Scientists redesign bacteria to tackle the antibiotic resistance crisis
Probiotic bacteria can be engineered to fight antibiotic-resistant superbugs by releasing chemicals that kill them.
In 1945, almost two decades after Alexander Fleming discovered penicillin, he warned that as antibiotics use grows, they may lose their efficiency. He was prescient—the first case of penicillin resistance was reported two years later. Back then, not many people paid attention to Fleming’s warning. After all, the “golden era” of the antibiotics age had just began. By the 1950s, three new antibiotics derived from soil bacteria — streptomycin, chloramphenicol, and tetracycline — could cure infectious diseases like tuberculosis, cholera, meningitis and typhoid fever, among others.
Today, these antibiotics and many of their successors developed through the 1980s are gradually losing their effectiveness. The extensive overuse and misuse of antibiotics led to the rise of drug resistance. The livestock sector buys around 80 percent of all antibiotics sold in the U.S. every year. Farmers feed cows and chickens low doses of antibiotics to prevent infections and fatten up the animals, which eventually causes resistant bacterial strains to evolve. If manure from cattle is used on fields, the soil and vegetables can get contaminated with antibiotic-resistant bacteria. Another major factor is doctors overprescribing antibiotics to humans, particularly in low-income countries. Between 2000 to 2018, the global rates of human antibiotic consumption shot up by 46 percent.
In recent years, researchers have been exploring a promising avenue: the use of synthetic biology to engineer new bacteria that may work better than antibiotics. The need continues to grow, as a Lancet study linked antibiotic resistance to over 1.27 million deaths worldwide in 2019, surpassing HIV/AIDS and malaria. The western sub-Saharan Africa region had the highest death rate (27.3 people per 100,000).
Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
To make it worse, our remedy pipelines are drying up. Out of the 18 biggest pharmaceutical companies, 15 abandoned antibiotic development by 2013. According to the AMR Action Fund, venture capital has remained indifferent towards biotech start-ups developing new antibiotics. In 2019, at least two antibiotic start-ups filed for bankruptcy. As of December 2020, there were 43 new antibiotics in clinical development. But because they are based on previously known molecules, scientists say they are inadequate for treating multidrug-resistant bacteria. Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
The rise of synthetic biology
To circumvent this dire future, scientists have been working on alternative solutions using synthetic biology tools, meaning genetically modifying good bacteria to fight the bad ones.
From the time life evolved on earth around 3.8 billion years ago, bacteria have engaged in biological warfare. They constantly strategize new methods to combat each other by synthesizing toxic proteins that kill competition.
For example, Escherichia coli produces bacteriocins or toxins to kill other strains of E.coli that attempt to colonize the same habitat. Microbes like E.coli (which are not all pathogenic) are also naturally present in the human microbiome. The human microbiome harbors up to 100 trillion symbiotic microbial cells. The majority of them are beneficial organisms residing in the gut at different compositions.
The chemicals that these “good bacteria” produce do not pose any health risks to us, but can be toxic to other bacteria, particularly to human pathogens. For the last three decades, scientists have been manipulating bacteria’s biological warfare tactics to our collective advantage.
In the late 1990s, researchers drew inspiration from electrical and computing engineering principles that involve constructing digital circuits to control devices. In certain ways, every cell in living organisms works like a tiny computer. The cell receives messages in the form of biochemical molecules that cling on to its surface. Those messages get processed within the cells through a series of complex molecular interactions.
Synthetic biologists can harness these living cells’ information processing skills and use them to construct genetic circuits that perform specific instructions—for example, secrete a toxin that kills pathogenic bacteria. “Any synthetic genetic circuit is merely a piece of information that hangs around in the bacteria’s cytoplasm,” explains José Rubén Morones-Ramírez, a professor at the Autonomous University of Nuevo León, Mexico. Then the ribosome, which synthesizes proteins in the cell, processes that new information, making the compounds scientists want bacteria to make. “The genetic circuit remains separated from the living cell’s DNA,” Morones-Ramírez explains. When the engineered bacteria replicates, the genetic circuit doesn’t become part of its genome.
Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut.
