Can You Trust Your Gut for Food Advice?
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.

Which foods are actually healthy for your individual gut microbiome? Several companies are offering personalized dietary guidance based on your test results, but their answers in one experiment turned up with some conflicting advice.
I recently got on the scale to weigh myself, thinking I've got to eat better. With so many trendy diets today claiming to improve health, from Keto to Paleo to Whole30, it can be confusing to figure out what we should and shouldn't eat for optimal nutrition.
A number of companies are now selling the concept of "personalized" nutrition based on the genetic makeup of your individual gut bugs.
My next thought was: I've got to lose a few pounds.
Consider a weird factoid: In addition to my fat, skin, bone and muscle, I'm carrying around two or three pounds of straight-up bacteria. Like you, I am the host to trillions of micro-organisms that live in my gut and are collectively known as my microbiome. An explosion of research has occurred in the last decade to try to understand exactly how these microbial populations, which are unique to each of us, may influence our overall health and potentially even our brains and behavior.
Lots of mysteries still remain, but it is established that these "bugs" are crucial to keeping our body running smoothly, performing functions like stimulating the immune system, synthesizing important vitamins, and aiding digestion. The field of microbiome science is evolving rapidly, and a number of companies are now selling the concept of "personalized" nutrition based on the genetic makeup of your individual gut bugs. The two leading players are Viome and DayTwo, but the landscape includes the newly launched startup Onegevity Health and others like Thryve, which offers customized probiotic supplements in addition to dietary recommendations.
The idea has immediate appeal – if science could tell you exactly what to make for lunch and what to avoid, you could forget about the fad diets and go with your own bespoke food pyramid. Wondering if the promise might be too good to be true, I decided to perform my own experiment.
Last fall, I sent the identical fecal sample to both Viome (I paid $425, but the price has since dropped to $299) and DayTwo ($349). A couple of months later, both reports finally arrived, and I eagerly opened each app to compare their recommendations.
First, I examined my results from Viome, which was founded in 2016 in Cupertino, Calif., and declares without irony on its website that "conflicting food advice is now obsolete."
I learned I have "average" metabolic fitness and "average" inflammatory activity in my gut, which are scores that the company defines based on a proprietary algorithm. But I have "low" microbial richness, with only 62 active species of bacteria identified in my sample, compared with the mean of 157 in their test population. I also received a list of the specific species in my gut, with names like Lactococcus and Romboutsia.
But none of it meant anything to me without actionable food advice, so I clicked through to the Recommendations page and found a list of My Superfoods (cranberry, garlic, kale, salmon, turmeric, watermelon, and bone broth) and My Foods to Avoid (chickpeas, kombucha, lentils, and rice noodles). There was also a searchable database of many foods that had been categorized for me, like "bell pepper; minimize" and "beef; enjoy."
"I just don't think sufficient data is yet available to make reliable personalized dietary recommendations based on one's microbiome."
Next, I looked at my results from DayTwo, which was founded in 2015 from research out of the Weizmann Institute of Science in Israel, and whose pitch to consumers is, "Blood sugar made easy. The algorithm diet personalized to you."
This app had some notable differences. There was no result about my metabolic fitness, microbial richness, or list of the species in my sample. There was also no list of superfoods or foods to avoid. Instead, the app encouraged me to build a meal by searching for foods in their database and combining them in beneficial ways for my blood sugar. Two slices of whole wheat bread received a score of 2.7 out of 10 ("Avoid"), but if combined with one cup of large curd cottage cheese, the score improved to 6.8 ("Limit"), and if I added two hard-boiled eggs, the score went up to 7.5 ("Good").
Perusing my list of foods with "Excellent" scores, I noticed some troubling conflicts with the other app. Lentils, which had been a no-no according to Viome, received high marks from DayTwo. Ditto for Kombucha. My purported superfood of cranberry received low marks. Almonds got an almost perfect score (9.7) while Viome told me to minimize them. I found similarly contradictory advice for foods I regularly eat, including navel oranges, peanuts, pork, and beets.
