Will Blockchain Technology Usher in a Healthcare Data Revolution?

Digital blockchain concept.
The hacker collective known as the Dark Overlord first surfaced in June 2016, when it advertised more than 600,000 patient files from three U.S. healthcare organizations for sale on the dark web. The group, which also attempted to extort ransom from its victims, soon offered another 9 million records pilfered from health insurance companies and provider networks across the country.
Since 2009, federal regulators have counted nearly 5,000 major data breaches in the United States alone, affecting some 260 million individuals.
Last October, apparently seeking publicity as well as cash, the hackers stole a trove of potentially scandalous data from a celebrity plastic surgery clinic in London—including photos of in-progress genitalia- and breast-enhancement surgeries. "We have TBs [terabytes] of this shit. Databases, names, everything," a gang representative told a reporter. "There are some royal families in here."
Bandits like these are prowling healthcare's digital highways in growing numbers. Since 2009, federal regulators have counted nearly 5,000 major data breaches in the United States alone, affecting some 260 million individuals. Although hacker incidents represent less than 20 percent of the total breaches, they account for almost 80 percent of the affected patients. Such attacks expose patients to potential blackmail or identity theft, enable criminals to commit medical fraud or file false tax returns, and may even allow hostile state actors to sabotage electric grids or other infrastructure by e-mailing employees malware disguised as medical notices. According to the consulting agency Accenture, data theft will cost the healthcare industry $305 billion between 2015 and 2019, with annual totals doubling from $40 billion to $80 billion.
Blockchain could put patients in control of their own data, empowering them to access, share, and even sell their medical information as they see fit.
One possible solution to this crisis involves radically retooling the way healthcare data is stored and shared—by using blockchain, the still-emerging information technology that underlies cryptocurrencies such as Bitcoin. And blockchain-enabled IT systems, boosters say, could do much more than prevent the theft of medical data. Such networks could revolutionize healthcare delivery on many levels, creating efficiencies that would reduce medical errors, improve coordination between providers, drive down costs, and give researchers unprecedented insights into patterns of disease. Perhaps most transformative, blockchain could put patients in control of their own data, empowering them to access, share, and even sell their medical information as they see fit. Widespread adoption could result in "a new kind of healthcare economy, in which data and services are quantifiable and exchangeable, with strong guarantees around both the security and privacy of sensitive information," wrote W. Brian Smith, chief scientist of healthcare-blockchain startup PokitDok, in a recent white paper.
Around the world, entrepreneurs, corporations, and government agencies are hopping aboard the blockchain train. A survey by the IBM Institute for Business Value, released in late 2016, found that 16 percent of healthcare executives in 16 countries planned to begin implementing some form of the technology in the coming year; 90 percent planned to launch a pilot program in the next two years. In 2017, Estonia became the first country to switch its medical-records system to a blockchain-based framework. Great Britain and Dubai are exploring a similar move. Yet in countries with more fragmented health systems, most notably the U.S., the challenges remain formidable. Some of the most advanced healthcare applications envisioned for blockchain, moreover, raise technological and ethical questions whose answers may not arrive anytime soon.
By creating a detailed, comprehensive, and immutable timeline of medical transactions, blockchain-based recordkeeping could help providers gauge a patient's long-term health patterns in a way that's never before been possible.
What Exactly Is Blockchain, Anyway?
To understand the buzz around blockchain, it's necessary to grasp (at least loosely) how the technology works. Ordinary digital recordkeeping systems rely on a central administrator that acts as gatekeeper to a treasury of data; if you can sneak past the guard, you can often gain access to the entire hoard, and your intrusion may go undetected indefinitely. Blockchain, by contrast, employs a network of synchronized, replicated databases. Information is scattered among these nodes, rather than on a single server, and is exchanged through encrypted, peer-to-peer pathways. Each transaction is visible to every computer on the network, and must be approved by a majority in order to be successfully completed. Each batch of transactions, or "block," is date- and time-stamped, marked with the user's identity, and given a cryptographic code, which is posted to every node. These blocks form a "chain," preserved in an electronic ledger, that can be read by all users but can't be edited. Any unauthorized access, or attempt at tampering, can be quickly neutralized by these overlapping safeguards. Even if a hacker managed to break into the system, penetrating deeply would be extraordinarily difficult.