In 2000, Boston-based researchers constructed an E.coli with a genetic switch that toggled between turning genes on and off two. Later, they built some safety checks into their bacteria. “To prevent unintentional or deleterious consequences, in 2009, we built a safety switch in the engineered bacteria’s genetic circuit that gets triggered after it gets exposed to a pathogen," says James Collins, a professor of biological engineering at MIT and faculty member at Harvard University’s Wyss Institute. “After getting rid of the pathogen, the engineered bacteria is designed to switch off and leave the patient's body.”
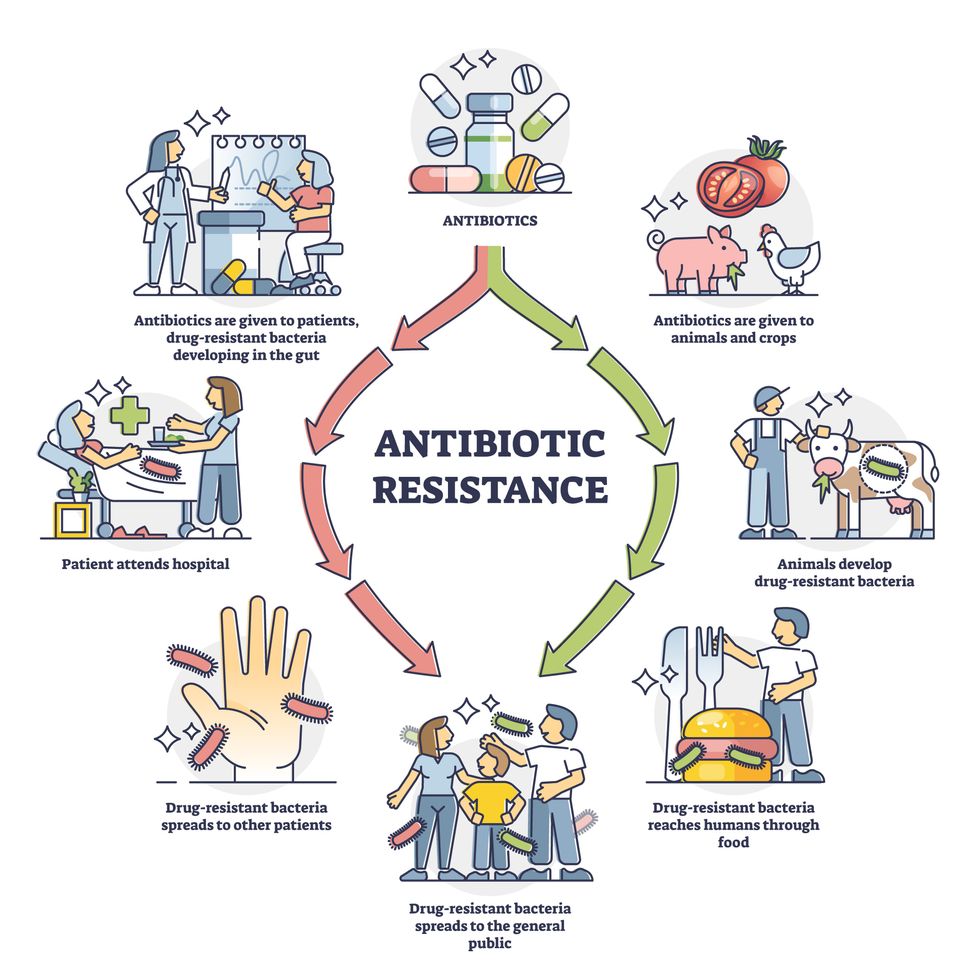
Overuse and misuse of antibiotics causes resistant strains to evolve
Adobe Stock
Seek and destroy
As the field of synthetic biology developed, scientists began using engineered bacteria to tackle superbugs. They first focused on Vibrio cholerae, which in the 19th and 20th century caused cholera pandemics in India, China, the Middle East, Europe, and Americas. Like many other bacteria, V. cholerae communicate with each other via quorum sensing, a process in which the microorganisms release different signaling molecules, to convey messages to its brethren. Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut. When untreated, cholera has a mortality rate of 25 to 50 percent and outbreaks frequently occur in developing countries, especially during floods and droughts.
Sometimes, however, V. cholerae makes mistakes. In 2008, researchers at Cornell University observed that when quorum sensing V. cholerae accidentally released high concentrations of a signaling molecule called CAI-1, it had a counterproductive effect—the pathogen couldn’t colonize the gut.
