Scientists redesign bacteria to tackle the antibiotic resistance crisis
Probiotic bacteria can be engineered to fight antibiotic-resistant superbugs by releasing chemicals that kill them.
In 1945, almost two decades after Alexander Fleming discovered penicillin, he warned that as antibiotics use grows, they may lose their efficiency. He was prescient—the first case of penicillin resistance was reported two years later. Back then, not many people paid attention to Fleming’s warning. After all, the “golden era” of the antibiotics age had just began. By the 1950s, three new antibiotics derived from soil bacteria — streptomycin, chloramphenicol, and tetracycline — could cure infectious diseases like tuberculosis, cholera, meningitis and typhoid fever, among others.
Today, these antibiotics and many of their successors developed through the 1980s are gradually losing their effectiveness. The extensive overuse and misuse of antibiotics led to the rise of drug resistance. The livestock sector buys around 80 percent of all antibiotics sold in the U.S. every year. Farmers feed cows and chickens low doses of antibiotics to prevent infections and fatten up the animals, which eventually causes resistant bacterial strains to evolve. If manure from cattle is used on fields, the soil and vegetables can get contaminated with antibiotic-resistant bacteria. Another major factor is doctors overprescribing antibiotics to humans, particularly in low-income countries. Between 2000 to 2018, the global rates of human antibiotic consumption shot up by 46 percent.
In recent years, researchers have been exploring a promising avenue: the use of synthetic biology to engineer new bacteria that may work better than antibiotics. The need continues to grow, as a Lancet study linked antibiotic resistance to over 1.27 million deaths worldwide in 2019, surpassing HIV/AIDS and malaria. The western sub-Saharan Africa region had the highest death rate (27.3 people per 100,000).
Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
To make it worse, our remedy pipelines are drying up. Out of the 18 biggest pharmaceutical companies, 15 abandoned antibiotic development by 2013. According to the AMR Action Fund, venture capital has remained indifferent towards biotech start-ups developing new antibiotics. In 2019, at least two antibiotic start-ups filed for bankruptcy. As of December 2020, there were 43 new antibiotics in clinical development. But because they are based on previously known molecules, scientists say they are inadequate for treating multidrug-resistant bacteria. Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
The rise of synthetic biology
To circumvent this dire future, scientists have been working on alternative solutions using synthetic biology tools, meaning genetically modifying good bacteria to fight the bad ones.
From the time life evolved on earth around 3.8 billion years ago, bacteria have engaged in biological warfare. They constantly strategize new methods to combat each other by synthesizing toxic proteins that kill competition.
For example, Escherichia coli produces bacteriocins or toxins to kill other strains of E.coli that attempt to colonize the same habitat. Microbes like E.coli (which are not all pathogenic) are also naturally present in the human microbiome. The human microbiome harbors up to 100 trillion symbiotic microbial cells. The majority of them are beneficial organisms residing in the gut at different compositions.
The chemicals that these “good bacteria” produce do not pose any health risks to us, but can be toxic to other bacteria, particularly to human pathogens. For the last three decades, scientists have been manipulating bacteria’s biological warfare tactics to our collective advantage.
In the late 1990s, researchers drew inspiration from electrical and computing engineering principles that involve constructing digital circuits to control devices. In certain ways, every cell in living organisms works like a tiny computer. The cell receives messages in the form of biochemical molecules that cling on to its surface. Those messages get processed within the cells through a series of complex molecular interactions.
Synthetic biologists can harness these living cells’ information processing skills and use them to construct genetic circuits that perform specific instructions—for example, secrete a toxin that kills pathogenic bacteria. “Any synthetic genetic circuit is merely a piece of information that hangs around in the bacteria’s cytoplasm,” explains José Rubén Morones-Ramírez, a professor at the Autonomous University of Nuevo León, Mexico. Then the ribosome, which synthesizes proteins in the cell, processes that new information, making the compounds scientists want bacteria to make. “The genetic circuit remains separated from the living cell’s DNA,” Morones-Ramírez explains. When the engineered bacteria replicates, the genetic circuit doesn’t become part of its genome.
Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut.
In 2000, Boston-based researchers constructed an E.coli with a genetic switch that toggled between turning genes on and off two. Later, they built some safety checks into their bacteria. “To prevent unintentional or deleterious consequences, in 2009, we built a safety switch in the engineered bacteria’s genetic circuit that gets triggered after it gets exposed to a pathogen," says James Collins, a professor of biological engineering at MIT and faculty member at Harvard University’s Wyss Institute. “After getting rid of the pathogen, the engineered bacteria is designed to switch off and leave the patient's body.”
