Your surgery could harm yourself and the planet. Here's what some doctors are doing about it.
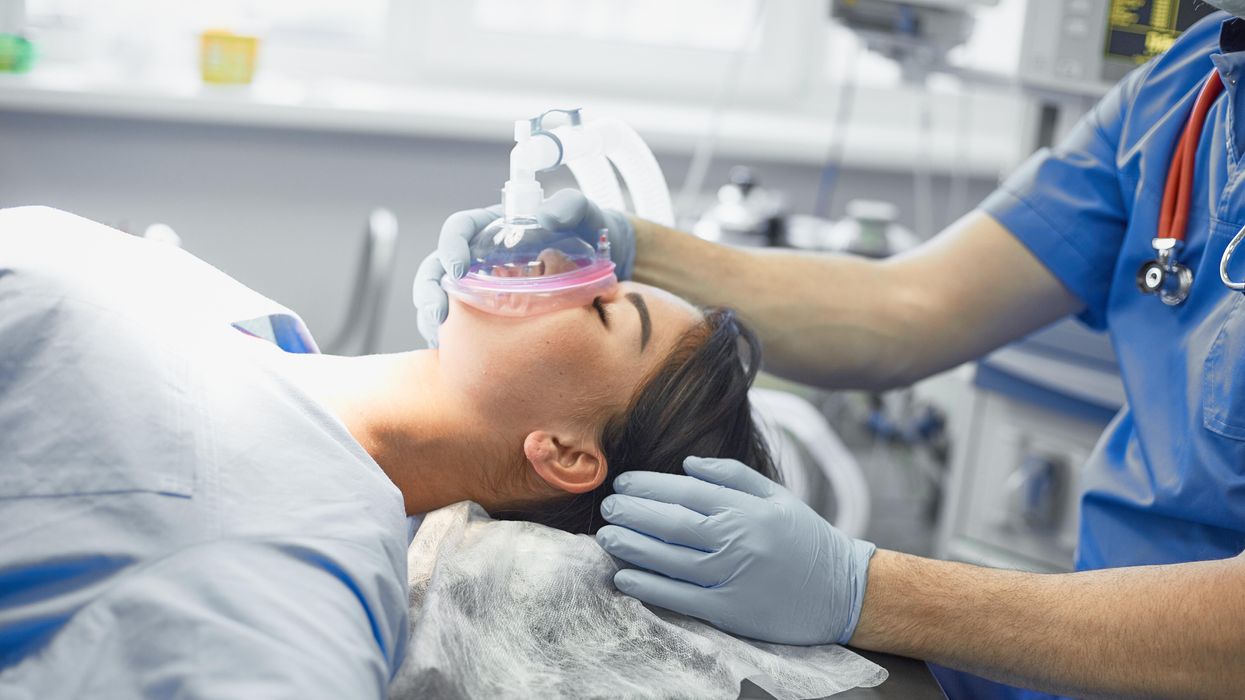
Certain gases used for anesthesia are 3,000 times more damaging for the climate than CO2. Some anesthesiologists are pointing to other solutions.
This is part 1 of a three part series on a new generation of doctors leading the charge to make the health care industry more sustainable - for the benefit of their patients and the planet. Read part 2 here and part 3 here.
Susanne Koch, an anesthesiologist and neurologist, reached a pivot point when she was up to her neck in water, almost literally. The basement of her house in Berlin had flooded in the summer of 2018, when Berlin was pummeled by unusually strong rains. After she drained the house, “I wanted to dig into facts, to understand how exactly these extreme weather events are related to climate change,” she says.
Studying the scientific literature, she realized how urgent the climate crisis is, but the biggest shock was to learn that her profession contributed substantially to the problem: Inhalation gases used during medical procedures are among the most damaging greenhouse gases. Some inhalation gases are 3,000 times more damaging for the climate than CO2, Koch discovered. “Spending seven hours in the surgery room is the equivalent of driving a car for four days nonstop,” she says. Her job of helping people at Europe’s largest university hospital, the Charité in Berlin, was inadvertently damaging both the people and the planet.
“Nobody had ever even mentioned a word about that during my training,” Koch says.