Because blockchain technology shares transaction records throughout a network, it could eliminate communication bottlenecks between different components of the healthcare system (primary care physicians, specialists, nurses, and so on). And because blockchain-based systems are designed to incorporate programs known as "smart contracts," which automate functions previously requiring human intervention, they could reduce dangerous slipups as well as tedious and costly paperwork. For example, when a patient gets a checkup, sees a specialist, and fills a prescription, all these actions could be automatically recorded on his or her electronic health record (EHR), checked for errors, submitted for billing, and entered on insurance claims—which could be adjudicated and reimbursed automatically as well. "Blockchain has the potential to remove a lot of intermediaries from existing workflows, whether digital or nondigital," says Kamaljit Behera, an industry analyst for the consulting firm Frost & Sullivan.
The possible upsides don't end there. By creating a detailed, comprehensive, and immutable timeline of medical transactions, blockchain-based recordkeeping could help providers gauge a patient's long-term health patterns in a way that's never before been possible. In addition to data entered by their caregivers, individuals could use app-based technologies or wearables to transmit other information to their records, such as diet, exercise, and sleep patterns, adding new depth to their medical portraits.
Many experts expect healthcare blockchain to take root more slowly in the U.S. than in nations with government-run national health services.
Smart contracts could also allow patients to specify who has access to their data. "If you get an MRI and want your orthopedist to see it, you can add him to your network instead of carrying a CD into his office," explains Andrew Lippman, associate director of the MIT Media Lab, who helped create a prototype healthcare blockchain system called MedRec that's currently being tested at Beth Israel Deaconess Hospital in Boston. "Or you might make a smart contract to allow your son or daughter to access your healthcare records if something happens to you." Another option: permitting researchers to analyze your data for scientific purposes, whether anonymously or with your name attached.
The Recent History, and Looking Ahead
Over the past two years, a crowd of startups has begun vying for a piece of the emerging healthcare blockchain market. Some, like PokitDok and Atlanta-based Patientory, plan to mint proprietary cryptocurrencies, which investors can buy in lieu of stock, medical providers may earn as a reward for achieving better outcomes, and patients might score for meeting wellness goals or participating in clinical trials. (Patientory's initial coin offering, or ICO, raised more than $7 million in three days.) Several fledgling healthcare-blockchain companies have found powerful corporate partners: Intel for Silicon Valley's PokitDok, Kaiser Permanente for Patientory, Philips for Los Angeles-based Gem Health. At least one established provider network, Change Healthcare, is developing blockchain-based systems of its own. Two months ago, Change launched what it calls the first "enterprise-scale" blockchain network in U.S. healthcare—a system to track insurance claim submissions and remittances.
No one, however, has set a roll-out date for a full-blown, blockchain-based EHR system in this country. "We have yet to see anything move from the pilot phase to some kind of production status," says Debbie Bucci, an IT architect in the federal government's Office of the National Coordinator for Health Information Technology. Indeed, many experts expect healthcare blockchain to take root more slowly here than in nations with government-run national health services. In America, a typical patient may have dealings with a family doctor who keeps everything on paper, an assortment of hospitals that use different EHR systems, and an insurer whose system for processing claims is separate from that of the healthcare providers. To help bridge these gaps, a consortium called the Hyperledger Healthcare Working Group (which includes many of the leading players in the field) is developing standard protocols for blockchain interoperability and other functions. Adding to the complexity is the federal Health Insurance and Portability Act (HIPAA), which governs who can access patient data and under what circumstances. "Healthcare blockchain is in a very nascent stage," says Behera. "Coming up with regulations and other guidelines, and achieving large-scale implementation, will take some time."