So the group, led by John March, professor of biological and environmental engineering, developed a novel strategy to combat V. cholerae. They genetically engineered E.coli to eavesdrop on V. cholerae communication networks and equipped it with the ability to release the CAI-1 molecules. That interfered with V. cholerae progress. Two years later, the Cornell team showed that V. cholerae-infected mice treated with engineered E.coli had a 92 percent survival rate.
These findings inspired researchers to sic the good bacteria present in foods like yogurt and kimchi onto the drug-resistant ones.
Three years later in 2011, Singapore-based scientists engineered E.coli to detect and destroy Pseudomonas aeruginosa, an often drug-resistant pathogen that causes pneumonia, urinary tract infections, and sepsis. Once the genetically engineered E.coli found its target through its quorum sensing molecules, it then released a peptide, that could eradicate 99 percent of P. aeruginosa cells in a test-tube experiment. The team outlined their work in a Molecular Systems Biology study.
“At the time, we knew that we were entering new, uncharted territory,” says lead author Matthew Chang, an associate professor and synthetic biologist at the National University of Singapore and lead author of the study. “To date, we are still in the process of trying to understand how long these microbes stay in our bodies and how they might continue to evolve.”
More teams followed the same path. In a 2013 study, MIT researchers also genetically engineered E.coli to detect P. aeruginosa via the pathogen’s quorum-sensing molecules. It then destroyed the pathogen by secreting a lab-made toxin.
Probiotics that fight
A year later in 2014, a Nature study found that the abundance of Ruminococcus obeum, a probiotic bacteria naturally occurring in the human microbiome, interrupts and reduces V.cholerae’s colonization— by detecting the pathogen’s quorum sensing molecules. The natural accumulation of R. obeum in Bangladeshi adults helped them recover from cholera despite living in an area with frequent outbreaks.
The findings from 2008 to 2014 inspired Collins and his team to delve into how good bacteria present in foods like yogurt and kimchi can attack drug-resistant bacteria. In 2018, Collins and his team developed the engineered probiotic strategy. They tweaked a bacteria commonly found in yogurt called Lactococcus lactis to treat cholera.
Engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut. --José Rubén Morones-Ramírez.
More scientists followed with more experiments. So far, researchers have engineered various probiotic organisms to fight pathogenic bacteria like Staphylococcus aureus (leading cause of skin, tissue, bone, joint and blood infections) and Clostridium perfringens (which causes watery diarrhea) in test-tube and animal experiments. In 2020, Russian scientists engineered a probiotic called Pichia pastoris to produce an enzyme called lysostaphin that eradicated S. aureus in vitro. Another 2020 study from China used an engineered probiotic bacteria Lactobacilli casei as a vaccine to prevent C. perfringens infection in rabbits.
In a study last year, Ramírez’s group at the Autonomous University of Nuevo León, engineered E. coli to detect quorum-sensing molecules from Methicillin-resistant Staphylococcus aureus or MRSA, a notorious superbug. The E. coli then releases a bacteriocin that kills MRSA. “An antibiotic is just a molecule that is not intelligent,” says Ramírez. “On the other hand, engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut.”
Collins and Timothy Lu, an associate professor of biological engineering at MIT, found that engineered E. coli can help treat other conditions—such as phenylketonuria, a rare metabolic disorder, that causes the build-up of an amino acid phenylalanine. Their start-up Synlogic aims to commercialize the technology, and has completed a phase 2 clinical trial.
Circumventing the challenges
The bacteria-engineering technique is not without pitfalls. One major challenge is that beneficial gut bacteria produce their own quorum-sensing molecules that can be similar to those that pathogens secrete. If an engineered bacteria’s biosensor is not specific enough, it will be ineffective.
Another concern is whether engineered bacteria might mutate after entering the gut. “As with any technology, there are risks where bad actors could have the capability to engineer a microbe to act quite nastily,” says Collins of MIT. But Collins and Ramírez both insist that the chances of the engineered bacteria mutating on its own are virtually non-existent. “It is extremely unlikely for the engineered bacteria to mutate,” Ramírez says. “Coaxing a living cell to do anything on command is immensely challenging. Usually, the greater risk is that the engineered bacteria entirely lose its functionality.”
