Black Participants Are Sorely Absent from Medical Research

Black participants are under-represented in clinical research.
After years of suffering from mysterious symptoms, my mother Janice Thomas finally found a doctor who correctly diagnosed her with two autoimmune diseases, Lupus and Sjogren's. Both diseases are more prevalent in the black population than in other races and are often misdiagnosed.
The National Institutes of Health has found that minorities make up less than 10 percent of trial participants.
Like many chronic health conditions, a lack of understanding persists about their causes, individual manifestations, and best treatment strategies.
On the search for relief from chronic pain, my mother started researching options and decided to participate in clinical trials as a way to gain much-needed insights. In return, she received discounted medical testing and has played an active role in finding answers for all.
"When my doctor told me I could get financial or medical benefits from participating in clinical trials for the same test I was already doing, I figured it would be an easy way to get some answers at little to no cost," she says.
As a person of color, her presence in clinical studies is rare. The National Institutes of Health has found that minorities make up less than 10 percent of trial participants.
Without trial participation that is reflective of the general population, pharmaceutical companies and medical professionals are left guessing how various drugs work across racial lines. For example, albuterol, a widely used asthma treatment, was found to have decreased effectiveness for black American and Puerto Rican children. Many high mortality conditions, like cancer, also show different outcomes based on race.
Over the last decade, the pervasive lack of representation has left communities of color demanding higher levels of involvement in the research process. However, no consensus yet exists on how best to achieve this.
But experts suggest that before we can improve black participation in medical studies, key misconceptions must be addressed, such as the false assumption that such patients are unwilling to participate because they distrust scientists.
Jill A. Fisher, a professor in the Center for Bioethics at the University of North Carolina at Chapel Hill, learned in one study that mistrust wasn't the main barrier for black Americans. "There is a lot of evidence that researchers' recruitment of black Americans is generally poorly done, with many black patients simply not asked," Fisher says. "Moreover, the underrepresentation of black Americans is primarily true for efficacy trials - those testing whether an investigational drug might therapeutically benefit patients with specific illnesses."
Without increased minority participation, research will not accurately reflect the diversity of the general population.
Dr. Joyce Balls-Berry, a psychiatric epidemiologist and health educator, agrees that black Americans are often overlooked in the process. One study she conducted found that "enrollment of minorities in clinical trials meant using a variety of culturally appropriate strategies to engage participants," she explained.
To overcome this hurdle, The National Black Church Initiative (NBCI), a faith-based organization made up of 34,000 churches and over 15.7 million African Americans, last year urged the Food and Drug Administration to mandate diversity in all clinical trials before approving a drug or device. However, the FDA declined to implement the mandate, declaring that they don't have the authority to regulate diversity in clinical trials.
"African Americans have not been successfully incorporated into the advancement of medicine and research technologies as legitimate and natural born citizens of this country," admonishes NBCI's president Rev. Anthony Evans.
His words conjure a reminder of the medical system's insidious history for people of color. The most infamous incident is the Tuskegee syphilis scandal, in which white government doctors perpetrated harmful experiments on hundreds of unsuspecting black men for forty years, until the research was shut down in the early 1970s.
Today, in the second decade of twenty-first century, the pernicious narrative that blacks are outsiders in science and medicine must be challenged, says Dr. Danielle N. Lee, assistant professor of biological sciences at Southern Illinois University. And having majority white participants in clinical trials only furthers the notion that "whiteness" is the default.
According to Lee, black individuals often see themselves disconnected from scientific and medical processes. "One of the critiques with science and medical research is that communities of color, and black communities in particular, regard ourselves as outsiders of science," Lee says. "We are othered."
Without increased minority participation, research will not accurately reflect the diversity of the general population.
"We are all human, but we are different, and yes, even different populations of people require modified medical responses," she points out.
Another obstacle is that many trials have health requirements that exclude black Americans, like not wanting individuals who have high blood pressure or a history of stroke. Considering that this group faces health disparities at a higher rate than whites, this eliminates eligibility for millions of potential participants.
One way to increase the diversity in sample participation without an FDA mandate is to include more black Americans in both volunteer and clinical roles during the research process to increase accountability in treatment, education, and advocacy.
"When more of us participate in clinical trials, we help build out the basic data sets that account for health disparities from the start, not after the fact," Lee says. She also suggests that researchers involve black patient representatives throughout the clinical trial process, from the study design to the interpretation of results.
"This allows for the black community to give insight on how to increase trial enrollment and help reduce stigma," she explains.
Thankfully, partnerships are popping up like the one between The Howard University's Cancer Center and Driver, a platform that connects cancer patients to treatment and trials. These sorts of targeted and culturally tailored efforts allow black patients to receive assistance from well-respected organizations.