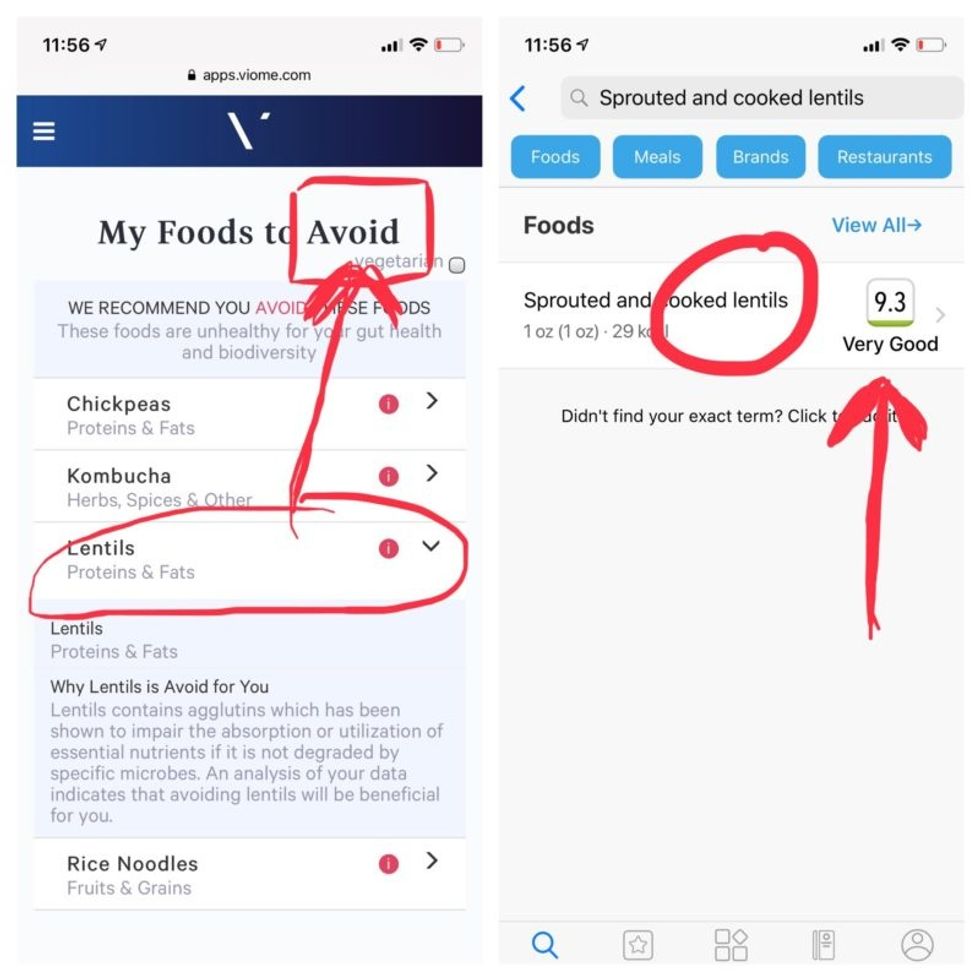
Contradictory dietary guidance that Kira Peikoff received from Viome (left) and DayTwo from an identical sample.
To be sure, there was some overlap. Both apps agreed on rice noodles (bad), chickpeas (bad), honey (bad), carrots (good), and avocado (good), among other foods.
But still, I was left scratching my head. Which set of recommendations should I trust, if either? And what did my results mean for the accuracy of this nascent field?
I called a couple of experts to find out.
"I have worked on the microbiome and nutrition for the last 20 years and I would be absolutely incapable of finding you evidence in the scientific literature that lentils have a detrimental effect based on the microbiome," said Dr. Jens Walter, an Associate Professor and chair for Nutrition, Microbes, and Gastrointestinal Health at the University of Alberta. "I just don't think sufficient data is yet available to make reliable personalized dietary recommendations based on one's microbiome. And even if they would have proprietary algorithms, at least one of them is not doing it right."