However, the biggest challenge is bringing the curative bacteria to consumers. Pharmaceutical companies aren’t interested in antibiotics or their alternatives because it’s less profitable than developing new medicines for non-infectious diseases. Unlike the more chronic conditions like diabetes or cancer that require long-term medications, infectious diseases are usually treated much quicker. Running clinical trials are expensive and antibiotic-alternatives aren’t lucrative enough.
“Unfortunately, new medications for antibiotic resistant infections have been pushed to the bottom of the field,” says Lu of MIT. “It's not because the technology does not work. This is more of a market issue. Because clinical trials cost hundreds of millions of dollars, the only solution is that governments will need to fund them.” Lu stresses that societies must lobby to change how the modern healthcare industry works. “The whole world needs better treatments for antibiotic resistance.”
A surprising weapon in the fight against food poisoning
Phages, which are harmless viruses that destroy specific bacteria, are becoming useful tools to protect our food supply.
Every year, one in seven people in America comes down with a foodborne illness, typically caused by a bacterial pathogen, including E.Coli, listeria, salmonella, or campylobacter. That adds up to 48 million people, of which 120,000 are hospitalized and 3000 die, according to the Centers for Disease Control. And the variety of foods that can be contaminated with bacterial pathogens is growing too. In the 20th century, E.Coli and listeria lurked primarily within meat. Now they find their way into lettuce, spinach, and other leafy greens, causing periodic consumer scares and product recalls. Onions are the most recent suspected culprit of a nationwide salmonella outbreak.
Some of these incidents are almost inevitable because of how Mother Nature works, explains Divya Jaroni, associate professor of animal and food sciences at Oklahoma State University. These common foodborne pathogens come from the cattle's intestines when the animals shed them in their manure—and then they get washed into rivers and lakes, especially in heavy rains. When this water is later used to irrigate produce farms, the bugs end up on salad greens. Plus, many small farms do both—herd cattle and grow produce.
"Unfortunately for us, these pathogens are part of the microflora of the cows' intestinal tract," Jaroni says. "Some farmers may have an acre or two of cattle pastures, and an acre of a produce farm nearby, so it's easy for this water to contaminate the crops."
Food producers and packagers fight bacteria by potent chemicals, with chlorine being the go-to disinfectant. Cattle carcasses, for example, are typically washed by chlorine solutions as the animals' intestines are removed. Leafy greens are bathed in water with added chlorine solutions. However, because the same "bath" can be used for multiple veggie batches and chlorine evaporates over time, the later rounds may not kill all of the bacteria, sparing some. The natural and organic producers avoid chlorine, substituting it with lactic acid, a more holistic sanitizer, but even with all these efforts, some pathogens survive, sickening consumers and causing food recalls. As we farm more animals and grow more produce, while also striving to use fewer chemicals and more organic growing methods, it will be harder to control bacteria's spread.
"It took us a long time to convince the FDA phages were safe and efficient alternatives. But we had worked with them to gather all the data they needed, and the FDA was very supportive in the end."
Luckily, bacteria have their own killers. Called bacteriophages, or phages for short, they are viruses that prey on bacteria only. Under the electron microscope, they look like fantasy spaceships, with oblong bodies, spider-like legs and long tails. Much smaller than a bacterium, phages pierce the microbes' cells with their tails, sneak in and begin multiplying inside, eventually bursting the microbes open—and then proceed to infect more of them.
The best part is that these phages are harmless to humans. Moreover, recent research finds that millions of phages dwell on us and in us—in our nose, throat, skin and gut, protecting us from bacterial infections as part of our healthy microbiome. A recent study suggested that we absorb about 30 billion phages into our bodies on a daily basis. Now, ingeniously, they are starting to be deployed as anti-microbial agents in the food industry.
A Maryland-based phage research company called Intralytix is doing just that. Founded by Alexander Sulakvelidze, a microbiologist and epidemiologist who came to the United States from Tbilisi, the capital of Georgia, Intralytix makes and sells five different FDA-approved phage cocktails that work against some of the most notorious food pathogens: ListShield for Listeria, SalmoFresh for Salmonella, ShigaShield for Shigella, another foodborne bug, and EcoShield for E.coli, including the infamous strain that caused the Jack in the Box outbreak in 1993 that killed four children and sickened 732 people across four states. Last year, the FDA granted its approval to yet another Intralytix phage for managing Campylobacter contamination, named CampyShield. "We call it safety by nature," Sulakvelidze says.