The ethical implications of buying and selling personal genomic data in an electronic marketplace are doubtless open to debate.
How long? Behera, like other analysts, estimates that relatively simple applications, such as revenue-cycle management systems, could become commonplace in the next five years. More ambitious efforts might reach fruition in a decade or so. But once the infrastructure for healthcare blockchain is fully established, its uses could go far beyond keeping better EHRs.
A handful of scientists and entrepreneurs are already working to develop one visionary application: managing genomic data. Last month, Harvard University geneticist George Church—one of the most influential figures in his discipline—launched a business called Nebula Genomics. It aims to set up an exchange in which individuals can use "Neptune tokens" to purchase DNA sequencing, which will be stored in the company's blockchain-based system; research groups will be able to pay clients for their data using the same cryptocurrency. Luna DNA, founded by a team of biotech veterans in San Diego, plans a similar service, as does a Moscow-based startup called the Zenome Project.
Hossein Rahnama, CEO of the mobile-tech company Flybits and director of research at the Ryerson Centre for Cloud and Context-Aware Computing in Toronto, envisions a more personalized way of sharing genomic data via blockchain. His firm is working with a U.S. insurance company to develop a service that would allow clients in their 20s and 30s to connect with people in their 70s or 80s with similar genomes. The young clients would learn how the elders' lifestyle choices had influenced their health, so that they could modify their own habits accordingly. "It's intergenerational wisdom-sharing," explains Rahnama, who is 38. "I would actually pay to be a part of that network."
The ethical implications of buying and selling personal genomic data in an electronic marketplace are doubtless open to debate. Such commerce could greatly expand the pool of subjects for research in many areas of medicine, enabling the kinds of breakthroughs that only Big Data can provide. Yet it could also lead millions to surrender the most private information of all—the secrets of their cells—to buyers with less benign intentions. The Dark Overlord, one might argue, could not hope for a more satisfying victory.
These scenarios, however, are pure conjecture. After the first web page was posted, in 1991, Lippman observes, "a whole universe developed that you couldn't have imagined on Day 1." The same, he adds, is likely true for healthcare blockchain. "Our vision is to make medical records useful for you and for society, and to give you more control over your own identity. Time will tell."
Nobel Prize goes to technology for mRNA vaccines
Katalin Karikó, pictured, and Drew Weissman won the Nobel Prize for advances in mRNA research that led to the first Covid vaccines.
When Drew Weissman received a call from Katalin Karikó in the early morning hours this past Monday, he assumed his longtime research partner was calling to share a nascent, nagging idea. Weissman, a professor of medicine at the Perelman School of Medicine at the University of Pennsylvania, and Karikó, a professor at Szeged University and an adjunct professor at UPenn, both struggle with sleep disturbances. Thus, middle-of-the-night discourses between the two, often over email, has been a staple of their friendship. But this time, Karikó had something more pressing and exciting to share: They had won the 2023 Nobel Prize in Physiology or Medicine.
The work for which they garnered the illustrious award and its accompanying $1,000,000 cash windfall was completed about two decades ago, wrought through long hours in the lab over many arduous years. But humanity collectively benefited from its life-saving outcome three years ago, when both Moderna and Pfizer/BioNTech’s mRNA vaccines against COVID were found to be safe and highly effective at preventing severe disease. Billions of doses have since been given out to protect humans from the upstart viral scourge.
“I thought of going somewhere else, or doing something else,” said Katalin Karikó. “I also thought maybe I’m not good enough, not smart enough. I tried to imagine: Everything is here, and I just have to do better experiments.”
