Scientists redesign bacteria to tackle the antibiotic resistance crisis
Probiotic bacteria can be engineered to fight antibiotic-resistant superbugs by releasing chemicals that kill them.
In 1945, almost two decades after Alexander Fleming discovered penicillin, he warned that as antibiotics use grows, they may lose their efficiency. He was prescient—the first case of penicillin resistance was reported two years later. Back then, not many people paid attention to Fleming’s warning. After all, the “golden era” of the antibiotics age had just began. By the 1950s, three new antibiotics derived from soil bacteria — streptomycin, chloramphenicol, and tetracycline — could cure infectious diseases like tuberculosis, cholera, meningitis and typhoid fever, among others.
Today, these antibiotics and many of their successors developed through the 1980s are gradually losing their effectiveness. The extensive overuse and misuse of antibiotics led to the rise of drug resistance. The livestock sector buys around 80 percent of all antibiotics sold in the U.S. every year. Farmers feed cows and chickens low doses of antibiotics to prevent infections and fatten up the animals, which eventually causes resistant bacterial strains to evolve. If manure from cattle is used on fields, the soil and vegetables can get contaminated with antibiotic-resistant bacteria. Another major factor is doctors overprescribing antibiotics to humans, particularly in low-income countries. Between 2000 to 2018, the global rates of human antibiotic consumption shot up by 46 percent.
In recent years, researchers have been exploring a promising avenue: the use of synthetic biology to engineer new bacteria that may work better than antibiotics. The need continues to grow, as a Lancet study linked antibiotic resistance to over 1.27 million deaths worldwide in 2019, surpassing HIV/AIDS and malaria. The western sub-Saharan Africa region had the highest death rate (27.3 people per 100,000).
Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
To make it worse, our remedy pipelines are drying up. Out of the 18 biggest pharmaceutical companies, 15 abandoned antibiotic development by 2013. According to the AMR Action Fund, venture capital has remained indifferent towards biotech start-ups developing new antibiotics. In 2019, at least two antibiotic start-ups filed for bankruptcy. As of December 2020, there were 43 new antibiotics in clinical development. But because they are based on previously known molecules, scientists say they are inadequate for treating multidrug-resistant bacteria. Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
The rise of synthetic biology
To circumvent this dire future, scientists have been working on alternative solutions using synthetic biology tools, meaning genetically modifying good bacteria to fight the bad ones.
From the time life evolved on earth around 3.8 billion years ago, bacteria have engaged in biological warfare. They constantly strategize new methods to combat each other by synthesizing toxic proteins that kill competition.
For example, Escherichia coli produces bacteriocins or toxins to kill other strains of E.coli that attempt to colonize the same habitat. Microbes like E.coli (which are not all pathogenic) are also naturally present in the human microbiome. The human microbiome harbors up to 100 trillion symbiotic microbial cells. The majority of them are beneficial organisms residing in the gut at different compositions.
The chemicals that these “good bacteria” produce do not pose any health risks to us, but can be toxic to other bacteria, particularly to human pathogens. For the last three decades, scientists have been manipulating bacteria’s biological warfare tactics to our collective advantage.
In the late 1990s, researchers drew inspiration from electrical and computing engineering principles that involve constructing digital circuits to control devices. In certain ways, every cell in living organisms works like a tiny computer. The cell receives messages in the form of biochemical molecules that cling on to its surface. Those messages get processed within the cells through a series of complex molecular interactions.
Synthetic biologists can harness these living cells’ information processing skills and use them to construct genetic circuits that perform specific instructions—for example, secrete a toxin that kills pathogenic bacteria. “Any synthetic genetic circuit is merely a piece of information that hangs around in the bacteria’s cytoplasm,” explains José Rubén Morones-Ramírez, a professor at the Autonomous University of Nuevo León, Mexico. Then the ribosome, which synthesizes proteins in the cell, processes that new information, making the compounds scientists want bacteria to make. “The genetic circuit remains separated from the living cell’s DNA,” Morones-Ramírez explains. When the engineered bacteria replicates, the genetic circuit doesn’t become part of its genome.
Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut.
In 2000, Boston-based researchers constructed an E.coli with a genetic switch that toggled between turning genes on and off two. Later, they built some safety checks into their bacteria. “To prevent unintentional or deleterious consequences, in 2009, we built a safety switch in the engineered bacteria’s genetic circuit that gets triggered after it gets exposed to a pathogen," says James Collins, a professor of biological engineering at MIT and faculty member at Harvard University’s Wyss Institute. “After getting rid of the pathogen, the engineered bacteria is designed to switch off and leave the patient's body.”
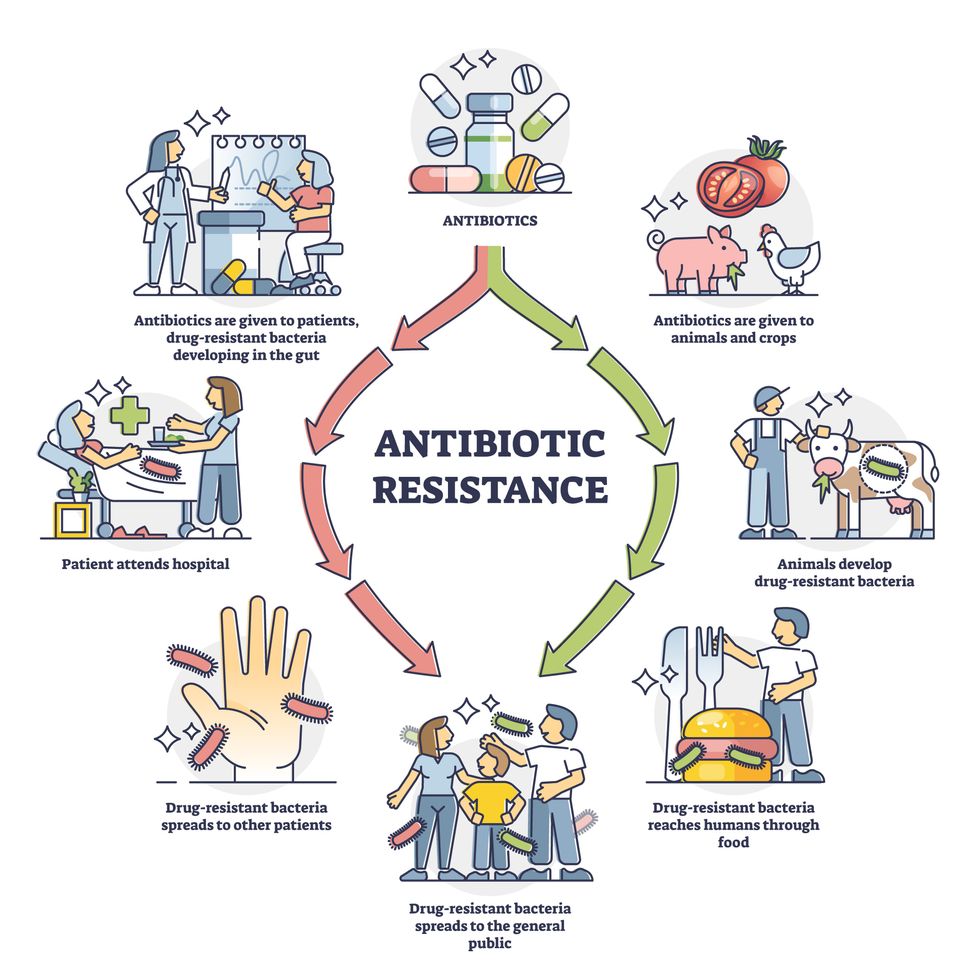
Overuse and misuse of antibiotics causes resistant strains to evolve
Adobe Stock
Seek and destroy
As the field of synthetic biology developed, scientists began using engineered bacteria to tackle superbugs. They first focused on Vibrio cholerae, which in the 19th and 20th century caused cholera pandemics in India, China, the Middle East, Europe, and Americas. Like many other bacteria, V. cholerae communicate with each other via quorum sensing, a process in which the microorganisms release different signaling molecules, to convey messages to its brethren. Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut. When untreated, cholera has a mortality rate of 25 to 50 percent and outbreaks frequently occur in developing countries, especially during floods and droughts.