Intralytix grows phages inside massive 1500-liter fermenters, feeding them bacterial "fodder."
Photo credit: Living Radiant Photography
Phage preparations are relatively straightforward to make. In nature, phages thrive in any body of water where bacteria live too, including rivers, lakes and bays. "I can dip a bucket into the Chesapeake Bay, and it will be full of all kinds of phages," Sulakvelidze says. "Sewage is another great place to look for specific phages of interest, because it's teeming with all sorts of bacteria—and therefore the viruses that prey on them."
In lab settings, Intralytix grows phages inside massive 1500-liter fermenters, feeding them bacterial "fodder." Once phages multiply enough, they are harvested, dispensed into containers and shipped to food producers who have adopted this disinfecting practice into their preparation process. Typically, it's done by computer-controlled sprayer systems that disperse mist-like phage preparations onto the food.
Unlike chemicals like chlorine or antibiotics, which kill a wide spectrum of bacteria, phages are more specialized, each feeding on specific microbial species. A phage that targets salmonella will not prey on listeria and vice versa. So food producers may sometimes use a combo of different phage preparations. Intralytix is continuously researching and testing new phages. With a contract from the National Institutes of Health, Intralytix is expanding its automated high-throughput robot that tests which phages work best against which bacteria, speeding up the development of the new phage cocktails.
Phages have other "talents." In her recent study, Jaroni found that phages have the ability to destroy bacterial biofilms—colonies of microorganisms that tend to grow on surfaces of the food processing equipment, surrounding themselves with protective coating that even very harsh chemicals can't crack.
"Phages are very clever," Jaroni says. "They produce enzymes that target the biofilms, and once they break through, they can reach the bacteria."
Convincing the FDA that phages were safe to use on food products was no easy feat, Sulakvelidze says. In his home country of Georgia, phages have been used as antimicrobial remedies for over a century, but the FDA was leery of using viruses as food safety agents. "It took us a long time to convince the FDA phages were safe and efficient alternatives," Sulakvelidze says. "But we had worked with them to gather all the data they needed, and the FDA was very supportive in the end."
The agency had granted Intralytix its first approval in 2006, and over the past 10 years, the company's sales increased by over 15-fold. "We currently sell to about 40 companies and are in discussions with several other large food producers," Sulakvelidze says. One indicator that the industry now understands and appreciates the science of phages was that his company was ranked as Top Food Safety Provider in 2021 by Food and Beverage Technology Review, he adds. Notably, phage sprays are kosher, halal and organic-certified.
Intralytix's phage cocktails to safeguard food from bacteria are approved for consumers in addition to food producers, but currently the company sells to food producers only. Selling retail requires different packaging like easy-to-use spray bottles and different marketing that would inform people about phages' antimicrobial qualities. But ultimately, giving people the ability to remove pathogens from their food with probiotic phage sprays is the goal, Sulakvelidze says.
It's not the company's only goal. Now Intralytix is going a step further, investigating phages' probiotic and therapeutic abilities. Because phages are highly specialized in the bacteria they target, they can be used to treat infections caused by specific pathogens while leaving the beneficial species of our microbiome intact. In an ongoing clinical trial with Mount Sinai, Intralytix is now investigating a potential phage treatment against a certain type of E. coli for patients with Crohn's disease, and is about to start another clinical trial for treating bacterial dysentery.
"Now that we have proved that phages are safe and effective against foodborne bacteria," Sulakvelidze says, "we are going to demonstrate their potential in therapeutic applications."
This article was first published by Leaps.org on October 27, 2021.
Lina Zeldovich has written about science, medicine and technology for Popular Science, Smithsonian, National Geographic, Scientific American, Reader’s Digest, the New York Times and other major national and international publications. A Columbia J-School alumna, she has won several awards for her stories, including the ASJA Crisis Coverage Award for Covid reporting, and has been a contributing editor at Nautilus Magazine. In 2021, Zeldovich released her first book, The Other Dark Matter, published by the University of Chicago Press, about the science and business of turning waste into wealth and health. You can find her on http://linazeldovich.com/ and @linazeldovich.