On the whole, the medical sector is responsible for a disproportionally large percentage of greenhouse gas emissions, with the U.S. as the biggest culprit. According to a key paper published in 2020 in Health Affairs, the health industry “is among the most carbon-intensive service sectors in the industrialized world,” accounting for between 4.4 percent and 4.6 percent of greenhouse gas emissions. “It’s not just anesthesia but health care that has a problem,” says Jodi Sherman, anesthesiology professor and Medical Director of the Program on Healthcare Environmental Sustainability at Yale University as well as co-director of the Lancet Planetary Health Commission on Sustainable Healthcare. In the U.S., health care greenhouse gas emissions make up about 8.5 percent of domestic greenhouse gas emissions. They rose 6 percent from 2010 to 2018, to nearly 1,700 kilograms per person, more than in any other nation.
Of course, patients worry primarily about safety, not sustainability. Yet, Koch emphasizes that “as doctors, we have the responsibility to do no harm, and this includes making sure that we use resources as sustainably as possible.” Studies show that 2018 greenhouse gas and toxic air pollutant emissions resulted in the loss of 388,000 disability-adjusted life years in the U.S. alone. “Disease burden from health care pollution is of the same order of magnitude as deaths from preventable medical errors, and should be taken just as seriously,” Sherman cautions.
When Koch, the anesthesiologist, started discussing sustainable options with colleagues, the topic was immediately met with plenty of interest. Her experience is consistent with the latest representative poll of the nonprofit Foundation Health in Germany. Nine out of ten doctors were interested in urgently finding sustainable solutions for medical services but lacked knowhow and resources. For teaching purposes, Sherman and her team have developed the Yale Gassing Greener app that allows anesthesiologists to compare how much pollution they can avoid through choosing different anesthesia methods. Sherman also published professional guidelines intended to help her colleagues better understand how various methods affect carbon emissions.
Significant traces of inhalation gases have been found in Antarctica and the Himalayas, far from the vast majority of surgery rooms.
A solution espoused by both Sherman and Koch is comparatively simple: They stopped using desflurane, which is by far the most damaging of all inhalation gases to the climate. Its greenhouse effect is 2,590 times stronger than carbon dioxide. The Yale New Haven Hospital already stopped using desflurane in 2013, becoming the first known healthcare organization to eliminate a drug based on environmental grounds. Sherman points out that this resulted in saving more than $1.2 million in costs and 1,600 tons of CO2 equivalents, about the same as the exhaust from 360 passenger vehicles per year.
At the Charité, Koch claims that switching to other anesthesiology choices, such as propofol, has eliminated 90 percent of the climate gas emissions in the anesthesiology department since 2016. Young anesthesiologists are still taught to use desflurane as the standard because desflurane is absorbed less into the patients’ bodies, and they wake up faster. However, Koch who has worked as an anesthesiologist since 2006, says that with a little bit of experience, you can learn when to stop giving the propofol so it's timed just as well with a person’s wake-up process. In addition, “patients are less likely to feel nauseous after being given propofol,” Koch says. Intravenous drugs might require more skill, she adds, "but there is nothing unique to the drug desflurane that cannot be accomplished with other medications.”
Desflurane isn’t the only gas to be concerned about. Nitrous oxide is the second most damaging because it’s extremely long-lived in the environment, and it depletes the ozone layer. Climate-conscious anesthesiologists are phasing out this gas, too, or have implemented measures to decrease leaks.
Internationally, 192 governments agreed in the Kyoto protocol of 2005 to reduce halogenated hydrocarbons – resulting from inhalation gases, including desflurane and nitrous oxide – because of their immense climate-warming potential, and in 2016, they pledged to eliminate them by 2035. However, the use of inhalation anesthetics continues to increase worldwide, not least because more people access healthcare in developing countries, and because people in industrialized countries live longer and therefore need more surgeries. Significant traces of inhalation gases have been found in Antarctica and the Himalayas, far from the vast majority of surgery rooms.