Unlocking the power of mRNA
Weissman and Karikó unlocked mRNA vaccines for the world back in the early 2000s when they made a key breakthrough. Messenger RNA molecules are essentially instructions for cells’ ribosomes to make specific proteins, so in the 1980s and 1990s, researchers started wondering if sneaking mRNA into the body could trigger cells to manufacture antibodies, enzymes, or growth agents for protecting against infection, treating disease, or repairing tissues. But there was a big problem: injecting this synthetic mRNA triggered a dangerous, inflammatory immune response resulting in the mRNA’s destruction.
While most other researchers chose not to tackle this perplexing problem to instead pursue more lucrative and publishable exploits, Karikó stuck with it. The choice sent her academic career into depressing doldrums. Nobody would fund her work, publications dried up, and after six years as an assistant professor at the University of Pennsylvania, Karikó got demoted. She was going backward.
“I thought of going somewhere else, or doing something else,” Karikó told Stat in 2020. “I also thought maybe I’m not good enough, not smart enough. I tried to imagine: Everything is here, and I just have to do better experiments.”
A tale of tenacity
Collaborating with Drew Weissman, a new professor at the University of Pennsylvania, in the late 1990s helped provide Karikó with the tenacity to continue. Weissman nurtured a goal of developing a vaccine against HIV-1, and saw mRNA as a potential way to do it.
“For the 20 years that we’ve worked together before anybody knew what RNA is, or cared, it was the two of us literally side by side at a bench working together,” Weissman said in an interview with Adam Smith of the Nobel Foundation.
In 2005, the duo made their 2023 Nobel Prize-winning breakthrough, detailing it in a relatively small journal, Immunity. (Their paper was rejected by larger journals, including Science and Nature.) They figured out that chemically modifying the nucleoside bases that make up mRNA allowed the molecule to slip past the body’s immune defenses. Karikó and Weissman followed up that finding by creating mRNA that’s more efficiently translated within cells, greatly boosting protein production. In 2020, scientists at Moderna and BioNTech (where Karikó worked from 2013 to 2022) rushed to craft vaccines against COVID, putting their methods to life-saving use.
The future of vaccines
Buoyed by the resounding success of mRNA vaccines, scientists are now hurriedly researching ways to use mRNA medicine against other infectious diseases, cancer, and genetic disorders. The now ubiquitous efforts stand in stark contrast to Karikó and Weissman’s previously unheralded struggles years ago as they doggedly worked to realize a shared dream that so many others shied away from. Katalin Karikó and Drew Weissman were brave enough to walk a scientific path that very well could have ended in a dead end, and for that, they absolutely deserve their 2023 Nobel Prize.
This article originally appeared on Big Think, home of the brightest minds and biggest ideas of all time.

Scientists turn pee into power in Uganda
With conventional fuel cells as their model, researchers learned to use similar chemical reactions to make a fuel from microbes in pee.
At the edge of a dirt road flanked by trees and green mountains outside the town of Kisoro, Uganda, sits the concrete building that houses Sesame Girls School, where girls aged 11 to 19 can live, learn and, at least for a while, safely use a toilet. In many developing regions, toileting at night is especially dangerous for children. Without electrical power for lighting, kids may fall into the deep pits of the latrines through broken or unsteady floorboards. Girls are sometimes assaulted by men who hide in the dark.
For the Sesame School girls, though, bright LED lights, connected to tiny gadgets, chased the fears away. They got to use new, clean toilets lit by the power of their own pee. Some girls even used the light provided by the latrines to study.
Urine, whether animal or human, is more than waste. It’s a cheap and abundant resource. Each day across the globe, 8.1 billion humans make 4 billion gallons of pee. Cows, pigs, deer, elephants and other animals add more. By spending money to get rid of it, we waste a renewable resource that can serve more than one purpose. Microorganisms that feed on nutrients in urine can be used in a microbial fuel cell that generates electricity – or "pee power," as the Sesame girls called it.