There is definite potential for personalized nutrition based on the microbiome, he said, but first, predictive models must be built and standardized, then linked to clinical endpoints, and tested in a large sample of healthy volunteers in order to enable extrapolations for the general population.
"It is mindboggling what you would need to do to make this work," he observed. "There are probably hundreds of relevant dietary compounds, then the microbiome has at least a hundred relevant species with a hundred or more relevant genes each, then you'd have to put all this together with relevant clinical outcomes. And there's a hundred-fold variation in that information between individuals."
However, Walter did acknowledge that the companies might be basing their algorithms on proprietary data that could potentially connect all the dots. I reached out to them to find out.
Amir Golan, the Chief Commercial Officer of DayTwo, told me, "It's important to emphasize this is a prediction, as the microbiome field is in a very early stage of research." But he added, "I believe we are the only company that has very solid science published in top journals and we can bring very actionable evidence and benefit to our uses."
He was referring to pioneering work out of the Weizmann Institute that was published in 2015 in the journal Cell, which logged the glycemic responses of 800 people in response to nearly 50,000 meals; adding information about the subjects' microbiomes enabled more accurate glycemic response predictions. Since then, Golan said, additional trials have been conducted, most recently with the Mayo Clinic, to duplicate the results, and other studies are ongoing whose results have not yet been published.
He also pointed out that the microbiome was merely one component that goes into building a client's profile, in addition to medical records, including blood glucose levels. (I provided my HbA1c levels, a measure of average blood sugar over the previous several months.)
"We are not saying we want to improve your gut microbiome. We provide a dynamic tool to help guide what you should eat to control your blood sugar and think about combinations," he said. "If you eat one thing, or with another, it will affect you in a different way."
Viome acknowledged that the two companies are taking very different approaches.
"DayTwo is primarily focused on the glycemic response," Naveen Jain, the CEO, told me. "If you can only eat butter for rest of your life, you will have no glycemic response but will probably die of a heart attack." He laughed. "Whereas we came from very different angle – what is happening inside the gut at a microbial level? When you eat food like spinach, how will that be metabolized in the gut? Will it produce the nutrients you need or cause inflammation?"
He said his team studied 1000 people who were on continuous glucose monitoring and fed them 45,000 meals, then built a proprietary data prediction model, looking at which microbes existed and how they actively broke down the food.
Jain pointed out that DayTwo sequences the DNA of the microbes, while Viome sequences the RNA – the active expression of DNA. That difference, in his opinion, is key to making accurate predictions.
"DNA is extremely stable, so when you eat any food and measure the DNA [in a fecal sample], you get all these false positives--you get DNA from plant food and meat, and you have no idea if those organisms are dead and simply transient, or actually exist. With RNA, you see what is actually alive in the gut."