Certain companies are now pushing new technology to capture inhalation gases before they are released into the atmosphere, but both Sherman and Koch believe marketing claims of 99 percent efficiency amount to greenwashing. After investigating the technology first-hand and visiting the company that is producing such filters in Germany, Koch concluded that such technology only reduces emissions by 25 percent. And Sherman believes such initiatives are akin to the fallacy of recycling plastic. In addition to questioning their efficiency, Sherman fears such technology “gives the illusion there is a magical solution that means I don’t need to change my behavior, reduce my waste and choose less harmful options.”
Financial interests are at play, too. “Desflurane is the most expensive inhalation gas, and some think, the most expensive must be the best,” Koch says. Both Koch and Sherman lament that efforts to increase sustainability in the medical sector are entirely voluntary in their countries and led by a few dedicated individual professionals while industry-wide standards and transparency are needed, a notion expressed in the American Hospital Association’s Sustainability Roadmap.

Susanne Koch, an anesthesiologist in Berlin, wants her colleagues to stop using a gas called desflurane, which is by far the most damaging of all inhalation gases to the climate.
Adobe Stock
Other countries have done more. The European Union recommends reducing inhalation gases and even contemplated a ban of desflurane, except in medical emergencies. In 2008, the National Health Service (NHS) created a Sustainable Development Unit, which measures CO2 emissions in the U.K. health sector. NHS is the first national health service that pledged to reach net zero carbon by 2040. The carbon footprint of the NHS fell by 26 percent from 1990 to 2019, mostly due to reduced use of certain inhalers and the switch to renewable energy for heat and power. “The evidence that the climate emergency is a health emergency is overwhelming,” said Nick Watts, the NHS Chief Sustainability Officer, in a press release, “with health professionals already needing to manage its symptoms.”
Sherman is a leading voice in demanding action in the U.S. To her, comprehensive solutions start with the mandatory, transparent measurement of emissions in the health sector to tackle the biggest sources of pollution. While the Biden administration highlighted its efforts to reduce these kinds of emissions during the United Nations Climate Conference (COP27) in November 2022 and U.S. delegates announced that more than 100 health care organizations signed the voluntary Health Sector Climate Pledge, with the aim to reduce emissions by 50 percent in the next eight years, Sherman is convinced that voluntary pledges are not enough. “Voluntary measures are insufficient,” she testified in congress. “The vast majority of U.S. health care organizations remain uncommitted to timely action. Those that are committed lack policies and knowledge to support necessary changes; even worse, existing policies drive inappropriate consumption of resources and pollution.”
Both Sherman and Koch look at the larger picture. “Health care organizations have an obligation to their communities to protect public health,” Sherman says. “We must lead by example. That includes setting ambitious, science-based carbon reduction targets to achieve net zero emissions before 2050. We must quantify current emissions and their sources, particularly throughout the health care supply chains.”
Researchers claimed they built a breakthrough superconductor. Social media shot it down almost instantly.
In July, South Korean scientists posted a paper finding they had achieved superconductivity - a claim that was debunked within days.
Harsh Mathur was a graduate physics student at Yale University in late 1989 when faculty announced they had failed to replicate claims made by scientists at the University of Utah and the University of Wolverhampton in England.
Such work is routine. Replicating or attempting to replicate the contraptions, calculations and conclusions crafted by colleagues is foundational to the scientific method. But in this instance, Yale’s findings were reported globally.
“I had a ringside view, and it was crazy,” recalls Mathur, now a professor of physics at Case Western Reserve University in Ohio.
Yale’s findings drew so much attention because initial experiments by Stanley Pons of Utah and Martin Fleischmann of Wolverhampton led to a startling claim: They were able to fuse atoms at room temperature – a scientific El Dorado known as “cold fusion.”
Nuclear fusion powers the stars in the universe. However, star cores must be at least 23.4 million degrees Fahrenheit and under extraordinary pressure to achieve fusion. Pons and Fleischmann claimed they had created an almost limitless source of power achievable at any temperature.
Like fusion, superconductivity can only be achieved in mostly impractical circumstances.
But about six months after they made their startling announcement, the pair’s findings were discredited by researchers at Yale and the California Institute of Technology. It was one of the first instances of a major scientific debunking covered by mass media.