Sometimes, however, V. cholerae makes mistakes. In 2008, researchers at Cornell University observed that when quorum sensing V. cholerae accidentally released high concentrations of a signaling molecule called CAI-1, it had a counterproductive effect—the pathogen couldn’t colonize the gut.
So the group, led by John March, professor of biological and environmental engineering, developed a novel strategy to combat V. cholerae. They genetically engineered E.coli to eavesdrop on V. cholerae communication networks and equipped it with the ability to release the CAI-1 molecules. That interfered with V. cholerae progress. Two years later, the Cornell team showed that V. cholerae-infected mice treated with engineered E.coli had a 92 percent survival rate.
These findings inspired researchers to sic the good bacteria present in foods like yogurt and kimchi onto the drug-resistant ones.
Three years later in 2011, Singapore-based scientists engineered E.coli to detect and destroy Pseudomonas aeruginosa, an often drug-resistant pathogen that causes pneumonia, urinary tract infections, and sepsis. Once the genetically engineered E.coli found its target through its quorum sensing molecules, it then released a peptide, that could eradicate 99 percent of P. aeruginosa cells in a test-tube experiment. The team outlined their work in a Molecular Systems Biology study.
“At the time, we knew that we were entering new, uncharted territory,” says lead author Matthew Chang, an associate professor and synthetic biologist at the National University of Singapore and lead author of the study. “To date, we are still in the process of trying to understand how long these microbes stay in our bodies and how they might continue to evolve.”
More teams followed the same path. In a 2013 study, MIT researchers also genetically engineered E.coli to detect P. aeruginosa via the pathogen’s quorum-sensing molecules. It then destroyed the pathogen by secreting a lab-made toxin.
Probiotics that fight
A year later in 2014, a Nature study found that the abundance of Ruminococcus obeum, a probiotic bacteria naturally occurring in the human microbiome, interrupts and reduces V.cholerae’s colonization— by detecting the pathogen’s quorum sensing molecules. The natural accumulation of R. obeum in Bangladeshi adults helped them recover from cholera despite living in an area with frequent outbreaks.
The findings from 2008 to 2014 inspired Collins and his team to delve into how good bacteria present in foods like yogurt and kimchi can attack drug-resistant bacteria. In 2018, Collins and his team developed the engineered probiotic strategy. They tweaked a bacteria commonly found in yogurt called Lactococcus lactis to treat cholera.
Engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut. --José Rubén Morones-Ramírez.
More scientists followed with more experiments. So far, researchers have engineered various probiotic organisms to fight pathogenic bacteria like Staphylococcus aureus (leading cause of skin, tissue, bone, joint and blood infections) and Clostridium perfringens (which causes watery diarrhea) in test-tube and animal experiments. In 2020, Russian scientists engineered a probiotic called Pichia pastoris to produce an enzyme called lysostaphin that eradicated S. aureus in vitro. Another 2020 study from China used an engineered probiotic bacteria Lactobacilli casei as a vaccine to prevent C. perfringens infection in rabbits.
In a study last year, Ramírez’s group at the Autonomous University of Nuevo León, engineered E. coli to detect quorum-sensing molecules from Methicillin-resistant Staphylococcus aureus or MRSA, a notorious superbug. The E. coli then releases a bacteriocin that kills MRSA. “An antibiotic is just a molecule that is not intelligent,” says Ramírez. “On the other hand, engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut.”
Collins and Timothy Lu, an associate professor of biological engineering at MIT, found that engineered E. coli can help treat other conditions—such as phenylketonuria, a rare metabolic disorder, that causes the build-up of an amino acid phenylalanine. Their start-up Synlogic aims to commercialize the technology, and has completed a phase 2 clinical trial.