Plus, urine contains water, phosphorus, potassium and nitrogen, the key ingredients plants need to grow and survive. Human urine could replace about 25 percent of current nitrogen and phosphorous fertilizers worldwide and could save water for gardens and crops. The average U.S. resident flushes a toilet bowl containing only pee and paper about six to seven times a day, which adds up to about 3,500 gallons of water down per year. Plus cows in the U.S. produce 231 gallons of the stuff each year.
Pee power
A conventional fuel cell uses chemical reactions to produce energy, as electrons move from one electrode to another to power a lightbulb or phone. Ioannis Ieropoulos, a professor and chair of Environmental Engineering at the University of Southampton in England, realized the same type of reaction could be used to make a fuel from microbes in pee.
Bacterial species like Shewanella oneidensis and Pseudomonas aeruginosa can consume carbon and other nutrients in urine and pop out electrons as a result of their digestion. In a microbial fuel cell, one electrode is covered in microbes, immersed in urine and kept away from oxygen. Another electrode is in contact with oxygen. When the microbes feed on nutrients, they produce the electrons that flow through the circuit from one electrod to another to combine with oxygen on the other side. As long as the microbes have fresh pee to chomp on, electrons keep flowing. And after the microbes are done with the pee, it can be used as fertilizer.
These microbes are easily found in wastewater treatment plants, ponds, lakes, rivers or soil. Keeping them alive is the easy part, says Ieropoulos. Once the cells start producing stable power, his group sequences the microbes and keeps using them.
Like many promising technologies, scaling these devices for mass consumption won’t be easy, says Kevin Orner, a civil engineering professor at West Virginia University. But it’s moving in the right direction. Ieropoulos’s device has shrunk from the size of about three packs of cards to a large glue stick. It looks and works much like a AAA battery and produce about the same power. By itself, the device can barely power a light bulb, but when stacked together, they can do much more—just like photovoltaic cells in solar panels. His lab has produced 1760 fuel cells stacked together, and with manufacturing support, there’s no theoretical ceiling, he says.
Although pure urine produces the most power, Ieropoulos’s devices also work with the mixed liquids of the wastewater treatment plants, so they can be retrofit into urban wastewater utilities.
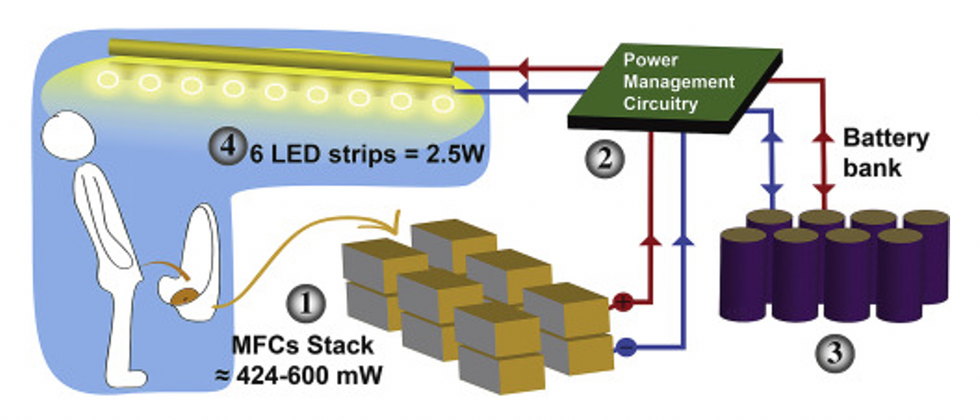
This image shows how the pee-powered system works. Pee feeds bacteria in the stack of fuel cells (1), which give off electrons (2) stored in parallel cylindrical cells (3). These cells are connected to a voltage regulator (4), which smooths out the electrical signal to ensure consistent power to the LED strips lighting the toilet.
Courtesy Ioannis Ieropoulos
Key to the long-term success of any urine reclamation effort, says Orner, is avoiding what he calls “parachute engineering”—when well-meaning scientists solve a problem with novel tech and then abandon it. “The way around that is to have either the need come from the community or to have an organization in a community that is committed to seeing a project operate and maintained,” he says.
