Your Digital Avatar May One Day Get Sick Before You Do

Artificial neurons in a concept of artificial intelligence.
Artificial intelligence is everywhere, just not in the way you think it is.
These networks, loosely designed after the human brain, are interconnected computers that have the ability to "learn."
"There's the perception of AI in the glossy magazines," says Anders Kofod-Petersen, a professor of Artificial Intelligence at the Norwegian University of Science and Technology. "That's the sci-fi version. It resembles the small guy in the movie AI. It might be benevolent or it might be evil, but it's generally intelligent and conscious."
"And this is, of course, as far from the truth as you can possibly get."
What Exactly Is Artificial Intelligence, Anyway?
Let's start with how you got to this piece. You likely came to it through social media. Your Facebook account, Twitter feed, or perhaps a Google search. AI influences all of those things, machine learning helping to run the algorithms that decide what you see, when, and where. AI isn't the little humanoid figure; it's the system that controls the figure.
"AI is being confused with robotics," Eleonore Pauwels, Director of the Anticipatory Intelligence Lab with the Science and Technology Innovation Program at the Wilson Center, says. "What AI is right now is a data optimization system, a very powerful data optimization system."
The revolution in recent years hasn't come from the method scientists and other researchers use. The general ideas and philosophies have been around since the late 1960s. Instead, the big change has been the dramatic increase in computing power, primarily due to the development of neural networks. These networks, loosely designed after the human brain, are interconnected computers that have the ability to "learn." An AI, for example, can be taught to spot a picture of a cat by looking at hundreds of thousands of pictures that have been labeled "cat" and "learning" what a cat looks like. Or an AI can beat a human at Go, an achievement that just five years ago Kofod-Petersen thought wouldn't be accomplished for decades.
"It's very difficult to argue that something is intelligent if it can't learn, and these algorithms are getting pretty good at learning stuff. What they are not good at is learning how to learn."
Medicine is the field where this expertise in perception tasks might have the most influence. It's already having an impact as iPhones use AI to detect cancer, Apple watches alert the wearer to a heart problem, AI spots tuberculosis and the spread of breast cancer with a higher accuracy than human doctors, and more. Every few months, another study demonstrates more possibility. (The New Yorker published an article about medicine and AI last year, so you know it's a serious topic.)
But this is only the beginning. "I personally think genomics and precision medicine is where AI is going to be the biggest game-changer," Pauwels says. "It's going to completely change how we think about health, our genomes, and how we think about our relationship between our genotype and phenotype."
The Fundamental Breakthrough That Must Be Solved
To get there, however, researchers will need to make another breakthrough, and there's debate about how long that will take. Kofod-Petersen explains: "If we want to move from this narrow intelligence to this broader intelligence, that's a very difficult problem. It basically boils down to that we haven't got a clue about what intelligence actually is. We don't know what intelligence means in a biological sense. We think we might recognize it but we're not completely sure. There isn't a working definition. We kind of agree with the biologists that learning is an aspect of it. It's very difficult to argue that something is intelligent if it can't learn, and these algorithms are getting pretty good at learning stuff. What they are not good at is learning how to learn. They can learn specific tasks but we haven't approached how to teach them to learn to learn."
In other words, current AI is very, very good at identifying that a picture of a cat is, in fact, a cat – and getting better at doing so at an incredibly rapid pace – but the system only knows what a "cat" is because that's what a programmer told it a furry thing with whiskers and two pointy ears is called. If the programmer instead decided to label the training images as "dogs," the AI wouldn't say "no, that's a cat." Instead, it would simply call a furry thing with whiskers and two pointy ears a dog. AI systems lack the explicit inference that humans do effortlessly, almost without thinking.
Pauwels believes that the next step is for AI to transition from supervised to unsupervised learning. The latter means that the AI isn't answering questions that a programmer asks it ("Is this a cat?"). Instead, it's almost like it's looking at the data it has, coming up with its own questions and hypothesis, and answering them or putting them to the test. Combining this ability with the frankly insane processing power of the computer system could result in game-changing discoveries.