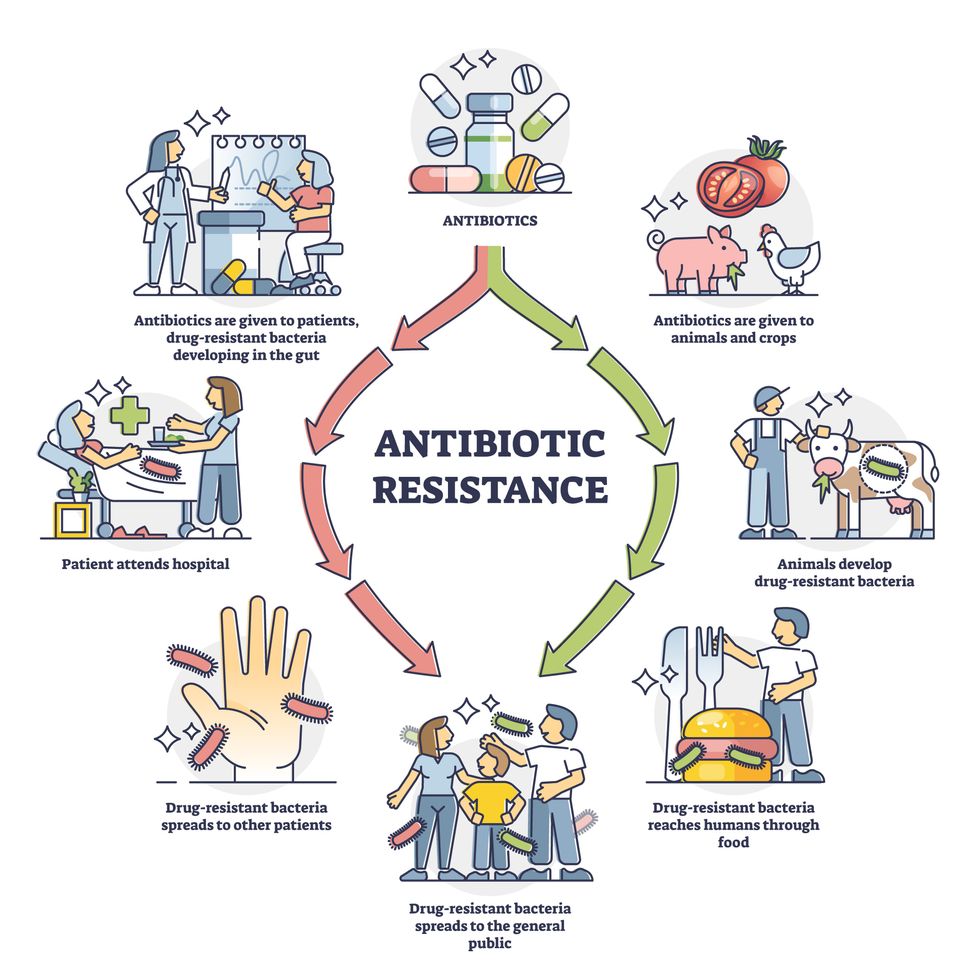
Overuse and misuse of antibiotics causes resistant strains to evolve
Adobe Stock
Seek and destroy
As the field of synthetic biology developed, scientists began using engineered bacteria to tackle superbugs. They first focused on Vibrio cholerae, which in the 19th and 20th century caused cholera pandemics in India, China, the Middle East, Europe, and Americas. Like many other bacteria, V. cholerae communicate with each other via quorum sensing, a process in which the microorganisms release different signaling molecules, to convey messages to its brethren. Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut. When untreated, cholera has a mortality rate of 25 to 50 percent and outbreaks frequently occur in developing countries, especially during floods and droughts.
Sometimes, however, V. cholerae makes mistakes. In 2008, researchers at Cornell University observed that when quorum sensing V. cholerae accidentally released high concentrations of a signaling molecule called CAI-1, it had a counterproductive effect—the pathogen couldn’t colonize the gut.
So the group, led by John March, professor of biological and environmental engineering, developed a novel strategy to combat V. cholerae. They genetically engineered E.coli to eavesdrop on V. cholerae communication networks and equipped it with the ability to release the CAI-1 molecules. That interfered with V. cholerae progress. Two years later, the Cornell team showed that V. cholerae-infected mice treated with engineered E.coli had a 92 percent survival rate.