Circumventing the challenges
The bacteria-engineering technique is not without pitfalls. One major challenge is that beneficial gut bacteria produce their own quorum-sensing molecules that can be similar to those that pathogens secrete. If an engineered bacteria’s biosensor is not specific enough, it will be ineffective.
Another concern is whether engineered bacteria might mutate after entering the gut. “As with any technology, there are risks where bad actors could have the capability to engineer a microbe to act quite nastily,” says Collins of MIT. But Collins and Ramírez both insist that the chances of the engineered bacteria mutating on its own are virtually non-existent. “It is extremely unlikely for the engineered bacteria to mutate,” Ramírez says. “Coaxing a living cell to do anything on command is immensely challenging. Usually, the greater risk is that the engineered bacteria entirely lose its functionality.”
However, the biggest challenge is bringing the curative bacteria to consumers. Pharmaceutical companies aren’t interested in antibiotics or their alternatives because it’s less profitable than developing new medicines for non-infectious diseases. Unlike the more chronic conditions like diabetes or cancer that require long-term medications, infectious diseases are usually treated much quicker. Running clinical trials are expensive and antibiotic-alternatives aren’t lucrative enough.
“Unfortunately, new medications for antibiotic resistant infections have been pushed to the bottom of the field,” says Lu of MIT. “It's not because the technology does not work. This is more of a market issue. Because clinical trials cost hundreds of millions of dollars, the only solution is that governments will need to fund them.” Lu stresses that societies must lobby to change how the modern healthcare industry works. “The whole world needs better treatments for antibiotic resistance.”
Every weekend since January, pediatrician Cora Collette Breuner has volunteered to give the COVID-19 vaccine to individuals from age 12 to 96 in an underserved community in Washington state.
Even though the COVID-19 vaccines have been shown to be incredibly safe and effective, there's still quite a bit of hesitancy among parents to vaccinate their teenage children, says Breuner, an adolescent medicine specialist at Seattle Children's Hospital and a past chair of the American Academy of Pediatrics' Committee on Adolescence. "They have questions and they have questions," she says.
Breuner patiently answers them all. Even then, parents—who have the final say in whether their child gets the vaccine—may be reluctant to sign off on it.
In 41 states, parents must consent for minors under age 18 to receive a COVID-19 vaccine. One state—Nebraska—requires parental consent for individuals under age 19, according to the Kaiser Family Foundation. Healthcare workers can't legally give teens COVID-19 vaccines otherwise. In a May report, the nonprofit healthcare organization highlights that from a legal perspective, "the landscape may be shifting slightly as more jurisdictions seek to encourage vaccination of young people."
Meanwhile, as the Delta variant creates a new surge in cases, some ethicists and pediatricians argue that state laws should be amended or loosened to allow minors to consent to COVID-19 vaccination on their own, without the need for parental permission.
"COVID-19 has killed millions of people around the world and disrupted the global economy," says pediatrician John Lantos. "It's a global catastrophe that requires special rules."
There are compelling arguments in favor of letting minors consent on their own, says Robyn Shapiro, a health care lawyer and a bioethicist in the Milwaukee area. "By that, I mean they're either old enough or they're evaluated in such a way that they have sufficient understanding of what they're agreeing to."
Shapiro and other ethicists argue that teens are perfectly capable of giving "informed consent"—a key principle in ethics that means fully understanding the benefits and risks of a medical intervention. To give informed consent, a person must be able to process that information in line with their own values. Only then can they make an autonomous choice and sign a consent form, Shapiro says.
Most states already have laws permitting minors to consent to testing and treatments related to sexually transmitted diseases, birth control, behavioral health, and substance abuse. It wouldn't be that much of a stretch to add COVID-19 vaccination to the list, Shapiro says. New Jersey and New York have introduced bills to let teens as young as 14 to consent to getting the COVID-19 vaccine and Minnesota has proposed a bill to allow children as young as 12 to give consent.
With any medical test or intervention, doctors often wrestle with how to best involve teens in conversations about their own health care, says John Lantos, a pediatrician and director of the Bioethics Center at Children's Mercy Kansas City.