In the not-too-distant future, a doctor could run diagnostics on a digital avatar, watching which medical conditions present themselves before the person gets sick in real life.
One company in China plans to develop a way to create a digital avatar of an individual person, then simulate that person's health and medical information into the future. In the not-too-distant future, a doctor could run diagnostics on a digital avatar, watching which medical conditions presented themselves – cancer or a heart condition or anything, really – and help the real-life version prevent those conditions from beginning or treating them before they became a life-threatening issue.
That, obviously, would be an incredibly powerful technology, and it's just one of the many possibilities that unsupervised AI presents. It's also terrifying in the potential for misuse. Even the term "unsupervised AI" brings to mind a dystopian landscape where AI takes over and enslaves humanity. (Pick your favorite movie. There are dozens.) This is a concern, something for developers, programmers, and scientists to consider as they build the systems of the future.
The Ethical Problem That Deserves More Attention
But the more immediate concern about AI is much more mundane. We think of AI as an unbiased system. That's incorrect. Algorithms, after all, are designed by someone or a team, and those people have explicit or implicit biases. Intentionally, or more likely not, they introduce these biases into the very code that forms the basis for the AI. Current systems have a bias against people of color. Facebook tried to rectify the situation and failed. These are two small examples of a larger, potentially systemic problem.
It's vital and necessary for the people developing AI today to be aware of these issues. And, yes, avoid sending us to the brink of a James Cameron movie. But AI is too powerful a tool to ignore. Today, it's identifying cats and on the verge of detecting cancer. In not too many tomorrows, it will be on the forefront of medical innovation. If we are careful, aware, and smart, it will help simulate results, create designer drugs, and revolutionize individualize medicine. "AI is the only way to get there," Pauwels says.
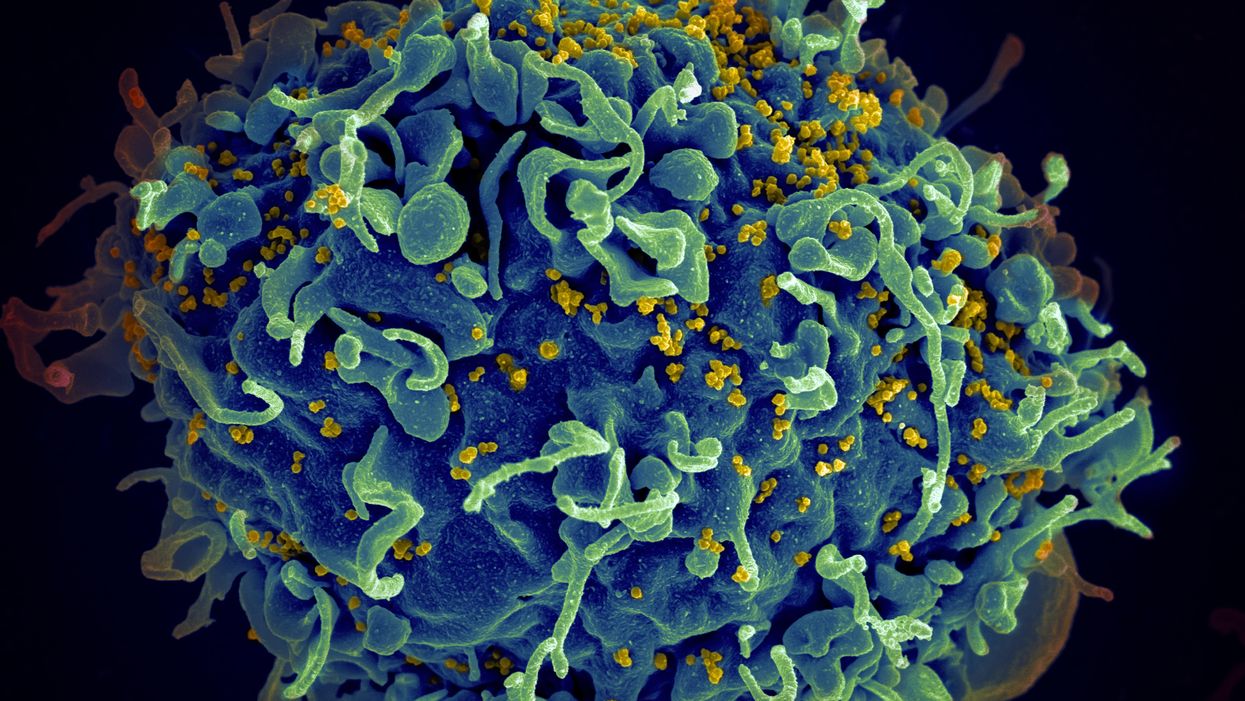
Is a Successful HIV Vaccine Finally on the Horizon?
