Scientists redesign bacteria to tackle the antibiotic resistance crisis
Probiotic bacteria can be engineered to fight antibiotic-resistant superbugs by releasing chemicals that kill them.
In 1945, almost two decades after Alexander Fleming discovered penicillin, he warned that as antibiotics use grows, they may lose their efficiency. He was prescient—the first case of penicillin resistance was reported two years later. Back then, not many people paid attention to Fleming’s warning. After all, the “golden era” of the antibiotics age had just began. By the 1950s, three new antibiotics derived from soil bacteria — streptomycin, chloramphenicol, and tetracycline — could cure infectious diseases like tuberculosis, cholera, meningitis and typhoid fever, among others.
Today, these antibiotics and many of their successors developed through the 1980s are gradually losing their effectiveness. The extensive overuse and misuse of antibiotics led to the rise of drug resistance. The livestock sector buys around 80 percent of all antibiotics sold in the U.S. every year. Farmers feed cows and chickens low doses of antibiotics to prevent infections and fatten up the animals, which eventually causes resistant bacterial strains to evolve. If manure from cattle is used on fields, the soil and vegetables can get contaminated with antibiotic-resistant bacteria. Another major factor is doctors overprescribing antibiotics to humans, particularly in low-income countries. Between 2000 to 2018, the global rates of human antibiotic consumption shot up by 46 percent.
In recent years, researchers have been exploring a promising avenue: the use of synthetic biology to engineer new bacteria that may work better than antibiotics. The need continues to grow, as a Lancet study linked antibiotic resistance to over 1.27 million deaths worldwide in 2019, surpassing HIV/AIDS and malaria. The western sub-Saharan Africa region had the highest death rate (27.3 people per 100,000).
Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
To make it worse, our remedy pipelines are drying up. Out of the 18 biggest pharmaceutical companies, 15 abandoned antibiotic development by 2013. According to the AMR Action Fund, venture capital has remained indifferent towards biotech start-ups developing new antibiotics. In 2019, at least two antibiotic start-ups filed for bankruptcy. As of December 2020, there were 43 new antibiotics in clinical development. But because they are based on previously known molecules, scientists say they are inadequate for treating multidrug-resistant bacteria. Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
The rise of synthetic biology
To circumvent this dire future, scientists have been working on alternative solutions using synthetic biology tools, meaning genetically modifying good bacteria to fight the bad ones.
From the time life evolved on earth around 3.8 billion years ago, bacteria have engaged in biological warfare. They constantly strategize new methods to combat each other by synthesizing toxic proteins that kill competition.
For example, Escherichia coli produces bacteriocins or toxins to kill other strains of E.coli that attempt to colonize the same habitat. Microbes like E.coli (which are not all pathogenic) are also naturally present in the human microbiome. The human microbiome harbors up to 100 trillion symbiotic microbial cells. The majority of them are beneficial organisms residing in the gut at different compositions.
The chemicals that these “good bacteria” produce do not pose any health risks to us, but can be toxic to other bacteria, particularly to human pathogens. For the last three decades, scientists have been manipulating bacteria’s biological warfare tactics to our collective advantage.
In the late 1990s, researchers drew inspiration from electrical and computing engineering principles that involve constructing digital circuits to control devices. In certain ways, every cell in living organisms works like a tiny computer. The cell receives messages in the form of biochemical molecules that cling on to its surface. Those messages get processed within the cells through a series of complex molecular interactions.
Synthetic biologists can harness these living cells’ information processing skills and use them to construct genetic circuits that perform specific instructions—for example, secrete a toxin that kills pathogenic bacteria. “Any synthetic genetic circuit is merely a piece of information that hangs around in the bacteria’s cytoplasm,” explains José Rubén Morones-Ramírez, a professor at the Autonomous University of Nuevo León, Mexico. Then the ribosome, which synthesizes proteins in the cell, processes that new information, making the compounds scientists want bacteria to make. “The genetic circuit remains separated from the living cell’s DNA,” Morones-Ramírez explains. When the engineered bacteria replicates, the genetic circuit doesn’t become part of its genome.
Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut.
In 2000, Boston-based researchers constructed an E.coli with a genetic switch that toggled between turning genes on and off two. Later, they built some safety checks into their bacteria. “To prevent unintentional or deleterious consequences, in 2009, we built a safety switch in the engineered bacteria’s genetic circuit that gets triggered after it gets exposed to a pathogen," says James Collins, a professor of biological engineering at MIT and faculty member at Harvard University’s Wyss Institute. “After getting rid of the pathogen, the engineered bacteria is designed to switch off and leave the patient's body.”