Some scholars say the media attention for cold fusion stemmed partly from a dazzling announcement made three years prior in 1986: Scientists had created the first “superconductor” – material that could transmit electrical current with little or no resistance. It drew global headlines – and whetted the public’s appetite for announcements of scientific breakthroughs that could cause economic transformations.
But like fusion, superconductivity can only be achieved in mostly impractical circumstances: It must operate either at temperatures of at least negative 100 degrees Fahrenheit, or under pressures of around 150,000 pounds per square inch. Superconductivity that functions in closer to a normal environment would cut energy costs dramatically while also opening infinite possibilities for computing, space travel and other applications.
In July, a group of South Korean scientists posted material claiming they had created an iron crystalline substance called LK-99 that could achieve superconductivity at slightly above room temperature and at ambient pressure. The group partners with the Quantum Energy Research Centre, a privately-held enterprise in Seoul, and their claims drew global headlines.
Their work was also debunked. But in the age of internet and social media, the process was compressed from half-a-year into days. And it did not require researchers at world-class universities.
One of the most compelling critiques came from Derrick VanGennep. Although he works in finance, he holds a Ph.D. in physics and held a postdoctoral position at Harvard. The South Korean researchers had posted a video of a nugget of LK-99 in what they claimed was the throes of the Meissner effect – an expulsion of the substance’s magnetic field that would cause it to levitate above a magnet. Unless Hollywood magic is involved, only superconducting material can hover in this manner.
That claim made VanGennep skeptical, particularly since LK-99’s levitation appeared unenthusiastic at best. In fact, a corner of the material still adhered to the magnet near its center. He thought the video demonstrated ferromagnetism – two magnets repulsing one another. He mixed powdered graphite with super glue, stuck iron filings to its surface and mimicked the behavior of LK-99 in his own video, which was posted alongside the researchers’ video.
VanGennep believes the boldness of the South Korean claim was what led to him and others in the scientific community questioning it so quickly.
“The swift replication attempts stemmed from the combination of the extreme claim, the fact that the synthesis for this material is very straightforward and fast, and the amount of attention that this story was getting on social media,” he says.
But practicing scientists were suspicious of the data as well. Michael Norman, director of the Argonne Quantum Institute at the Argonne National Laboratory just outside of Chicago, had doubts immediately.
Will this saga hurt or even affect the careers of the South Korean researchers? Possibly not, if the previous fusion example is any indication.
“It wasn’t a very polished paper,” Norman says of the Korean scientists’ work. That opinion was reinforced, he adds, when it turned out the paper had been posted online by one of the researchers prior to seeking publication in a peer-reviewed journal. Although Norman and Mathur say that is routine with scientific research these days, Norman notes it was posted by one of the junior researchers over the doubts of two more senior scientists on the project.
Norman also raises doubts about the data reported. Among other issues, he observes that the samples created by the South Korean researchers contained traces of copper sulfide that could inadvertently amplify findings of conductivity.
The lack of the Meissner effect also caught Mathur’s attention. “Ferromagnets tend to be unstable when they levitate,” he says, adding that the video “just made me feel unconvinced. And it made me feel like they hadn't made a very good case for themselves.”
Will this saga hurt or even affect the careers of the South Korean researchers? Possibly not, if the previous fusion example is any indication. Despite being debunked, cold fusion claimants Pons and Fleischmann didn’t disappear. They moved their research to automaker Toyota’s IMRA laboratory in France, which along with the Japanese government spent tens of millions of dollars on their work before finally pulling the plug in 1998.
Fusion has since been created in laboratories, but being unable to reproduce the density of a star’s core would require excruciatingly high temperatures to achieve – about 160 million degrees Fahrenheit. A recently released Government Accountability Office report concludes practical fusion likely remains at least decades away.
However, like Pons and Fleischman, the South Korean researchers are not going anywhere. They claim that LK-99’s Meissner effect is being obscured by the fact the substance is both ferromagnetic and diamagnetic. They have filed for a patent in their country. But for now, those claims remain chimerical.
