Deep Brain Stimulation for Mental Illnesses Raises Ethical Concerns
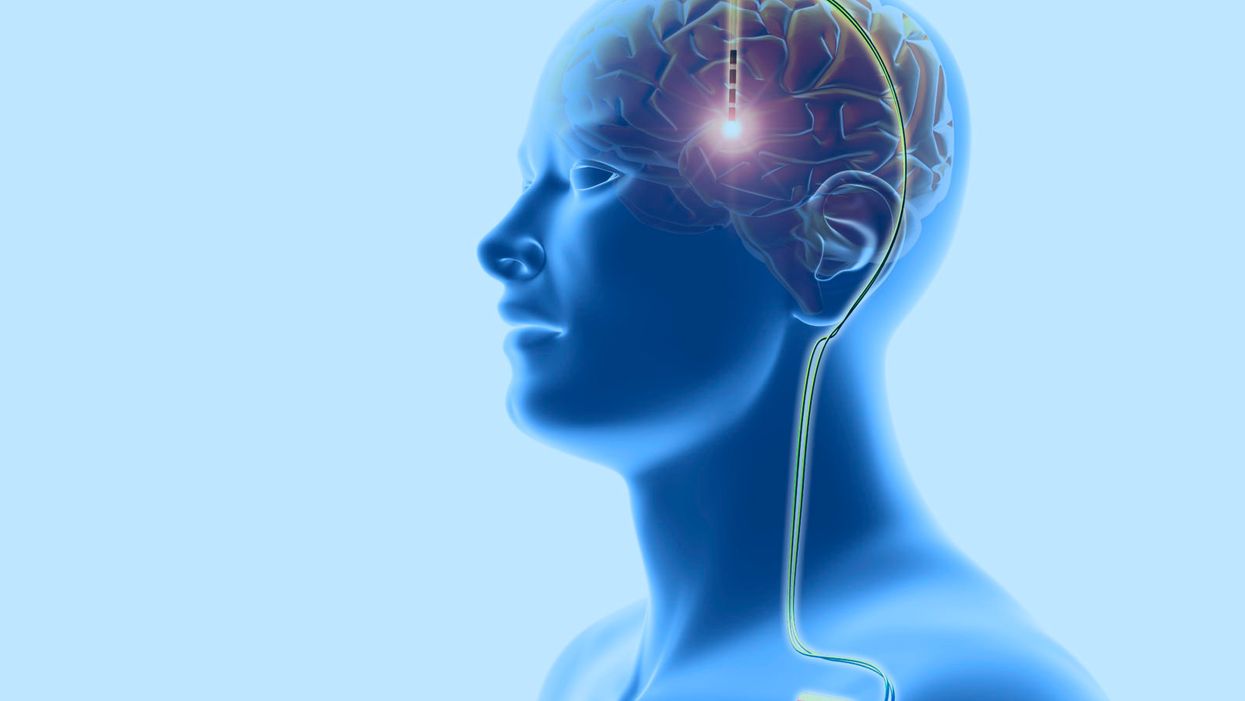
Deep brain stimulation: This neurosurgical treatment involves the implantation of electrodes in the cerebral lobes of the brain, linked through the scalp (top) to wires (down right) leading to a battery implanted below the skin. This sends electrical impulses to specific areas of the brain. DBS was developed for the treatment of Parkinson's disease, but is being investigated for use in other conditions.
Imagine that you are one of the hundreds of millions of people who suffer from depression. Medication hasn't helped you, so you're looking for another treatment option. Something powerful enough to change your mood as soon as you need a lift.
"If a participant experiences a personality change, does this change who they are or dehumanize them by altering their nature?"
Enter deep brain stimulation: a type of therapy in which one or more electrodes are inserted into your brain and connected to a surgically implanted, battery-operated medical device in your chest. This device, which is approximately the size of a stopwatch, sends electric pulses to a targeted region of your brain. The idea is to control a variety of neurological symptoms that can't be adequately managed by drugs.
Over the last twenty years, deep brain stimulation, known as DBS, has become an efficient and safe alternative for the treatment of chronic neurological diseases such as epilepsy, Parkinson's disease and neuropathic pain. According to the International Neuromodulation Society, there have been more than 80,000 deep brain stimulation implants performed around the world.
The Food and Drug Administration approved DBS as a treatment for essential tremor and Parkinson's in 1997, dystonia in 2003 and obsessive compulsive disorder in 2009. Since doctors can use drugs and treatments "off-label" (not approved by the FDA) to treat patients with any disease, DBS is now also being investigated as a treatment for chronic pain, PTSD and major depression.
And these new applications are raising profound ethical questions about individuality, personality, and even what it means to be human.
"These patients are essentially having a computer that can modify and influence emotional processing, mood and motor outputs inserted into the brain," said Gabriel Lazaro-Munoz, an assistant professor at The Center for Medical Ethics and Health Policy at Baylor College of Medicine. "These responses define us as human beings and dictate our autonomy. If a participant experiences a personality change, does this change who they are or dehumanize them by altering their nature? These are some of the questions we have to consider."
"When we are not in control of ourselves, are we ourselves?"