Some observers suggest that the federal government and pharmaceutical industries must step up to address the gap.
However, some experts say that the black community should not be held solely responsible for solving a problem it did not cause. Instead, some observers suggest that the federal government and pharmaceutical industries must step up to address the gap.
According to Balls-Berry, socioeconomic barriers like transportation, time off work, and childcare related to trial participation must be removed. "These are real-world issues and yet many times researchers have not included these things in their budgets."
When asked to comment, a spokesperson for BIO, the world's largest biotech trade association, emailed the following statement:
"BIO believes that that our members' products and services should address the needs of a diverse population, and enhancing participation in clinical trials by a diverse patient population is a priority for BIO and our member companies. By investing in patient education to improve awareness of clinical trial opportunities, we can reduce disparities in clinical research to better reflect the country's changing demographics."
For my mother, the patient suffering from autoimmune disease, the need for broad participation in medical research is clear. "Without clinical trials, we would have less diagnosis and solutions to diseases," she says. "I think it's an underutilized resource."
When doctors couldn’t stop her daughter’s seizures, this mom earned a PhD and found a treatment herself.
Savannah Salazar (left) and her mother, Tracy Dixon-Salazaar, who earned a PhD in neurobiology in the quest for a treatment of her daughter's seizure disorder.
Twenty-eight years ago, Tracy Dixon-Salazaar woke to the sound of her daughter, two-year-old Savannah, in the midst of a medical emergency.
“I entered [Savannah’s room] to see her tiny little body jerking about violently in her bed,” Tracy said in an interview. “I thought she was choking.” When she and her husband frantically called 911, the paramedic told them it was likely that Savannah had had a seizure—a term neither Tracy nor her husband had ever heard before.
Over the next several years, Savannah’s seizures continued and worsened. By age five Savannah was having seizures dozens of times each day, and her parents noticed significant developmental delays. Savannah was unable to use the restroom and functioned more like a toddler than a five-year-old.
Doctors were mystified: Tracy and her husband had no family history of seizures, and there was no event—such as an injury or infection—that could have caused them. Doctors were also confused as to why Savannah’s seizures were happening so frequently despite trying different seizure medications.
Doctors eventually diagnosed Savannah with Lennox-Gaustaut Syndrome, or LGS, an epilepsy disorder with no cure and a poor prognosis. People with LGS are often resistant to several kinds of anti-seizure medications, and often suffer from developmental delays and behavioral problems. People with LGS also have a higher chance of injury as well as a higher chance of sudden unexpected death (SUDEP) due to the frequent seizures. In about 70 percent of cases, LGS has an identifiable cause such as a brain injury or genetic syndrome. In about 30 percent of cases, however, the cause is unknown.
Watching her daughter struggle through repeated seizures was devastating to Tracy and the rest of the family.
“This disease, it comes into your life. It’s uninvited. It’s unannounced and it takes over every aspect of your daily life,” said Tracy in an interview with Today.com. “Plus it’s attacking the thing that is most precious to you—your kid.”
Desperate to find some answers, Tracy began combing the medical literature for information about epilepsy and LGS. She enrolled in college courses to better understand the papers she was reading.
“Ironically, I thought I needed to go to college to take English classes to understand these papers—but soon learned it wasn’t English classes I needed, It was science,” Tracy said. When she took her first college science course, Tracy says, she “fell in love with the subject.”
Tracy was now a caregiver to Savannah, who continued to have hundreds of seizures a month, as well as a full-time student, studying late into the night and while her kids were at school, using classwork as “an outlet for the pain.”
“I couldn’t help my daughter,” Tracy said. “Studying was something I could do.”
Twelve years later, Tracy had earned a PhD in neurobiology.
After her post-doctoral training, Tracy started working at a lab that explored the genetics of epilepsy. Savannah’s doctors hadn’t found a genetic cause for her seizures, so Tracy decided to sequence her genome again to check for other abnormalities—and what she found was life-changing.
Tracy discovered that Savannah had a calcium channel mutation, meaning that too much calcium was passing through Savannah’s neural pathways, leading to seizures. The information made sense to Tracy: Anti-seizure medications often leech calcium from a person’s bones. When doctors had prescribed Savannah calcium supplements in the past to counteract these effects, her seizures had gotten worse every time she took the medication. Tracy took her discovery to Savannah’s doctor, who agreed to prescribe her a calcium blocker.
The change in Savannah was almost immediate.
Within two weeks, Savannah’s seizures had decreased by 95 percent. Once on a daily seven-drug regimen, she was soon weaned to just four, and then three. Amazingly, Tracy started to notice changes in Savannah’s personality and development, too.