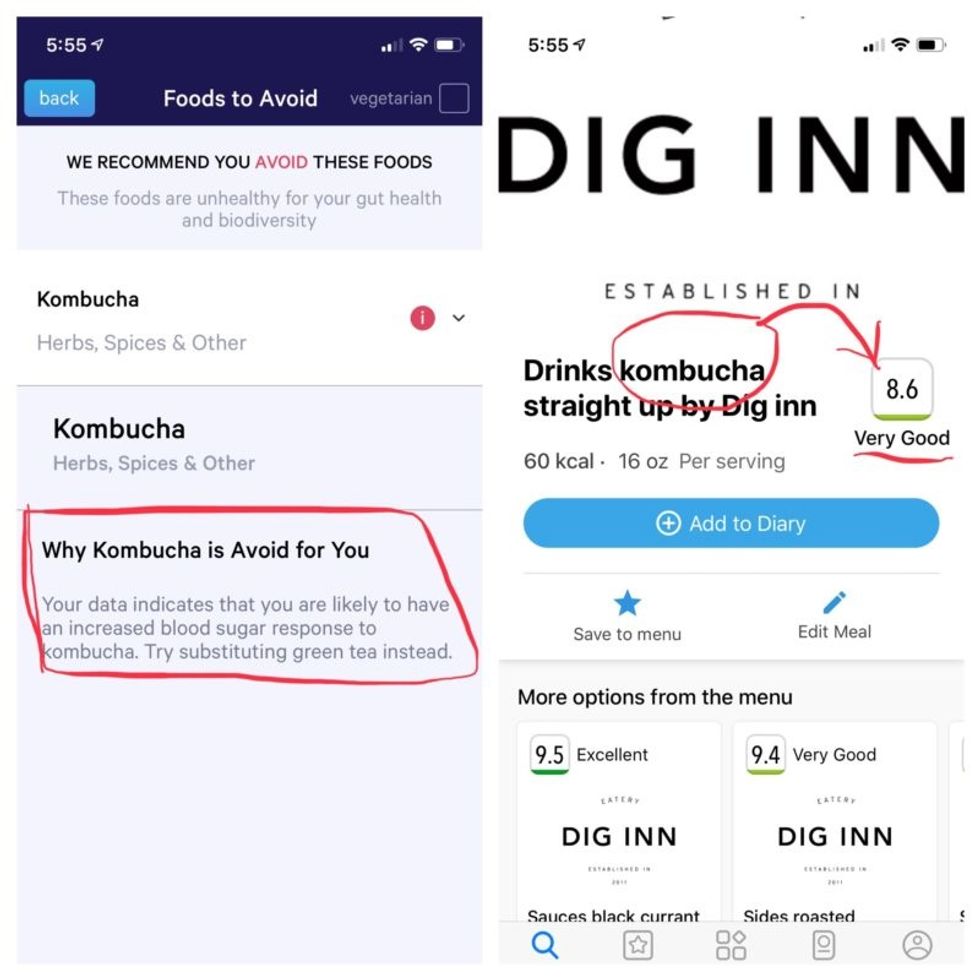
More contradictory food advice from Viome (left) and DayTwo.
Note that controversy exists over how it is possible with a fecal sample to effectively measure RNA, which degrades within minutes, though Jain said that his company has the technology to keep RNA stable for fourteen days.
Viome's approach, Jain maintains, is 90 percent accurate, based on as-yet unpublished data; a patent was filed just last week. DayTwo's approach is 66 percent accurate according to the latest published research.
Natasha Haskey, a registered dietician and doctoral student conducting research in the field of microbiome science and nutrition, is skeptical of both companies. "We can make broad statements, like eat more fruits and vegetables and fiber, but when it comes to specific foods, the science is just not there yet," she said. "I think there is a future, and we will be doing that someday, but not yet. Maybe we will be closer in ten years."
Professor Walter wholeheartedly agrees with Haskey, and suggested that if people want to eat a gut-healthy diet, they should focus on beneficial oils, fruits and vegetables, fish, a variety of whole grains, poultry and beans, and limit red meat and cheese, as well as avoid processed meats.
"These services are far over the tips of their science skis," Arthur Caplan, the founding head of New York University's Division of Medical Ethics, said in an email. "We simply don't know enough about the gut microbiome, its fluctuations and variability from person to person to support general [direct-to-consumer] testing. This is simply premature. We need standards for accuracy, specificity, and sensitivity, plus mandatory competent counseling for all such testing. They don't exist. Neither should DTC testing—yet."
Meanwhile, it's time for lunch. I close out my Viome and DayTwo apps and head to the kitchen to prepare a peanut butter sandwich. My gut tells me I'll be just fine.
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
A new way to help kids with ADHD: Treat adult ADHD
Adults who have ADHD can benefit from treatment, and some researchers think their kids' ADHD symptoms may get better as a result.
When a child is diagnosed with attention deficit hyperactivity disorder (ADHD), it can often be a surprise to the parents that one of them has ADHD as well. They may have experienced some of the symptoms but never had the condition diagnosed.