"Most bioethicists would say that [teens] should be included to the degree that they have decision-making capacity," he says. "In most cases, that means including them in discussions with their parents in trying to achieve consensus about what the best choice may be."
COVID-19 vaccination also presents a unique circumstance, Lantos notes. It raises the question: Should teens have greater decisional authority because it's a public health emergency? In his opinion, the answer is yes. "COVID-19 has killed millions of people around the world and disrupted the global economy," says pediatrician Lantos. "It's a global catastrophe that requires special rules."
In North Carolina, state legislators are moving to do the opposite. State law currently allows those under 18 to make vaccination decisions on their own, but on Aug. 5, North Carolina's General Assembly approved a Republican-sponsored bill requiring parental consent for 12- to 17-year-olds to get a COVID-19 vaccine.
Kyle Brothers, a pediatrician in Louisville, Kentucky, says it's "ethically justifiable" for states to permit adolescents, especially those on the verge of adulthood, to consent to COVID-19 vaccination and other straightforward medical care.
In many cases, 16- and 17-year-old adolescents are capable of making well-informed decisions, says Brothers, a member of the American Academy of Pediatrics' Section on Bioethics. "The problem is, the law tends not to have that level of nuance," he adds. "We know in the real world that maturing and developing the ability to make decisions is a continuous process, but the law sets a bright line at age 18."
Lacking parental consent, some defiant teens are researching avenues to get vaccinated without their mom's or dad's knowledge. They may have turned to VaxTeen.org, a site operated by a Los Angeles teenager that provides information on consent laws by state.
If parents are wavering on the decision to give consent, Breuner recommends that they speak with a trusted healthcare provider about their specific concerns. These kinds of dialogues often can clarify lingering worries and may help drive up consent rates for teen vaccination.
Vaccine-hesitant parents should hear out their teens who wish to be vaccinated. Teenagers have their own opinions and belief systems, and parents should respect their child's choice to be vaccinated if they wish, considering the minimal risk of harm and the significant benefit to society as a whole.
George J. Annas, professor and director at the Center for Health Law, Ethics & Human Rights at Boston University, says parents have a legal obligation to provide their children with necessary medical treatment, or they could be found guilty of child neglect. The circumstances vary, but in the face of unrelenting COVID-19, he says parents have an ethical duty to consent to teens' vaccination because "the disease is rampant and children are dying."
The Nose Knows: Dogs Are Being Trained to Detect the Coronavirus
Security workers walk with detection dogs in an airport terminal.
Asher is eccentric and inquisitive. He loves an audience, likes keeping busy, and howls to be let through doors. He is a six-year-old working Cocker Spaniel, who, with five other furry colleagues, has now been trained to sniff body odor samples from humans to detect COVID-19 infections.
As the Delta variant and other new versions of the SARS-CoV-2 virus emerge, public health agencies are once again recommending masking while employers contemplate mandatory vaccination. While PCR tests remain the "gold standard" of COVID-19 tests, they can take hours to flag infections. To accelerate the process, scientists are turning to a new testing tool: sniffer dogs.
At the London School of Hygiene and Tropical Medicine (LSHTM), researchers deployed Asher and five other trained dogs to test sock samples from 200 asymptomatic, infected individuals and 200 healthy individuals. In May, they published the findings of the yearlong study in a preprint, concluding that dogs could identify COVID-19 infections with a high degree of accuracy – they could correctly identify a COVID-positive sample up to 94% of the time and a negative sample up to 92% of the time. The paper has yet to be peer-reviewed.
"Dogs can screen lots of people very quickly – 300 people per dog per hour. This means they could be used in places like airports or public venues like stadiums and maybe even workplaces," says James Logan, who heads the Department of Disease Control at LSHTM, adding that canines can also detect variants of SARS-CoV-2. "We included samples from two variants and the dogs could still detect them."