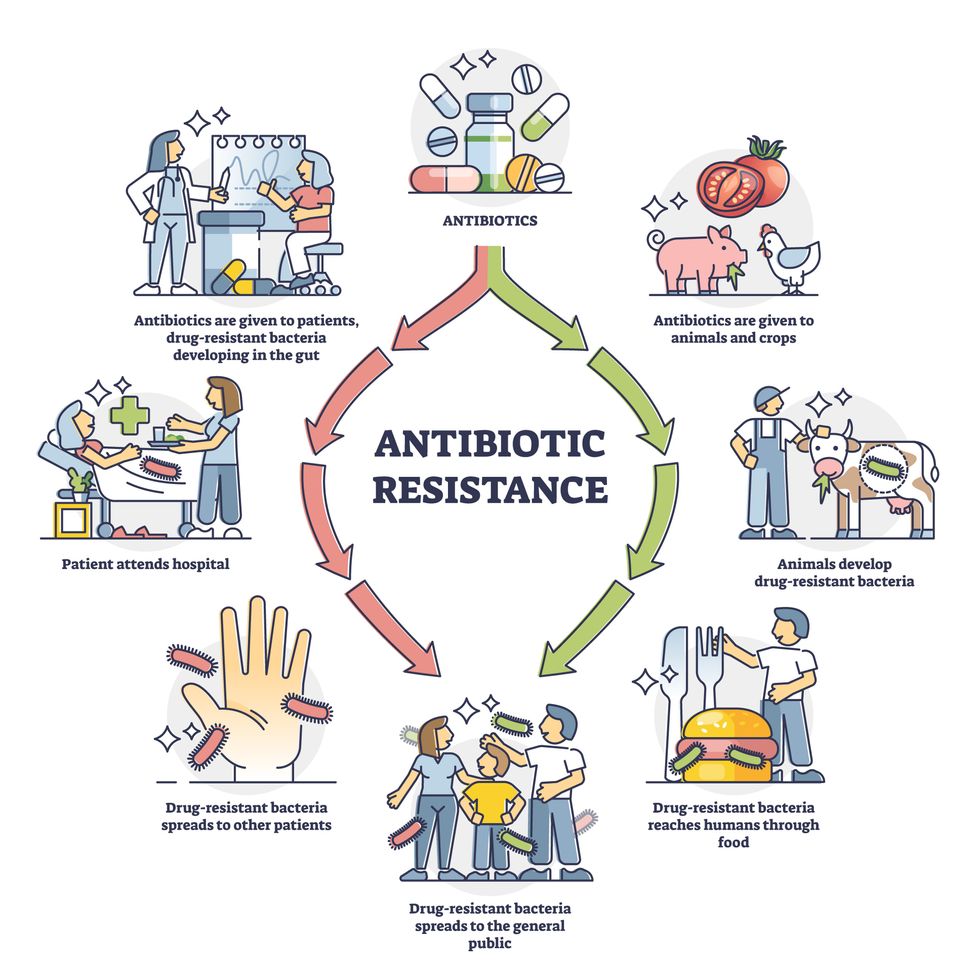
Overuse and misuse of antibiotics causes resistant strains to evolve
Adobe Stock
Seek and destroy
As the field of synthetic biology developed, scientists began using engineered bacteria to tackle superbugs. They first focused on Vibrio cholerae, which in the 19th and 20th century caused cholera pandemics in India, China, the Middle East, Europe, and Americas. Like many other bacteria, V. cholerae communicate with each other via quorum sensing, a process in which the microorganisms release different signaling molecules, to convey messages to its brethren. Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut. When untreated, cholera has a mortality rate of 25 to 50 percent and outbreaks frequently occur in developing countries, especially during floods and droughts.
Sometimes, however, V. cholerae makes mistakes. In 2008, researchers at Cornell University observed that when quorum sensing V. cholerae accidentally released high concentrations of a signaling molecule called CAI-1, it had a counterproductive effect—the pathogen couldn’t colonize the gut.
So the group, led by John March, professor of biological and environmental engineering, developed a novel strategy to combat V. cholerae. They genetically engineered E.coli to eavesdrop on V. cholerae communication networks and equipped it with the ability to release the CAI-1 molecules. That interfered with V. cholerae progress. Two years later, the Cornell team showed that V. cholerae-infected mice treated with engineered E.coli had a 92 percent survival rate.