Physicians, however, are usually less surprised because they know that ADHD is a very heritable disorder. According to a 2015 study, if a parent has ADHD, the child has up to a 57 percent chance of having it, and the child’s risk is 32 percent if their sibling has it.
“There have been 20 to 30 twin studies that show that the heritability of ADHD is about 70 percent,” meaning that both twins have it, says Stephen Faraone, distinguished professor and vice chair for research at SUNY Upstate Medical University. “It is as heritable as schizophrenia, bipolar disorder, autism or other psychiatric disorders that people tend to think are more biological than ADHD for some reason.”
More attention needed for adult ADHD
Brad McAlister, CMSE, executive director of the American Professional Society of ADHD & Related Disorders, or APSARD, explains that the consequences of untreated ADHD in adults are very well documented. The prevalence of ADHD in U.S. adults is 4.4 percent or about 11 million people.
Many adults go undiagnosed for decades or are misdiagnosed by providers. McAlister says that 75 percent are not receiving treatment. “The U.S. economic burden of adult ADHD is $105 to $194 billion annually,” he says. “The negative consequences on peoples’ lives include higher risks of dropping out of school, losing jobs, financial debt, divorce, fractured relationships, substance use disorders, and co-occurring depression/anxiety.”
One of the negative impacts of undiagnosed ADHD in adults is the effect that it can have on their children who have ADHD.
Adult ADHD is currently treated by a broad range of health care providers with different educational backgrounds and in different practice settings. In August, APSARD published the first U.S. guidelines for adult ADHD. “The creation of guidelines for ADHD in adults will allow all practitioners to benefit from the best evidence about diagnosing and treating the disorder,” McAlister says.
Faraone explains that the guidelines are intended to help practitioners understand the best practices for adults with ADHD, including screening and other ways of evaluating whether someone has it. He recently completed a study of what he calls the Metrics of Quality Care for adults with ADHD.
“We looked at a sizable group of primary care practices in the U.S., and we learned that although quality care for adults with ADHD has been gradually improving over the past decade, there are many areas where it is still far behind where it needs to be,” he says. “That’s consistent with other studies that show that in primary care for adults, ADHD is not treated nearly as well as it is treated in specialty and psychiatry care.”
How kids with ADHD are affected
One of the negative impacts of undiagnosed ADHD in adults is the effect that it can have on their children who have ADHD because their ability to care for that child’s special needs may be impaired.
“The treatments that are most effective in treating children with ADHD are medication and behavioral interventions as their reward bait, and at home, it’s the parent that administers them,” says Mark A. Stein, director of the ADHD and Related Disorders Program at Seattle Children’s Hospital. “Adults with ADHD have difficulties with time management and organization skills, so they will have a hard time making sure their child is ready for school, has breakfast, has their medications, etcetera.”
Even more challenging than getting a prescription, Stein adds, is finding a psychologist or therapist who is skilled in evaluating and working with children with ADHD and their parents. If left undiagnosed and untreated, adult ADHD may also interfere with getting a good evaluation for the child.
“If you have ADHD and your mind is wandering and you don’t have all of the forms from the school for your provider, and you’re focused on the bad day you’re having rather than giving a history of your child, all of that is going to delay getting an effective treatment for your child,” Stein says. “So that’s why it’s important to identify ADHD in parents.”
Promising research and training
After delays due to the pandemic, Stein and his colleague Andrea Chronis-Tuscano, professor and director of the Maryland ADHD Program at the University of Maryland, are now about two years into what they anticipate will be a six-year study that involves treating parents who have children with untreated ADHD symptoms. The goal is to see whether treating the parent first with medication and training, or just the training, helps the child’s symptoms due to improved parenting. They are also studying whether they can postpone the need for medication until children are older, when it’s more effective.
“Pediatricians are more aware of ADHD in parents because of our study,” Stein says. “They’re also more aware of the shortcomings in our healthcare delivery system in terms of how hard it is to find providers who are comfortable treating adult ADHD.”