“She just exploded in her personality and her talking and her walking and her potty training and oh my gosh she is just so sassy,” Tracy said in an interview.
Since starting the calcium blocker eleven years ago, Savannah has continued to make enormous strides. Though still unable to read or write, Savannah enjoys puzzles and social media. She’s “obsessed” with boys, says Tracy. And while Tracy suspects she’ll never be able to live independently, she and her daughter can now share more “normal” moments—something she never anticipated at the start of Savannah’s journey with LGS. While preparing for an event, Savannah helped Tracy get ready.
“We picked out a dress and it was the first time in our lives that we did something normal as a mother and a daughter,” she said. “It was pretty cool.”
A robot server, controlled remotely by a disabled worker, delivers drinks to patrons at the DAWN cafe in Tokyo.
A sleek, four-foot tall white robot glides across a cafe storefront in Tokyo’s Nihonbashi district, holding a two-tiered serving tray full of tea sandwiches and pastries. The cafe’s patrons smile and say thanks as they take the tray—but it’s not the robot they’re thanking. Instead, the patrons are talking to the person controlling the robot—a restaurant employee who operates the avatar from the comfort of their home.

It’s a typical scene at DAWN, short for Diverse Avatar Working Network—a cafe that launched in Tokyo six years ago as an experimental pop-up and quickly became an overnight success. Today, the cafe is a permanent fixture in Nihonbashi, staffing roughly 60 remote workers who control the robots remotely and communicate to customers via a built-in microphone.
More than just a creative idea, however, DAWN is being hailed as a life-changing opportunity. The workers who control the robots remotely (known as “pilots”) all have disabilities that limit their ability to move around freely and travel outside their homes. Worldwide, an estimated 16 percent of the global population lives with a significant disability—and according to the World Health Organization, these disabilities give rise to other problems, such as exclusion from education, unemployment, and poverty.
These are all problems that Kentaro Yoshifuji, founder and CEO of Ory Laboratory, which supplies the robot servers at DAWN, is looking to correct. Yoshifuji, who was bedridden for several years in high school due to an undisclosed health problem, launched the company to help enable people who are house-bound or bedridden to more fully participate in society, as well as end the loneliness, isolation, and feelings of worthlessness that can sometimes go hand-in-hand with being disabled.
“It’s heartbreaking to think that [people with disabilities] feel they are a burden to society, or that they fear their families suffer by caring for them,” said Yoshifuji in an interview in 2020. “We are dedicating ourselves to providing workable, technology-based solutions. That is our purpose.”
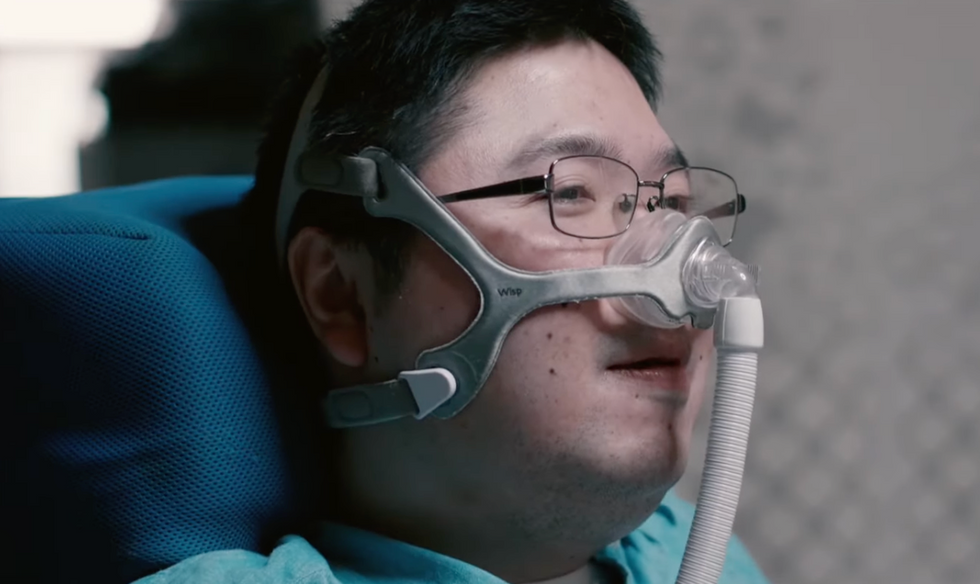
Shota, Kuwahara, a DAWN employee with muscular dystrophy, agrees. "There are many difficulties in my daily life, but I believe my life has a purpose and is not being wasted," he says. "Being useful, able to help other people, even feeling needed by others, is so motivational."