In the meantime, the consensus as to when a room temperature superconductor will be achieved is mixed. VenGennep – who studied the issue during his graduate and postgraduate work – puts the chance of creating such a superconductor by 2050 at perhaps 50-50. Mathur believes it could happen sooner, but adds that research on the topic has been going on for nearly a century, and that it has seen many plateaus.
“There's always this possibility that there's going to be something out there that we're going to discover unexpectedly,” Norman notes. The only certainty in this age of social media is that it will be put through the rigors of replication instantly.
Scientists implant brain cells to counter Parkinson's disease
In a recent research trial, patients with Parkinson's disease reported that their symptoms had improved after stem cells were implanted into their brains. Martin Taylor, far right, was diagnosed at age 32.
Martin Taylor was only 32 when he was diagnosed with Parkinson's, a disease that causes tremors, stiff muscles and slow physical movement - symptoms that steadily get worse as time goes on.
“It's horrible having Parkinson's,” says Taylor, a data analyst, now 41. “It limits my ability to be the dad and husband that I want to be in many cruel and debilitating ways.”
Today, more than 10 million people worldwide live with Parkinson's. Most are diagnosed when they're considerably older than Taylor, after age 60. Although recent research has called into question certain aspects of the disease’s origins, Parkinson’s eventually kills the nerve cells in the brain that produce dopamine, a signaling chemical that carries messages around the body to control movement. Many patients have lost 60 to 80 percent of these cells by the time they are diagnosed.
For years, there's been little improvement in the standard treatment. Patients are typically given the drug levodopa, a chemical that's absorbed by the brain’s nerve cells, or neurons, and converted into dopamine. This drug addresses the symptoms but has no impact on the course of the disease as patients continue to lose dopamine producing neurons. Eventually, the treatment stops working effectively.
BlueRock Therapeutics, a cell therapy company based in Massachusetts, is taking a different approach by focusing on the use of stem cells, which can divide into and generate new specialized cells. The company makes the dopamine-producing cells that patients have lost and inserts these cells into patients' brains. “We have a disease with a high unmet need,” says Ahmed Enayetallah, the senior vice president and head of development at BlueRock. “We know [which] cells…are lost to the disease, and we can make them. So it really came together to use stem cells in Parkinson's.”
In a phase 1 research trial announced late last month, patients reported that their symptoms had improved after a year of treatment. Brain scans also showed an increased number of neurons generating dopamine in patients’ brains.
Increases in dopamine signals
The recent phase 1 trial focused on deploying BlueRock’s cell therapy, called bemdaneprocel, to treat 12 patients suffering from Parkinson’s. The team developed the new nerve cells and implanted them into specific locations on each side of the patient's brain through two small holes in the skull made by a neurosurgeon. “We implant cells into the places in the brain where we think they have the potential to reform the neural networks that are lost to Parkinson's disease,” Enayetallah says. The goal is to restore motor function to patients over the long-term.
Five patients were given a relatively low dose of cells while seven got higher doses. Specialized brain scans showed evidence that the transplanted cells had survived, increasing the overall number of dopamine producing cells. The team compared the baseline number of these cells before surgery to the levels one year later. “The scans tell us there is evidence of increased dopamine signals in the part of the brain affected by Parkinson's,” Enayetallah says. “Normally you’d expect the signal to go down in untreated Parkinson’s patients.”
"I think it has a real chance to reverse motor symptoms, essentially replacing a missing part," says Tilo Kunath, a professor of regenerative neurobiology at the University of Edinburgh.
The team also asked patients to use a specific type of home diary to log the times when symptoms were well controlled and when they prevented normal activity. After a year of treatment, patients taking the higher dose reported symptoms were under control for an average of 2.16 hours per day above their baselines. At the smaller dose, these improvements were significantly lower, 0.72 hours per day. The higher-dose patients reported a corresponding decrease in the amount of time when symptoms were uncontrolled, by an average of 1.91 hours, compared to 0.75 hours for the lower dose. The trial was safe, and patients tolerated the year of immunosuppression needed to make sure their bodies could handle the foreign cells.
Claire Bale, the associate director of research at Parkinson's U.K., sees the promise of BlueRock's approach, while noting the need for more research on a possible placebo effect. The trial participants knew they were getting the active treatment, and placebo effects are known to be a potential factor in Parkinson’s research. Even so, “The results indicate that this therapy produces improvements in symptoms for Parkinson's, which is very encouraging,” Bale says.