The HIV virus (yellow) infecting a human cell.
Few vaccines have been as complicated—and filled with false starts and crushed hopes—as the development of an HIV vaccine.
While antivirals help HIV-positive patients live longer and reduce viral transmission to virtually nil, these medications must be taken for life, and preventative medications like pre-exposure prophylaxis, known as PrEP, need to be taken every day to be effective. Vaccines, even if they need boosters, would make prevention much easier.
In August, Moderna began human trials for two HIV vaccine candidates based on messenger RNA.
As they have with the Covid-19 pandemic, mRNA vaccines could change the game. The technology could be applied for gene editing therapy, cancer, other infectious diseases—even a universal influenza vaccine.
In the past, three other mRNA vaccines completed phase-2 trials without success. But the easily customizable platforms mean the vaccines can be tweaked better to target HIV as researchers learn more.
Ever since HIV was discovered as the virus causing AIDS, researchers have been searching for a vaccine. But the decades-long journey has so far been fruitless; while some vaccine candidates showed promise in early trials, none of them have worked well among later-stage clinical trials.
There are two main reasons for this: HIV evolves incredibly quickly, and the structure of the virus makes it very difficult to neutralize with antibodies.
"We in HIV medicine have been desperate to find a vaccine that has effectiveness, but this goal has been elusive so far."
"You know the panic that goes on when a new coronavirus variant surfaces?" asked John Moore, professor of microbiology and immunology at Weill Cornell Medicine who has researched HIV vaccines for 25 years. "With HIV, that kind of variation [happens] pretty much every day in everybody who's infected. It's just orders of magnitude more variable a virus."
Vaccines like these usually work by imitating the outer layer of a virus to teach cells how to recognize and fight off the real thing off before it enters the cell. "If you can prevent landing, you can essentially keep the virus out of the cell," said Larry Corey, the former president and director of the Fred Hutchinson Cancer Research Center who helped run a recent trial of a Johnson & Johnson HIV vaccine candidate, which failed its first efficacy trial.
Like the coronavirus, HIV also has a spike protein with a receptor-binding domain—what Moore calls "the notorious RBD"—that could be neutralized with antibodies. But while that target sticks out like a sore thumb in a virus like SARS-CoV-2, in HIV it's buried under a dense shield. That's not the only target for neutralizing the virus, but all of the targets evolve rapidly and are difficult to reach.
"We understand these targets. We know where they are. But it's still proving incredibly difficult to raise antibodies against them by vaccination," Moore said.
In fact, mRNA vaccines for HIV have been under development for years. The Covid vaccines were built on decades of that research. But it's not as simple as building on this momentum, because of how much more complicated HIV is than SARS-CoV-2, researchers said.
"They haven't succeeded because they were not designed appropriately and haven't been able to induce what is necessary for them to induce," Moore said. "The mRNA technology will enable you to produce a lot of antibodies to the HIV envelope, but if they're the wrong antibodies that doesn't solve the problem."