Success with urine reclamation also depends on the economy. “If energy prices are low, it may not make sense to recover energy,” says Orner. “But right now, fertilizer prices worldwide are generally pretty high, so it may make sense to recover fertilizer and nutrients.” There are obstacles, too, such as few incentives for builders to incorporate urine recycling into new construction. And any hiccups like leaks or waste seepage will cost builders money and reputation. Right now, Orner says, the risks are just too high.
Despite the challenges, Ieropoulos envisions a future in which urine is passed through microbial fuel cells at wastewater treatment plants, retrofitted septic tanks, and building basements, and is then delivered to businesses to use as agricultural fertilizers. Although pure urine produces the most power, Ieropoulos’s devices also work with the mixed liquids of the wastewater treatment plants, so they can be retrofitted into urban wastewater utilities where they can make electricity from the effluent. And unlike solar cells, which are a common target of theft in some areas, nobody wants to steal a bunch of pee.
When Ieropoulos’s team returned to wrap up their pilot project 18 months later, the school’s director begged them to leave the fuel cells in place—because they made a major difference in students’ lives. “We replaced it with a substantial photovoltaic panel,” says Ieropoulos, They couldn’t leave the units forever, he explained, because of intellectual property reasons—their funders worried about theft of both the technology and the idea. But the photovoltaic replacement could be stolen, too, leaving the girls in the dark.
The story repeated itself at another school, in Nairobi, Kenya, as well as in an informal settlement in Durban, South Africa. Each time, Ieropoulos vowed to return. Though the pandemic has delayed his promise, he is resolute about continuing his work—it is a moral and legal obligation. “We've made a commitment to ourselves and to the pupils,” he says. “That's why we need to go back.”
Urine as fertilizer
Modern day industrial systems perpetuate the broken cycle of nutrients. When plants grow, they use up nutrients the soil. We eat the plans and excrete some of the nutrients we pass them into rivers and oceans. As a result, farmers must keep fertilizing the fields while our waste keeps fertilizing the waterways, where the algae, overfertilized with nitrogen, phosphorous and other nutrients grows out of control, sucking up oxygen that other marine species need to live. Few global communities remain untouched by the related challenges this broken chain create: insufficient clean water, food, and energy, and too much human and animal waste.
The Rich Earth Institute in Vermont runs a community-wide urine nutrient recovery program, which collects urine from homes and businesses, transports it for processing, and then supplies it as fertilizer to local farms.
One solution to this broken cycle is reclaiming urine and returning it back to the land. The Rich Earth Institute in Vermont is one of several organizations around the world working to divert and save urine for agricultural use. “The urine produced by an adult in one day contains enough fertilizer to grow all the wheat in one loaf of bread,” states their website.
Notably, while urine is not entirely sterile, it tends to harbor fewer pathogens than feces. That’s largely because urine has less organic matter and therefore less food for pathogens to feed on, but also because the urinary tract and the bladder have built-in antimicrobial defenses that kill many germs. In fact, the Rich Earth Institute says it’s safe to put your own urine onto crops grown for home consumption. Nonetheless, you’ll want to dilute it first because pee usually has too much nitrogen and can cause “fertilizer burn” if applied straight without dilution. Other projects to turn urine into fertilizer are in progress in Niger, South Africa, Kenya, Ethiopia, Sweden, Switzerland, The Netherlands, Australia, and France.
Eleven years ago, the Institute started a program that collects urine from homes and businesses, transports it for processing, and then supplies it as fertilizer to local farms. By 2021, the program included 180 donors producing over 12,000 gallons of urine each year. This urine is helping to fertilize hay fields at four partnering farms. Orner, the West Virginia professor, sees it as a success story. “They've shown how you can do this right--implementing it at a community level scale."