These findings inspired researchers to sic the good bacteria present in foods like yogurt and kimchi onto the drug-resistant ones.
Three years later in 2011, Singapore-based scientists engineered E.coli to detect and destroy Pseudomonas aeruginosa, an often drug-resistant pathogen that causes pneumonia, urinary tract infections, and sepsis. Once the genetically engineered E.coli found its target through its quorum sensing molecules, it then released a peptide, that could eradicate 99 percent of P. aeruginosa cells in a test-tube experiment. The team outlined their work in a Molecular Systems Biology study.
“At the time, we knew that we were entering new, uncharted territory,” says lead author Matthew Chang, an associate professor and synthetic biologist at the National University of Singapore and lead author of the study. “To date, we are still in the process of trying to understand how long these microbes stay in our bodies and how they might continue to evolve.”
More teams followed the same path. In a 2013 study, MIT researchers also genetically engineered E.coli to detect P. aeruginosa via the pathogen’s quorum-sensing molecules. It then destroyed the pathogen by secreting a lab-made toxin.
Probiotics that fight
A year later in 2014, a Nature study found that the abundance of Ruminococcus obeum, a probiotic bacteria naturally occurring in the human microbiome, interrupts and reduces V.cholerae’s colonization— by detecting the pathogen’s quorum sensing molecules. The natural accumulation of R. obeum in Bangladeshi adults helped them recover from cholera despite living in an area with frequent outbreaks.
The findings from 2008 to 2014 inspired Collins and his team to delve into how good bacteria present in foods like yogurt and kimchi can attack drug-resistant bacteria. In 2018, Collins and his team developed the engineered probiotic strategy. They tweaked a bacteria commonly found in yogurt called Lactococcus lactis to treat cholera.
Engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut. --José Rubén Morones-Ramírez.
More scientists followed with more experiments. So far, researchers have engineered various probiotic organisms to fight pathogenic bacteria like Staphylococcus aureus (leading cause of skin, tissue, bone, joint and blood infections) and Clostridium perfringens (which causes watery diarrhea) in test-tube and animal experiments. In 2020, Russian scientists engineered a probiotic called Pichia pastoris to produce an enzyme called lysostaphin that eradicated S. aureus in vitro. Another 2020 study from China used an engineered probiotic bacteria Lactobacilli casei as a vaccine to prevent C. perfringens infection in rabbits.
In a study last year, Ramírez’s group at the Autonomous University of Nuevo León, engineered E. coli to detect quorum-sensing molecules from Methicillin-resistant Staphylococcus aureus or MRSA, a notorious superbug. The E. coli then releases a bacteriocin that kills MRSA. “An antibiotic is just a molecule that is not intelligent,” says Ramírez. “On the other hand, engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut.”
Collins and Timothy Lu, an associate professor of biological engineering at MIT, found that engineered E. coli can help treat other conditions—such as phenylketonuria, a rare metabolic disorder, that causes the build-up of an amino acid phenylalanine. Their start-up Synlogic aims to commercialize the technology, and has completed a phase 2 clinical trial.
Circumventing the challenges
The bacteria-engineering technique is not without pitfalls. One major challenge is that beneficial gut bacteria produce their own quorum-sensing molecules that can be similar to those that pathogens secrete. If an engineered bacteria’s biosensor is not specific enough, it will be ineffective.
Another concern is whether engineered bacteria might mutate after entering the gut. “As with any technology, there are risks where bad actors could have the capability to engineer a microbe to act quite nastily,” says Collins of MIT. But Collins and Ramírez both insist that the chances of the engineered bacteria mutating on its own are virtually non-existent. “It is extremely unlikely for the engineered bacteria to mutate,” Ramírez says. “Coaxing a living cell to do anything on command is immensely challenging. Usually, the greater risk is that the engineered bacteria entirely lose its functionality.”
However, the biggest challenge is bringing the curative bacteria to consumers. Pharmaceutical companies aren’t interested in antibiotics or their alternatives because it’s less profitable than developing new medicines for non-infectious diseases. Unlike the more chronic conditions like diabetes or cancer that require long-term medications, infectious diseases are usually treated much quicker. Running clinical trials are expensive and antibiotic-alternatives aren’t lucrative enough.
“Unfortunately, new medications for antibiotic resistant infections have been pushed to the bottom of the field,” says Lu of MIT. “It's not because the technology does not work. This is more of a market issue. Because clinical trials cost hundreds of millions of dollars, the only solution is that governments will need to fund them.” Lu stresses that societies must lobby to change how the modern healthcare industry works. “The whole world needs better treatments for antibiotic resistance.”