Debates over transgender athletes rage on, with new state bans and rules for Olympians, NCAA sports
Some argue that transgender females should be banned from competing with women in sports, while others think such bans are unfair, as the NCAA and other organizations try to keep up with research on how testosterone affects performance.
Ashley O’Connor, who was biologically male at birth but identifies as female, decided to compete in badminton as a girl during her senior year of high school in Downers Grove, Illinois. There was no team for boys, and a female friend and badminton player “practically bullied me into joining” the girls’ team. O’Connor, who is 18 and taking hormone replacement therapy for her gender transition, recalled that “it was easily one of the best decisions I have ever made.”
She believes there are many reasons why it’s important for transgender people to have the option of playing sports on the team of their choice. “It provides a sense of community,” said O’Connor, now a first-year student concentrating in psychology at the College of DuPage in Glen Ellyn, Illinois.
“It’s a great way to get a workout, which is good for physical and mental health,” she added. She also enjoyed the opportunity to be competitive, learn about her strengths and weaknesses, and just be normal. “Trans people have friends and trans people want to play sports with their friends, especially in adolescence,” she said.
However, in 18 states, many of which are politically conservative, laws prohibit transgender students from participating in sports consistent with their gender identity, according to the Movement Advancement Project, an independent, nonprofit think tank based in Boulder, Colo., that focuses on the rights of LGBTQ people. The first ban was passed in Idaho in 2020, although federal district judges have halted this legislation and a similar law in West Virginia from taking effect.
Proponents of the bans caution that transgender females would have an unfair biological advantage in competitive school sports with other girls or women as a result of being born as stronger males, potentially usurping the athletic accomplishments of other athletes.
“The future of women’s sports is at risk, and the equal rights of female athletes is being infringed,” said Penny Nance, CEO and president of Concerned Women for America, a legislative action committee in D.C. that seeks to impact culture to promote religious values.
“As the tidal wave of gender activism consumes sports from the Olympics on down, a backlash is being felt as parents are furious about the disregard for their daughters who have worked very hard to achieve success as athletes,” Nance added. “Former athletes, whose records are being shattered, are demanding answers.”
Meanwhile, opponents of the bans contend that they bar transgender athletes from playing sports with friends and learning the value of teamwork and other life lessons. These laws target transgender girls most often in kindergarten through high school but sometimes in college as well. Many local schools and state athletic associations already have their own guidelines “to both protect transgender people and ensure a level playing field for all athletes,” according to the Movement Advancement Project’s website. But statewide bans take precedence over these policies.
"It’s easy to sympathize on some level with arguments on both sides, and it’s likely going to be impossible to make everyone happy,” said Liz Joy, a past president of the American College of Sports Medicine.
In January, the National Collegiate Athletic Association (NCAA), based in Indianapolis, tried to sort out the controversy by implementing a new policy. It requires transgender students participating in female sports to prove that they’ve been taking treatments to suppress testosterone for at least one year before competition, as well as demonstrating that their testosterone level is sufficiently low, depending on the sport, through a blood test.
Then, in August, the NCAA clarified that these athletes also must take another blood test six months after their season has started that shows their testosterone levels aren’t too high. Additional guidelines will take effect next August.
Even with these requirements, “there is no plan that is going to be considered equitable and fair to all,” said Bradley Anawalt, an endocrinologist at the University of Washington School of Medicine. Biologically, he noted, there is still some evidence that a transgender female who initiates hormone therapy with estrogen and drops her testosterone to very low levels may have some advantage over other females, based on characteristics such as hand and foot size, height and perhaps strength.
Liz Joy, a past president of the American College of Sports Medicine, agrees that allowing transgender athletes to compete on teams of their self-identifying gender poses challenges. “It’s easy to sympathize on some level with arguments on both sides, and it’s likely going to be impossible to make everyone happy,” said Joy, a physician and senior medical director of wellness and nutrition at Intermountain Healthcare in Salt Lake City, Utah. While advocating for inclusion, she added that “sport was incredibly important in my life. I just want everyone to be able to benefit from it.”
One solution may be to allow transgender youth to play sports in a way that aligns with their gender identity until a certain age and before an elite level. “There are minimal or no potential financial stakes for most youth sports before age 13 or 14, and you do not have a lot of separation in athlete performance between most boys and girls until about age 13,” said Anwalt, who was a reviewer of the Endocrine Society’s national guidelines on transgender care.