These findings inspired researchers to sic the good bacteria present in foods like yogurt and kimchi onto the drug-resistant ones.
Three years later in 2011, Singapore-based scientists engineered E.coli to detect and destroy Pseudomonas aeruginosa, an often drug-resistant pathogen that causes pneumonia, urinary tract infections, and sepsis. Once the genetically engineered E.coli found its target through its quorum sensing molecules, it then released a peptide, that could eradicate 99 percent of P. aeruginosa cells in a test-tube experiment. The team outlined their work in a Molecular Systems Biology study.
“At the time, we knew that we were entering new, uncharted territory,” says lead author Matthew Chang, an associate professor and synthetic biologist at the National University of Singapore and lead author of the study. “To date, we are still in the process of trying to understand how long these microbes stay in our bodies and how they might continue to evolve.”
More teams followed the same path. In a 2013 study, MIT researchers also genetically engineered E.coli to detect P. aeruginosa via the pathogen’s quorum-sensing molecules. It then destroyed the pathogen by secreting a lab-made toxin.
Probiotics that fight
A year later in 2014, a Nature study found that the abundance of Ruminococcus obeum, a probiotic bacteria naturally occurring in the human microbiome, interrupts and reduces V.cholerae’s colonization— by detecting the pathogen’s quorum sensing molecules. The natural accumulation of R. obeum in Bangladeshi adults helped them recover from cholera despite living in an area with frequent outbreaks.
The findings from 2008 to 2014 inspired Collins and his team to delve into how good bacteria present in foods like yogurt and kimchi can attack drug-resistant bacteria. In 2018, Collins and his team developed the engineered probiotic strategy. They tweaked a bacteria commonly found in yogurt called Lactococcus lactis to treat cholera.
Engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut. --José Rubén Morones-Ramírez.
More scientists followed with more experiments. So far, researchers have engineered various probiotic organisms to fight pathogenic bacteria like Staphylococcus aureus (leading cause of skin, tissue, bone, joint and blood infections) and Clostridium perfringens (which causes watery diarrhea) in test-tube and animal experiments. In 2020, Russian scientists engineered a probiotic called Pichia pastoris to produce an enzyme called lysostaphin that eradicated S. aureus in vitro. Another 2020 study from China used an engineered probiotic bacteria Lactobacilli casei as a vaccine to prevent C. perfringens infection in rabbits.
In a study last year, Ramírez’s group at the Autonomous University of Nuevo León, engineered E. coli to detect quorum-sensing molecules from Methicillin-resistant Staphylococcus aureus or MRSA, a notorious superbug. The E. coli then releases a bacteriocin that kills MRSA. “An antibiotic is just a molecule that is not intelligent,” says Ramírez. “On the other hand, engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut.”
Collins and Timothy Lu, an associate professor of biological engineering at MIT, found that engineered E. coli can help treat other conditions—such as phenylketonuria, a rare metabolic disorder, that causes the build-up of an amino acid phenylalanine. Their start-up Synlogic aims to commercialize the technology, and has completed a phase 2 clinical trial.
Circumventing the challenges
The bacteria-engineering technique is not without pitfalls. One major challenge is that beneficial gut bacteria produce their own quorum-sensing molecules that can be similar to those that pathogens secrete. If an engineered bacteria’s biosensor is not specific enough, it will be ineffective.
Another concern is whether engineered bacteria might mutate after entering the gut. “As with any technology, there are risks where bad actors could have the capability to engineer a microbe to act quite nastily,” says Collins of MIT. But Collins and Ramírez both insist that the chances of the engineered bacteria mutating on its own are virtually non-existent. “It is extremely unlikely for the engineered bacteria to mutate,” Ramírez says. “Coaxing a living cell to do anything on command is immensely challenging. Usually, the greater risk is that the engineered bacteria entirely lose its functionality.”
However, the biggest challenge is bringing the curative bacteria to consumers. Pharmaceutical companies aren’t interested in antibiotics or their alternatives because it’s less profitable than developing new medicines for non-infectious diseases. Unlike the more chronic conditions like diabetes or cancer that require long-term medications, infectious diseases are usually treated much quicker. Running clinical trials are expensive and antibiotic-alternatives aren’t lucrative enough.
“Unfortunately, new medications for antibiotic resistant infections have been pushed to the bottom of the field,” says Lu of MIT. “It's not because the technology does not work. This is more of a market issue. Because clinical trials cost hundreds of millions of dollars, the only solution is that governments will need to fund them.” Lu stresses that societies must lobby to change how the modern healthcare industry works. “The whole world needs better treatments for antibiotic resistance.”