“Besides depression, ADHD is the other disorder that parents have that really impacts kids significantly," Stein says. “With treatment, many people with ADHD do very well."
That said, he’s seen a significant improvement in the past decade with increased recognition of ADHD in adults. “It started with pediatricians recognizing that post-partum depression impacted the mother’s ability to care for her children and making it routine to screen for depression in parents of kids,” he says. “Besides depression, ADHD is the other disorder that parents have that really impacts kids significantly, so it’s important for them to be aware of characteristics of [ADHD in] parents and have resources they can give parents to help them.”
Stein emphasizes that even if someone displays symptoms of ADHD, that does not mean that they have it. They should seek a physician’s evaluation to confirm a diagnosis, which would enable them to get the medication and behavioral treatment they need.
The medication can take effect in parents within an hour. Meanwhile, when parents participate in the behavioral parent training courses, their kids with ADHD start showing significant improvement within about four to five weeks, according to Stein.
“With treatment, many people with ADHD do very well,” he says. “Especially if they get through formal schooling, find the right fit with their job, and if they make the right choices with their relationships, those three things can go a long way to make their ADHD fade into the background.”
Patients voice hope and relief as FDA gives third-ever drug approval for ALS
On Sept. 29, the FDA approved Relyvrio, a new drug for ALS, even though a study of 137 ALS patients did not result in “substantial evidence” that Relyvrio was effective.
At age 52, Glen Rouse suffered from arm weakness and a lot of muscle twitches. “I first thought something was wrong when I could not throw a 50-pound bag of dog food over the tailgate of my truck—something I use to do effortlessly,” said the 54-year-old resident of Anderson, California, about three hours north of San Francisco.
In August, Rouse retired as a forester for a private timber company, a job he had held for 31 years. The impetus: amyotrophic lateral sclerosis, or ALS, a progressive neuromuscular disease that is commonly known as Lou Gehrig’s disease, named after the New York Yankees’ first baseman who succumbed to it less than a month shy of his 38th birthday in 1941. ALS eventually robs an individual of the ability to talk, walk, chew, swallow and breathe.
Rouse is now dependent on ventilation through a nasal mask and uses a powerchair to get around. “I can no longer walk or use my arms very well,” he said. “I can still move my wrists and fingers. I can also transfer from my chair to the toilet if I have two of my friends help me.”
It’s “shocking” that modern medicine has very little to offer to people with this devastating condition, Rouse said. But there is hope on the horizon. Yesterday, the U.S. Food and Drug Administration approved Relyvrio, a drug made up of two parts, sodium phenylbutyrate and taurursodiol, to treat patients with ALS.
“This approval provides another important treatment option for ALS, a life-threatening disease that currently has no cure,” said Billy Dunn, director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research, in a statement. “The FDA remains committed to facilitating the development of additional ALS treatments.”
Until this point, the FDA had approved only two other medications—Riluzole (rilutek) in 1995 and Radicava (edaravone) in 2017—to extend life in patients with ALS, which typically kills within two to five years after diagnosis. That’s why earlier this week, Rouse was optimistic about the FDA’s likely approval of a controversial new drug for ALS.
When Relyvrio is taken in addition to Riluzole, it appears to slow functional decline by an additional 25 percent and extend life by another 6 to 10 months, said Richard Bedlak, director of the Duke ALS Clinic. “It is not a cure, but it is definitely a step forward.”
“The whole ALS community is extremely excited about it,” he said the day before Relyvrio’s expected approval. “We are very hopeful. We’re on pins and needles.”
A study of 137 ALS patients did not result in “substantial evidence” that Relyvrio was effective, the agency’s Peripheral and Central Nervous System Drugs Advisory Committee concluded in March. However, after some persuasion from FDA officials, patients and their families, the committee met again and decided to recommend approving the drug.
In January 2019, following an ALS diagnosis at age 58 in October the previous year, Jeff Sarnacki, of Chester, Maryland, was accepted into a trial for Relyvrio. “Because of the trial, we did experience hope and a greater sense of help than had we not had that opportunity,” said Juliet Taylor, his wife and caregiver. They both believed the drug “worked for him in giving him more time.”