Your surgery could harm yourself and the planet. Here's what some doctors are doing about it.
Certain gases used for anesthesia are 3,000 times more damaging for the climate than CO2. Some anesthesiologists are pointing to other solutions.
This is part 1 of a three part series on a new generation of doctors leading the charge to make the health care industry more sustainable - for the benefit of their patients and the planet. Read part 2 here and part 3 here.
Susanne Koch, an anesthesiologist and neurologist, reached a pivot point when she was up to her neck in water, almost literally. The basement of her house in Berlin had flooded in the summer of 2018, when Berlin was pummeled by unusually strong rains. After she drained the house, “I wanted to dig into facts, to understand how exactly these extreme weather events are related to climate change,” she says.
Studying the scientific literature, she realized how urgent the climate crisis is, but the biggest shock was to learn that her profession contributed substantially to the problem: Inhalation gases used during medical procedures are among the most damaging greenhouse gases. Some inhalation gases are 3,000 times more damaging for the climate than CO2, Koch discovered. “Spending seven hours in the surgery room is the equivalent of driving a car for four days nonstop,” she says. Her job of helping people at Europe’s largest university hospital, the Charité in Berlin, was inadvertently damaging both the people and the planet.
“Nobody had ever even mentioned a word about that during my training,” Koch says.
On the whole, the medical sector is responsible for a disproportionally large percentage of greenhouse gas emissions, with the U.S. as the biggest culprit. According to a key paper published in 2020 in Health Affairs, the health industry “is among the most carbon-intensive service sectors in the industrialized world,” accounting for between 4.4 percent and 4.6 percent of greenhouse gas emissions. “It’s not just anesthesia but health care that has a problem,” says Jodi Sherman, anesthesiology professor and Medical Director of the Program on Healthcare Environmental Sustainability at Yale University as well as co-director of the Lancet Planetary Health Commission on Sustainable Healthcare. In the U.S., health care greenhouse gas emissions make up about 8.5 percent of domestic greenhouse gas emissions. They rose 6 percent from 2010 to 2018, to nearly 1,700 kilograms per person, more than in any other nation.
Of course, patients worry primarily about safety, not sustainability. Yet, Koch emphasizes that “as doctors, we have the responsibility to do no harm, and this includes making sure that we use resources as sustainably as possible.” Studies show that 2018 greenhouse gas and toxic air pollutant emissions resulted in the loss of 388,000 disability-adjusted life years in the U.S. alone. “Disease burden from health care pollution is of the same order of magnitude as deaths from preventable medical errors, and should be taken just as seriously,” Sherman cautions.
When Koch, the anesthesiologist, started discussing sustainable options with colleagues, the topic was immediately met with plenty of interest. Her experience is consistent with the latest representative poll of the nonprofit Foundation Health in Germany. Nine out of ten doctors were interested in urgently finding sustainable solutions for medical services but lacked knowhow and resources. For teaching purposes, Sherman and her team have developed the Yale Gassing Greener app that allows anesthesiologists to compare how much pollution they can avoid through choosing different anesthesia methods. Sherman also published professional guidelines intended to help her colleagues better understand how various methods affect carbon emissions.
Significant traces of inhalation gases have been found in Antarctica and the Himalayas, far from the vast majority of surgery rooms.
A solution espoused by both Sherman and Koch is comparatively simple: They stopped using desflurane, which is by far the most damaging of all inhalation gases to the climate. Its greenhouse effect is 2,590 times stronger than carbon dioxide. The Yale New Haven Hospital already stopped using desflurane in 2013, becoming the first known healthcare organization to eliminate a drug based on environmental grounds. Sherman points out that this resulted in saving more than $1.2 million in costs and 1,600 tons of CO2 equivalents, about the same as the exhaust from 360 passenger vehicles per year.
At the Charité, Koch claims that switching to other anesthesiology choices, such as propofol, has eliminated 90 percent of the climate gas emissions in the anesthesiology department since 2016. Young anesthesiologists are still taught to use desflurane as the standard because desflurane is absorbed less into the patients’ bodies, and they wake up faster. However, Koch who has worked as an anesthesiologist since 2006, says that with a little bit of experience, you can learn when to stop giving the propofol so it's timed just as well with a person’s wake-up process. In addition, “patients are less likely to feel nauseous after being given propofol,” Koch says. Intravenous drugs might require more skill, she adds, "but there is nothing unique to the drug desflurane that cannot be accomplished with other medications.”