Podcast: The Friday Five weekly roundup in health research
Researchers are making progress on a vaccine for Lyme disease, sex differences in cancer, new research on reducing your risk of dementia with leisure activities, and more in this week's Friday Five
The Friday Five covers five stories in health research that you may have missed this week. There are plenty of controversies and troubling ethical issues in science – and we get into many of them in our online magazine – but this news roundup focuses on scientific creativity and progress to give you a therapeutic dose of inspiration headed into the weekend.
Covered in this week's Friday Five:
- Sex differences in cancer
- Promising research on a vaccine for Lyme disease
- Using a super material for brain-like devices
- Measuring your immunity to Covid
- Reducing dementia risk with leisure activities
Giving robots self-awareness as they move through space - and maybe even providing them with gene-like methods for storing rules of behavior - could be important steps toward creating more intelligent machines.
One day in recent past, scientists at Columbia University’s Creative Machines Lab set up a robotic arm inside a circle of five streaming video cameras and let the robot watch itself move, turn and twist. For about three hours the robot did exactly that—it looked at itself this way and that, like toddlers exploring themselves in a room full of mirrors. By the time the robot stopped, its internal neural network finished learning the relationship between the robot’s motor actions and the volume it occupied in its environment. In other words, the robot built a spatial self-awareness, just like humans do. “We trained its deep neural network to understand how it moved in space,” says Boyuan Chen, one of the scientists who worked on it.
For decades robots have been doing helpful tasks that are too hard, too dangerous, or physically impossible for humans to carry out themselves. Robots are ultimately superior to humans in complex calculations, following rules to a tee and repeating the same steps perfectly. But even the biggest successes for human-robot collaborations—those in manufacturing and automotive industries—still require separating the two for safety reasons. Hardwired for a limited set of tasks, industrial robots don't have the intelligence to know where their robo-parts are in space, how fast they’re moving and when they can endanger a human.
Over the past decade or so, humans have begun to expect more from robots. Engineers have been building smarter versions that can avoid obstacles, follow voice commands, respond to human speech and make simple decisions. Some of them proved invaluable in many natural and man-made disasters like earthquakes, forest fires, nuclear accidents and chemical spills. These disaster recovery robots helped clean up dangerous chemicals, looked for survivors in crumbled buildings, and ventured into radioactive areas to assess damage.
Now roboticists are going a step further, training their creations to do even better: understand their own image in space and interact with humans like humans do. Today, there are already robot-teachers like KeeKo, robot-pets like Moffin, robot-babysitters like iPal, and robotic companions for the elderly like Pepper.
But even these reasonably intelligent creations still have huge limitations, some scientists think. “There are niche applications for the current generations of robots,” says professor Anthony Zador at Cold Spring Harbor Laboratory—but they are not “generalists” who can do varied tasks all on their own, as they mostly lack the abilities to improvise, make decisions based on a multitude of facts or emotions, and adjust to rapidly changing circumstances. “We don’t have general purpose robots that can interact with the world. We’re ages away from that.”
Robotic spatial self-awareness – the achievement by the team at Columbia – is an important step toward creating more intelligent machines. Hod Lipson, professor of mechanical engineering who runs the Columbia lab, says that future robots will need this ability to assist humans better. Knowing how you look and where in space your parts are, decreases the need for human oversight. It also helps the robot to detect and compensate for damage and keep up with its own wear-and-tear. And it allows robots to realize when something is wrong with them or their parts. “We want our robots to learn and continue to grow their minds and bodies on their own,” Chen says. That’s what Zador wants too—and on a much grander level. “I want a robot who can drive my car, take my dog for a walk and have a conversation with me.”

Columbia scientists have trained a robot to become aware of its own "body," so it can map the right path to touch a ball without running into an obstacle, in this case a square.
Jane Nisselson and Yinuo Qin/ Columbia Engineering
Today’s technological advances are making some of these leaps of progress possible. One of them is the so-called Deep Learning—a method that trains artificial intelligence systems to learn and use information similar to how humans do it. Described as a machine learning method based on neural network architectures with multiple layers of processing units, Deep Learning has been used to successfully teach machines to recognize images, understand speech and even write text.