Tilo Kunath, a professor of regenerative neurobiology at the University of Edinburgh, also finds the results intriguing. “I think it's excellent,” he says. “I think it has a real chance to reverse motor symptoms, essentially replacing a missing part.” However, it could take time for this therapy to become widely available, Kunath says, and patients in the late stages of the disease may not benefit as much. “Data from cell transplantation with fetal tissue in the 1980s and 90s show that cells did not survive well and release dopamine in these [late-stage] patients.”
Searching for the right approach
There's a long history of using cell therapy as a treatment for Parkinson's. About four decades ago, scientists at the University of Lund in Sweden developed a method in which they transferred parts of fetal brain tissue to patients with Parkinson's so that their nerve cells would produce dopamine. Many benefited, and some were able to stop their medication. However, the use of fetal tissue was highly controversial at that time, and the tissues were difficult to obtain. Later trials in the U.S. showed that people benefited only if a significant amount of the tissue was used, and several patients experienced side effects. Eventually, the work lost momentum.
“Like many in the community, I'm aware of the long history of cell therapy,” says Taylor, the patient living with Parkinson's. “They've long had that cure over the horizon.”
In 2000, Lorenz Studer led a team at the Memorial Sloan Kettering Centre, in New York, to find the chemical signals needed to get stem cells to differentiate into cells that release dopamine. Back then, the team managed to make cells that produced some dopamine, but they led to only limited improvements in animals. About a decade later, in 2011, Studer and his team found the specific signals needed to guide embryonic cells to become the right kind of dopamine producing cells. Their experiments in mice, rats and monkeys showed that their implanted cells had a significant impact, restoring lost movement.
Studer then co-founded BlueRock Therapeutics in 2016. Forming the most effective stem cells has been one of the biggest challenges, says Enayetallah, the BlueRock VP. “It's taken a lot of effort and investment to manufacture and make the cells at the right scale under the right conditions.” The team is now using cells that were first isolated in 1998 at the University of Wisconsin, a major advantage because they’re available in a virtually unlimited supply.
Other efforts underway
In the past several years, University of Lund researchers have begun to collaborate with the University of Cambridge on a project to use embryonic stem cells, similar to BlueRock’s approach. They began clinical trials this year.
A company in Japan called Sumitomo is using a different strategy; instead of stem cells from embryos, they’re reprogramming adults' blood or skin cells into induced pluripotent stem cells - meaning they can turn into any cell type - and then directing them into dopamine producing neurons. Although Sumitomo started clinical trials earlier than BlueRock, they haven’t yet revealed any results.
“It's a rapidly evolving field,” says Emma Lane, a pharmacologist at the University of Cardiff who researches clinical interventions for Parkinson’s. “But BlueRock’s trial is the first full phase 1 trial to report such positive findings with stem cell based therapies.” The company’s upcoming phase 2 research will be critical to show how effectively the therapy can improve disease symptoms, she added.
The cure over the horizon
BlueRock will continue to look at data from patients in the phase 1 trial to monitor the treatment’s effects over a two-year period. Meanwhile, the team is planning the phase 2 trial with more participants, including a placebo group.
For patients with Parkinson’s like Martin Taylor, the therapy offers some hope, though Taylor recognizes that more research is needed.

BlueRock Therapeutics
“Like many in the community, I'm aware of the long history of cell therapy,” he says. “They've long had that cure over the horizon.” His expectations are somewhat guarded, he says, but, “it's certainly positive to see…movement in the field again.”
"If we can demonstrate what we’re seeing today in a more robust study, that would be great,” Enayetallah says. “At the end of the day, we want to address that unmet need in a field that's been waiting for a long time.”
Editor's note: The company featured in this piece, BlueRock Therapeutics, is a portfolio company of Leaps by Bayer, which is a sponsor of Leaps.org. BlueRock was acquired by Bayer Pharmaceuticals in 2019. Leaps by Bayer and other sponsors have never exerted influence over Leaps.org content or contributors.