Desflurane isn’t the only gas to be concerned about. Nitrous oxide is the second most damaging because it’s extremely long-lived in the environment, and it depletes the ozone layer. Climate-conscious anesthesiologists are phasing out this gas, too, or have implemented measures to decrease leaks.
Internationally, 192 governments agreed in the Kyoto protocol of 2005 to reduce halogenated hydrocarbons – resulting from inhalation gases, including desflurane and nitrous oxide – because of their immense climate-warming potential, and in 2016, they pledged to eliminate them by 2035. However, the use of inhalation anesthetics continues to increase worldwide, not least because more people access healthcare in developing countries, and because people in industrialized countries live longer and therefore need more surgeries. Significant traces of inhalation gases have been found in Antarctica and the Himalayas, far from the vast majority of surgery rooms.
Certain companies are now pushing new technology to capture inhalation gases before they are released into the atmosphere, but both Sherman and Koch believe marketing claims of 99 percent efficiency amount to greenwashing. After investigating the technology first-hand and visiting the company that is producing such filters in Germany, Koch concluded that such technology only reduces emissions by 25 percent. And Sherman believes such initiatives are akin to the fallacy of recycling plastic. In addition to questioning their efficiency, Sherman fears such technology “gives the illusion there is a magical solution that means I don’t need to change my behavior, reduce my waste and choose less harmful options.”
Financial interests are at play, too. “Desflurane is the most expensive inhalation gas, and some think, the most expensive must be the best,” Koch says. Both Koch and Sherman lament that efforts to increase sustainability in the medical sector are entirely voluntary in their countries and led by a few dedicated individual professionals while industry-wide standards and transparency are needed, a notion expressed in the American Hospital Association’s Sustainability Roadmap.

Susanne Koch, an anesthesiologist in Berlin, wants her colleagues to stop using a gas called desflurane, which is by far the most damaging of all inhalation gases to the climate.
Adobe Stock
Other countries have done more. The European Union recommends reducing inhalation gases and even contemplated a ban of desflurane, except in medical emergencies. In 2008, the National Health Service (NHS) created a Sustainable Development Unit, which measures CO2 emissions in the U.K. health sector. NHS is the first national health service that pledged to reach net zero carbon by 2040. The carbon footprint of the NHS fell by 26 percent from 1990 to 2019, mostly due to reduced use of certain inhalers and the switch to renewable energy for heat and power. “The evidence that the climate emergency is a health emergency is overwhelming,” said Nick Watts, the NHS Chief Sustainability Officer, in a press release, “with health professionals already needing to manage its symptoms.”
Sherman is a leading voice in demanding action in the U.S. To her, comprehensive solutions start with the mandatory, transparent measurement of emissions in the health sector to tackle the biggest sources of pollution. While the Biden administration highlighted its efforts to reduce these kinds of emissions during the United Nations Climate Conference (COP27) in November 2022 and U.S. delegates announced that more than 100 health care organizations signed the voluntary Health Sector Climate Pledge, with the aim to reduce emissions by 50 percent in the next eight years, Sherman is convinced that voluntary pledges are not enough. “Voluntary measures are insufficient,” she testified in congress. “The vast majority of U.S. health care organizations remain uncommitted to timely action. Those that are committed lack policies and knowledge to support necessary changes; even worse, existing policies drive inappropriate consumption of resources and pollution.”
Both Sherman and Koch look at the larger picture. “Health care organizations have an obligation to their communities to protect public health,” Sherman says. “We must lead by example. That includes setting ambitious, science-based carbon reduction targets to achieve net zero emissions before 2050. We must quantify current emissions and their sources, particularly throughout the health care supply chains.”
Have You Heard of the Best Sport for Brain Health?
In this week's Friday Five, research points to this brain healthiest of sports. Plus, the natural way to reprogram cells to a younger state, the network that could underlie many different mental illnesses, and a new test could diagnose autism in newborns. Plus, scientists 3D print an ear and attach it to woman
The Friday Five covers five stories in research that you may have missed this week. There are plenty of controversies and troubling ethical issues in science – and we get into many of them in our online magazine – but this news roundup focuses on scientific creativity and progress to give you a therapeutic dose of inspiration headed into the weekend.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
Here are the promising studies covered in this week's Friday Five:
- Reprogram cells to a younger state
- Pick up this sport for brain health
- Do all mental illnesses have the same underlying cause?
- New test could diagnose autism in newborns
- Scientists 3D print an ear and attach it to woman