Trained by Google, one of these language machine learning geniuses, BERT, can finish sentences. Another one called GPT3, designed by San Francisco-based company OpenAI, can write little stories. Yet, both of them still make funny mistakes in their linguistic exercises that even a child wouldn’t. According to a paper published by Stanford’s Center for Research on Foundational Models, BERT seems to not understand the word “not.” When asked to fill in the word after “A robin is a __” it correctly answers “bird.” But try inserting the word “not” into that sentence (“A robin is not a __”) and BERT still completes it the same way. Similarly, in one of its stories, GPT3 wrote that if you mix a spoonful of grape juice into your cranberry juice and drink the concoction, you die. It seems that robots, and artificial intelligence systems in general, are still missing some rudimentary facts of life that humans and animals grasp naturally and effortlessly.
How does one give robots a genome? Zador has an idea. We can’t really equip machines with real biological nucleotide-based genes, but we can mimic the neuronal blueprint those genes create.
It's not exactly the robots’ fault. Compared to humans, and all other organisms that have been around for thousands or millions of years, robots are very new. They are missing out on eons of evolutionary data-building. Animals and humans are born with the ability to do certain things because they are pre-wired in them. Flies know how to fly, fish knows how to swim, cats know how to meow, and babies know how to cry. Yet, flies don’t really learn to fly, fish doesn’t learn to swim, cats don’t learn to meow, and babies don’t learn to cry—they are born able to execute such behaviors because they’re preprogrammed to do so. All that happens thanks to the millions of years of evolutions wired into their respective genomes, which give rise to the brain’s neural networks responsible for these behaviors. Robots are the newbies, missing out on that trove of information, Zador argues.
A neuroscience professor who studies how brain circuitry generates various behaviors, Zador has a different approach to developing the robotic mind. Until their creators figure out a way to imbue the bots with that information, robots will remain quite limited in their abilities. Each model will only be able to do certain things it was programmed to do, but it will never go above and beyond its original code. So Zador argues that we have to start giving robots a genome.
How does one do that? Zador has an idea. We can’t really equip machines with real biological nucleotide-based genes, but we can mimic the neuronal blueprint those genes create. Genomes lay out rules for brain development. Specifically, the genome encodes blueprints for wiring up our nervous system—the details of which neurons are connected, the strength of those connections and other specs that will later hold the information learned throughout life. “Our genomes serve as blueprints for building our nervous system and these blueprints give rise to a human brain, which contains about 100 billion neurons,” Zador says.
If you think what a genome is, he explains, it is essentially a very compact and compressed form of information storage. Conceptually, genomes are similar to CliffsNotes and other study guides. When students read these short summaries, they know about what happened in a book, without actually reading that book. And that’s how we should be designing the next generation of robots if we ever want them to act like humans, Zador says. “We should give them a set of behavioral CliffsNotes, which they can then unwrap into brain-like structures.” Robots that have such brain-like structures will acquire a set of basic rules to generate basic behaviors and use them to learn more complex ones.
Currently Zador is in the process of developing algorithms that function like simple rules that generate such behaviors. “My algorithms would write these CliffsNotes, outlining how to solve a particular problem,” he explains. “And then, the neural networks will use these CliffsNotes to figure out which ones are useful and use them in their behaviors.” That’s how all living beings operate. They use the pre-programmed info from their genetics to adapt to their changing environments and learn what’s necessary to survive and thrive in these settings.
For example, a robot’s neural network could draw from CliffsNotes with “genetic” instructions for how to be aware of its own body or learn to adjust its movements. And other, different sets of CliffsNotes may imbue it with the basics of physical safety or the fundamentals of speech.
At the moment, Zador is working on algorithms that are trying to mimic neuronal blueprints for very simple organisms—such as earthworms, which have only 302 neurons and about 7000 synapses compared to the millions we have. That’s how evolution worked, too—expanding the brains from simple creatures to more complex to the Homo Sapiens. But if it took millions of years to arrive at modern humans, how long would it take scientists to forge a robot with human intelligence? That’s a billion-dollar question. Yet, Zador is optimistic. “My hypotheses is that if you can build simple organisms that can interact with the world, then the higher level functions will not be nearly as challenging as they currently are.”
Lina Zeldovich has written about science, medicine and technology for Popular Science, Smithsonian, National Geographic, Scientific American, Reader’s Digest, the New York Times and other major national and international publications. A Columbia J-School alumna, she has won several awards for her stories, including the ASJA Crisis Coverage Award for Covid reporting, and has been a contributing editor at Nautilus Magazine. In 2021, Zeldovich released her first book, The Other Dark Matter, published by the University of Chicago Press, about the science and business of turning waste into wealth and health. You can find her on http://linazeldovich.com/ and @linazeldovich.