Part of the problem is that the HIV vaccines have to perform better than our own immune systems. Many vaccines are created by imitating how our bodies overcome an infection, but that doesn't happen with HIV. Once you have the virus, you can't fight it off on your own.
"The human immune system actually does not know how to innately cure HIV," Corey said. "We needed to improve upon the human immune system to make it quicker… with Covid. But we have to actually be better than the human immune system" with HIV.
But in the past few years, there have been impressive leaps in understanding how an HIV vaccine might work. Scientists have known for decades that neutralizing antibodies are key for a vaccine. But in 2010 or so, they were able to mimic the HIV spike and understand how antibodies need to disable the virus. "It helps us understand the nature of the problem, but doesn't instantly solve the problem," Moore said. "Without neutralizing antibodies, you don't have a chance."
Because the vaccines need to induce broadly neutralizing antibodies, and because it's very difficult to neutralize the highly variable HIV, any vaccine will likely be a series of shots that teach the immune system to be on the lookout for a variety of potential attacks.
"Each dose is going to have to have a different purpose," Corey said. "And we hope by the end of the third or fourth dose, we will achieve the level of neutralization that we want."
That's not ideal, because each individual component has to be made and tested—and four shots make the vaccine harder to administer.
"You wouldn't even be going down that route, if there was a better alternative," Moore said. "But there isn't a better alternative."
The mRNA platform is exciting because it is easily customizable, which is especially important in fighting against a shapeshifting, complicated virus. And the mRNA platform has shown itself, in the Covid pandemic, to be safe and quick to make. Effective Covid vaccines were comparatively easy to develop, since the coronavirus is easier to battle than HIV. But companies like Moderna are capitalizing on their success to launch other mRNA therapeutics and vaccines, including the HIV trial.
"You can make the vaccine in two months, three months, in a research lab, and not a year—and the cost of that is really less," Corey said. "It gives us a chance to try many more options, if we've got a good response."
In a trial on macaque monkeys, the Moderna vaccine reduced the chances of infection by 85 percent. "The mRNA platform represents a very promising approach for the development of an HIV vaccine in the future," said Dr. Peng Zhang, who is helping lead the trial at the National Institute of Allergy and Infectious Diseases.
Moderna's trial in humans represents "a very exciting possibility for the prevention of HIV infection," Dr. Monica Gandhi, director of the UCSF-Gladstone Center for AIDS Research, said in an email. "We in HIV medicine have been desperate to find a vaccine that has effectiveness, but this goal has been elusive so far."
If a successful HIV vaccine is developed, the series of shots could include an mRNA shot that primes the immune system, followed by protein subunits that generate the necessary antibodies, Moore said.
"I think it's the only thing that's worth doing," he said. "Without something complicated like that, you have no chance of inducing broadly neutralizing antibodies."
"I can't guarantee you that's going to work," Moore added. "It may completely fail. But at least it's got some science behind it."
New Podcast: The Lead Scientist for the NASA Mission to Venus
The volcano Sapas Mons is displayed in the center of this computer-generated three-dimensional perspective view of the surface of Venus.
The "Making Sense of Science" podcast features interviews with leading medical and scientific experts about the latest developments and the big ethical and societal questions they raise. This monthly podcast is hosted by journalist Kira Peikoff, founding editor of the award-winning science outlet Leaps.org.
This month, our guest is JPL's Dr. Suzanne Smrekar, who will be pushing the boundaries of knowledge about the planet Venus during the upcoming VERITAS mission set to launch in 2028. Why did Earth's twin planet develop so differently than our own? Could Venus ever have hosted life? What is the bigger purpose for humanity in studying the solar system -- is it purely scientific, or is it also a matter of art and philosophy? Hear Dr. Smrekar discuss all this and more on the latest episode.
Watch the 30-Second Trailer:
Listen to the Episode:
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.