Detection dogs have been one of the most reliable biosensors for identifying the odor of human disease. According to Gemma Butlin, a spokesperson of Medical Detection Dogs, the UK-based charity that trained canines for the LSHTM study, the olfactory capabilities of dogs have been deployed to detect malaria, Parkinson's disease, different types of cancers, as well as pseudomonas, a type of bacteria known to cause infections in blood, lungs, eyes, and other parts of the human body.
COVID-19 has a distinctive smell — a result of chemicals known as volatile organic compounds released by infected body cells, which give off an odor "fingerprint."
"It's estimated that the percentage of a dog's brain devoted to analyzing odors is 40 times larger than that of a human," says Butlin. "Humans have around 5 million scent receptors dedicated to smell. Dogs have 350 million and can detect odors at parts per trillion. To put this into context, a dog can detect a teaspoon of sugar in a million gallons of water: two Olympic-sized pools full."
According to LSHTM scientists, COVID-19 has a distinctive smell — a result of chemicals known as volatile organic compounds released by infected body cells, which give off an odor "fingerprint." Other studies, too, have revealed that the SARS-CoV-2 virus has a distinct olfactory signature, detectable in the urine, saliva, and sweat of infected individuals. Humans can't smell the disease in these fluids, but dogs can.
"Our research shows that the smell associated with COVID-19 is at least partly due to small and volatile chemicals that are produced by the virus growing in the body or the immune response to the virus or both," said Steve Lindsay, a public health entomologist at Durham University, whose team collaborated with LSHTM for the study. He added, "There is also a further possibility that dogs can actually smell the virus, which is incredible given how small viruses are."
In April this year, researchers from the University of Pennsylvania and collaborators published a similar study in the scientific journal PLOS One, revealing that detection dogs could successfully discriminate between urine samples of infected and uninfected individuals. The accuracy rate of canines in this study was 96%. Similarly, last December, French scientists found that dogs were 76-100% effective at identifying individuals with COVID-19 when presented with sweat samples.
Grandjean Dominique, a professor at France's National Veterinary School of Alfort, who led the French study, said that the researchers used two types of dogs — search and rescue dogs, as they can sniff sweat, and explosive detection dogs, because they're often used at airports to find bomb ingredients. Dogs may very well be as good as PCR tests, said Dominique, but the goal, he added, is not to replace these tests with canines.
In France, the government gave the green light to train hundreds of disease detection dogs and deploy them in airports. "They will act as mass pre-test, and only people who are positive will undergo a PCR test to check their level of infection and the kind of variant," says Dominique. He thinks the dogs will be able to decrease the amount of PCR testing and potentially save money.
Since the accuracy rate for bio-detection dogs is fairly high, scientists think they could prove to be a quick diagnosis and mass screening tool, especially at ports, airports, train stations, stadiums, and public gatherings. Countries like Finland, Thailand, UAE, Italy, Chile, India, Australia, Pakistan, Saudi Arabia, Switzerland, and Mexico are already training and deploying canines for COVID-19 detection. The dogs are trained to sniff the area around a person, and if they find the odor of COVID-19 they will sit or stand back from an individual as a signal that they've identified an infection.
While bio-detection dogs seem promising for cheap, large-volume screening, many of the studies that have been performed to date have been small and in controlled environments. The big question is whether this approach work on people in crowded airports, not just samples of shirts and socks in a lab.
"The next step is 'real world' testing where they [canines] are placed in airports to screen people and see how they perform," says Anna Durbin, professor of international health at the John Hopkins Bloomberg School of Public Health. "Testing in real airports with lots of passengers and competing scents will need to be done."
According to Butlin of Medical Detection Dogs, scalability could be a challenge. However, scientists don't intend to have a dog in every waiting room, detecting COVID-19 or other diseases, she said.
"Dogs are the most reliable bio sensors on the planet and they have proven time and time again that they can detect diseases as accurately, if not more so, than current technological diagnostics," said Butlin. "We are learning from them all the time and what their noses know will one day enable the creation an 'E-nose' that does the same job – imagine a day when your mobile phone can tell you that you are unwell."