In June 2019, Sarnacki chose an open-label extension, offered to patients by drug researchers after a study ends, and took the active drug until he died peacefully at home under hospice care in May 2020, five days after his 60th birthday. A retired agent with the federal Bureau of Alcohol, Tobacco, Firearms and Explosives who later worked as a security consultant, Sarnacki lived about 19 months after diagnosis, which is shorter than the typical prognosis.
His symptoms began with leg cramps in fall 2017 and foot drop in early 2018. A feeding tube was placed in 2019, as it became necessary early in his illness, Taylor said. He also took Radicava and Riluzole, the two previously approved drugs, for his ALS. “We were both incredulous that, so many years after Lou Gehrig’s own diagnosis, there were so few treatments available,” she said.
The dearth of successful treatments for ALS is “certainly not for lack of trying,” said Karen Raley Steffens, a registered nurse and ALS support services coordinator at the Les Turner ALS Foundation in Skokie, Ill. “There are thousands of researchers and scientists all over the world working tirelessly to try to develop treatments for ALS.”
Unfortunately, she added, research takes time and exorbitant amounts of funding, while bureaucratic challenges persist. The rare disease also manifests and progresses in many different ways, so many treatments are needed.
As of 2017, the Centers for Disease Control and Prevention estimated that more than 31,000 people in the U.S. live with ALS, and an average of 5,000 people are newly diagnosed every year. It is slightly more common in men than women. Most people are diagnosed between the ages of 55 and 75.
Most cases of ALS are sporadic, meaning that doctors don’t know the cause. There is about a one-year interval between symptom onset and an ALS diagnosis for most patients, so many motor neurons are lost by the time individuals can enroll in a clinical trial, said Richard Bedlack, professor of neurology and director of the Duke ALS Clinic in Durham, North Carolina.
Bedlack found the new drug, Relyvrio, to be “very promising,” which is why he testified to the FDA in favor of approval. (He’s a consultant and disease state speaker for multiple companies including Amylyx, manufacturer of Relyvrio.)
The “drug has different mechanisms of action than the currently approved treatments,” Bedlack said. He added that, when Relyvrio is taken in addition to Riluzole, it appears to slow functional decline by an additional 25 percent and extend life by another 6 to 10 months. “It is not a cure, but it is definitely a step forward.”
T. Scott Diesing, a neurohospitalist and director of general neurology at the University of Nebraska Medical Center in Omaha, said he hopes the drug is “as good as people anticipated it should be, because there are not too many options for these patients.”
"FDA went out on a limb in approving Relyvrio based on limited results from a small trial while a larger study remains in progress," said Florian P. Thomas, co-director of the ALS Center at Hackensack University Medical Center and Hackensack Meridian School of Medicine in New Jersey. "While it is definitely promising, clearly, the last word on this drug has not been spoken."
So far, Rouse's voice is holding up, but he knows the day will come when ALS will steal that and much more from him.
ALS is 100 percent fatal, with some patients dying as soon as a year after diagnosis. A few have lasted as long as 15 years, but those are the exceptions, Diesing said.
“If this drug can provide even months of additional life, or would maintain quality of life, that’s a big deal,” he noted, adding that “the patients are saying, ‘I know it’s not proven conclusively, but what do we have to lose?’ So, they would like to try it while additional studies are ongoing.” The drug has already been conditionally approved in Canada.
As his disease progresses, Rouse hopes to get a speech-to-text voice-generating computer that he can control with his eyes. So far, his voice is holding up, but he knows the day will come when ALS will steal that and much more from him. He works at I AM ALS, a patient-led community, and six of his friends have already died of the disease.
“Every time I lose a friend to ALS, I grieve and am sad but I resolve myself to keep working harder for them, myself and others,” Rouse said. “People living with ALS find great purpose in life advocating and trying to make a difference.”

