitigation, at least on a small scale. While the rest of the industry sorts out larger intricacies like getting ocean carbon markets up and running and driving carbon removal at billion-ton scale in centralized infrastructure, his decentralized method could have important, more immediate implications.
Specifically, for shellfish hatcheries, which grow seafood for human consumption and for coastal restoration projects. Some of these incubators for oysters and clams and scallops are already feeling the negative effects of excess carbon in water, and Vycarb’s tech could improve outcomes for the larval- and juvenile-stage mollusks they’re raising. “We’re learning from these folks about what their needs are, so that we’re developing our system as a solution that’s relevant,” Boudinot says.
Ocean acidification can wreak havoc on developing shellfish, inhibiting their shells from growing and leading to mass die-offs.
Ocean waters naturally absorb CO2 gas from the atmosphere. When CO2 accumulates faster than nature can dissipate it, it reacts with H2O molecules, forming carbonic acid, H2CO3, which makes the water column more acidic. On the West Coast, acidification occurs when deep, carbon dioxide-rich waters upwell onto the coast. This can wreak havoc on developing shellfish, inhibiting their shells from growing and leading to mass die-offs; this happened, disastrously, at Pacific Northwest oyster hatcheries in 2007.
This type of acidification will eventually come for the East Coast, too, says Ryan Wallace, assistant professor and graduate director of environmental studies and sciences at Long Island’s Adelphi University, who studies acidification. But at the moment, East Coast acidification has other sources: agricultural runoff, usually in the form of nitrogen, and human and animal waste entering coastal areas. These excess nutrient loads cause algae to grow, which isn’t a problem in and of itself, Wallace says; but when algae die, they’re consumed by bacteria, whose respiration in turn bumps up CO2 levels in water.
“Unfortunately, this is occurring at the bottom [of the water column], where shellfish organisms live and grow,” Wallace says. Acidification on the East Coast is minutely localized, occurring closest to where nutrients are being released, as well as seasonally; at least one local shellfish farm, on Fishers Island in the Long Island Sound, has contended with its effects.

The second Vycarb pilot, ready to be installed at the East Hampton shellfish hatchery.
Courtesy of Vycarb
Besides CO2, ocean water contains two other forms of dissolved carbon — carbonate (CO3-) and bicarbonate (HCO3) — at all times, at differing levels. At low pH (acidic), CO2 prevails; at medium pH, HCO3 is the dominant form; at higher pH, CO3 dominates. Boudinot’s invention is the first real-time measurement for all three, he says. From the dock at the Navy Yard, his pilot system uses carefully calibrated but low-cost sensors to gauge the water’s pH and its corresponding levels of CO2. When it detects elevated levels of the greenhouse gas, the system mitigates it on the spot. It does this by adding a bicarbonate powder that’s a byproduct of agricultural limestone mining in nearby Pennsylvania. Because the bicarbonate powder is alkaline, it increases the water pH and reduces the acidity. “We drive a chemical reaction to increase the pH to convert greenhouse gas- and acid-causing CO2 into bicarbonate, which is HCO3,” Boudinot says. “And HCO3 is what shellfish and fish and lots of marine life prefers over CO2.”
This de-acidifying “buffering” is something shellfish operations already do to water, usually by adding soda ash (NaHCO3), which is also alkaline. Some hatcheries add soda ash constantly, just in case; some wait till acidification causes significant problems. Generally, for an overly busy shellfish farmer to detect acidification takes time and effort. “We’re out there daily, taking a look at the pH and figuring out how much we need to dose it,” explains John “Barley” Dunne, director of the East Hampton Shellfish Hatchery on Long Island. “If this is an automatic system…that would be much less labor intensive — one less thing to monitor when we have so many other things we need to monitor.”
Across the Sound at the hatchery he runs, Dunne annually produces 30 million hard clams, 6 million oysters, and “if we’re lucky, some years we get a million bay scallops,” he says. These mollusks are destined for restoration projects around the town of East Hampton, where they’ll create habitat, filter water, and protect the coastline from sea level rise and storm surge. So far, Dunne’s hatchery has largely escaped the ill effects of acidification, although his bay scallops are having a finicky year and he’s checking to see if acidification might be part of the problem. But “I think it's important to have these solutions ready-at-hand for when the time comes,” he says. That’s why he’s hosting a second, 70-liter Vycarb pilot starting this summer on a dock adjacent to his East Hampton operation; it will amp up to a 50,000 liter-system in a few months.
If it can buffer water over a large area, absolutely this will benefit natural spawns. -- John “Barley” Dunne.
Boudinot hopes this new pilot will act as a proof of concept for hatcheries up and down the East Coast. The area from Maine to Nova Scotia is experiencing the worst of Atlantic acidification, due in part to increased Arctic meltwater combining with Gulf of St. Lawrence freshwater; that decreases saturation of calcium carbonate, making the water more acidic. Boudinot says his system should work to adjust low pH regardless of the cause or locale. The East Hampton system will eventually test and buffer-as-necessary the water that Dunne pumps from the Sound into 100-gallon land-based tanks where larvae grow for two weeks before being transferred to an in-Sound nursery to plump up.
Dunne says this could have positive effects — not only on his hatchery but on wild shellfish populations, too, reducing at least one stressor their larvae experience (others include increasing water temperatures and decreased oxygen levels). “If it can buffer water over a large area, absolutely this will [benefit] natural spawns,” he says.
No one believes the Vycarb model — even if it proves capable of functioning at much greater scale — is the sole solution to acidification in the ocean. Wallace says new water treatment plants in New York City, which reduce nitrogen released into coastal waters, are an important part of the equation. And “certainly, some green infrastructure would help,” says Boudinot, like restoring coastal and tidal wetlands to help filter nutrient runoff.
In the meantime, Boudinot continues to collect data in advance of amping up his own operations. Still unknown is the effect of releasing huge amounts of alkalinity into the ocean. Boudinot says a pH of 9 or higher can be too harsh for marine life, plus it can also trigger a release of CO2 from the water back into the atmosphere. For a third pilot, on Governor’s Island in New York Harbor, Vycarb will install yet another system from which Boudinot’s team will frequently sample to analyze some of those and other impacts. “Let's really make sure that we know what the results are,” he says. “Let's have data to show, because in this carbon world, things behave very differently out in the real world versus on paper.”