Myron Genel, a professor emeritus and former chief of pediatric endocrinology at Yale School of Medicine, said it’s difficult to argue that height gives transgender females an edge because in some sports tall women already dominate over their shorter counterparts.
He added that the decision to allow transgender females to compete with other girls or women could hinge on when athletes began taking testosterone blockers. “If the process of conversion from male to female has been undertaken in the early stages of puberty, from my perspective, they have very little unique advantage,” said Genel, who advised the International Olympic Committee (IOC), based in Switzerland, on testosterone limits for transgender athletes.
Because young athletes’ bodies are still developing, “the differences in natural abilities are so massive that they would overwhelm any advantage a transgender athlete might have,” said Thomas H. Murray, president emeritus of The Hastings Center, a pioneering bioethics research institute in Garrison, New York, and author of the book “Good Sport,” which focuses on the ethics and values in the Olympics and other competitions.
“There’s no good reason to limit the participation of transgender athletes in the sports where male athletes don’t have an advantage over women,” such as sailing, archery and shooting events, Murray said. “The burden of proof rests on those who want to restrict participation by transgender athletes. They must show that in this sport, at this level of competition, transgender athletes have a conspicuous advantage.”
Last year, the IOC issued a new framework emphasizing that the Olympic rules related to transgender participation should be specific to each sport. “This is an evolving topic and there has been—as it will continue to be—new research coming out and new developments informing our approach,” and there’s currently no consensus on how testosterone affects performance across all sports, an IOC spokesperson told Leaps.org.
Many of the new laws prohibiting transgender people from competing in sports consistent with their gender identity specifically apply to transgender females. Yet, some experts say the issue also affects transgender males, nonbinary and intersex athletes.
“There has been quite a bit of attention paid to transgender females and their participation in biological female sports and almost minimal focus on transgender male competition in male sports or in any sports,” said Katherine Drabiak, associate professor of public health law and medical ethics at University of South Florida in Tampa. In fact, “transgender men, because they were born female, would be at a disadvantage of having less lean body mass, less strength and less muscular area as a general category compared to a biological male.”
While discussing transgender students’ participation in sports, it’s important to call attention to the toll that anti-transgender legislation can take on these young people’s well-being, said Jonah DeChants, a research scientist at The Trevor Project, a suicide prevention and mental health organization for LGBTQ youth. Recent polling found that 85 percent of transgender and nonbinary youth said that debates around anti-transgender laws had a negative impact on their mental health.
“The reality is simple: Most transgender girls want to play sports for the same reasons as any student—to benefit their health, to have fun, and to build connection with friends,” DeChants said. According to a new peer-reviewed qualitative study by researchers at The Trevor Project, many trans girls who participated in sports experienced harassment and stigma based on their gender identity, which can contribute to poor mental health outcomes and suicide risk.

In addition to badminton, O'Connor played other sports such as volleyball, and she plans to become an assistant coach or manager of her old high school's badminton team.
Ashley O'Connor
However, DeChants added, research also shows that young people who reported living in an accepting community, had access to LGBTQ-affirming spaces, or had social support from family and friends reported significantly lower rates of attempting suicide in the past year. “We urge coaches, educators and school administrators to seek LGBTQ-cultural competency training, implement zero tolerance policies for anti-trans bullying, and create safe, affirming environments for all transgender students on and off the field,” DeChants said.
O’Connor said her experiences on the athletic scene have been mostly positive. The politics of her community lean somewhat liberal, and she thinks it’s probably more supportive than some other areas of the country, though she noted the local library has received threats for hosting LGBTQ events. In addition to badminton, she also played baseball, lacrosse, volleyball, basketball and hockey. In the spring, she plans to become an assistant coach or manager for the girls’ badminton team at her old high school.
“When I played badminton, I never got any direct backlash from any coaches, competitors or teammates,” she said. “I had a few other teammates that identified as trans or nonbinary, [and] nearly all of the people I ever interacted with were super pleasant and treated me like any other normal person.” She added that transgender athletes “have aspirations. We have wants and needs. We have dreams. And at the end of the day, we just want to live our lives and be happy like everyone else.”