The U.S. government has similar concerns about DBS. The National Institutes of Health recently awarded grants to study the neuroethical issues surrounding the use of DBS in neuropsychiatric and movement disorders and appropriate consent for brain research. The grants are part of the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative. Walter Koroshetz, director of NIH's National Institute of Neurological Disorders and Stroke said, "Neuroscience is rapidly moving toward a new frontier of research on human brains that may have long-lasting and unforeseen effects. These new awards signal our commitment to research conducted in a responsible way as to anticipate all potential consequences, and to ensure that research subjects have a clear understanding of the potential benefits and risks of participating in studies."
Dr. Lazaro-Munoz's Center was awarded one of the grants to identify and evaluate the ethical, legal and social concerns with adaptive deep brain stimulation (aDBS) technologies. Adaptive DBS is a relatively new version of the technology that enables recording of brain cell activity that is then used to regulate the brain in real time. He and his team will closely observe researchers conducting aDBS studies and administering in-depth interviews to trial participants, their caregivers, and researchers, as well as individuals who declined to participate in such studies. The goal is to gain a better understanding of the ethical concerns at stake in order to guide responsible research.
Dr. Lazaro-Munoz said one of the concerns is dehumanization. "By using this technology are we compromising what makes us human? When we are not in control of ourselves, are we ourselves?" He notes that similar concerns were raised about pharmaceutical treatments for illnesses. "Both change behaviors and emotional processing. However, there is a difference. Culturally we are more used to using drugs, not implanting devices into brain and computer interfaces. Many people think of it as science fiction."
The changes in behavior due to DBS can be dramatic, perhaps none more so than with Parkinson's disease; patients may see their chronic tremors suddenly vanish.
Pills for OCD and depression take longer than DBS to see significant improvement, sometimes months. "A DBS device is either on or off. And patients and families see changes immediately," Dr. Lazaro-Munoz said. "Family members are often startled by these changes, as are the patients." He's observed that patients feel more in control with pills because they can alter and "play" with the dose or even skip a dose.
The changes in behavior due to DBS can be dramatic, perhaps none more so than with Parkinson's disease; patients may see their chronic tremors suddenly vanish, like in this must-see video.
But surgical procedures to treat motor symptoms are also increasingly being implicated as a cause of behavioral changes, both positive and negative, in patients with Parkinson's. The personality changes reported in patients who undergo DBS include hypermania, pathological gambling, hypersexuality, impulsivity and aggressiveness. One patient who suffered from OCD fell in love with the music of Johnny Cash when his brain was stimulated. On the positive side, patients report memory enhancement.
One patient who is pleased with DBS is Greg Barstead, who was diagnosed with Parkinson's in 2003, when he was the president of Colonial Penn Life Insurance Company. He also has dystonia, which affects his neck and shoulders. Barstead said that DBS has been helpful for a range of symptoms: "My shoulder is a lot less stiff and my neck hurts less. And my tremors are under control. It is not perfect, as it doesn't relieve all the Parkinson's symptoms, but it does enough of a good job that both my wife and I are very happy I had DBS."
"We are not exactly sure what part of the brain causes depression. Doctors have not identified where to implant the device."
He said he hasn't noticed any personality changes, but noted that the disease itself can cause such changes. In fact, studies have shown that it can cause many psychiatric problems including depression and hallucinations. And, approximately a third of Parkinson's patients develop dementia.
Arthur L. Caplan, founding head of the Division of Medical Ethics at NYU School of Medicine, notes that unlike psychosurgery, DBS can be turned on and off and the device can be removed. "There are less ethical concerns around treating patients with Parkinson's disease than other illnesses because surgeons know exactly where to implant the device and have many years of experience with it," he said, adding that he is concerned about using DBS for other illnesses, such as depression. "We are not exactly sure what part of the brain causes depression. Doctors have not identified where to implant the device. And I would certainly not advocate its use in patients with mild depression."
Dr. Lazaro-Munoz said of the personality changes possible with DBS, physicians need to consider how the patients were functioning without it. "Patients who are candidates for DBS typically used many medications as well as psychotherapy before opting for DBS," he explained. "To me, the question is what is the net result of using this technology? Does the patient have regrets? Are the changes in personality significant or not? Although most DBS patients report being happy they underwent the procedure, some say they don't feel like themselves after DBS. Others feel they are more like themselves, especially if there are dramatic improvements in movement problems or relief of OCD symptoms."
And then there is the question of money. The costs of DBS are covered by most insurance companies and Medicare only for FDA-approved targets like Parkinson's. Off-label uses are not covered, at least for now.
Caplan reminds people that DBS devices are manufactured by companies that are interested in making money and the average cost per treatment is around $50,000. "I am interested in seeing DBS move forward," he said. "But we must be careful and not allow industry to make it go too fast, or be used on too many people, before we know it is effective."
A company uses AI to fight muscle loss and unhealthy aging
In December 2022, a company called BioAge Labs published findings on a drug that worked to prevent muscular atrophy, or the loss of muscle strength and mass, in older people.
There’s a growing need to slow down the aging process. The world’s population is getting older and, according to one estimate, 80 million Americans will be 65 or older by 2040. As we age, the risk of many chronic diseases goes up, from cancer to heart disease to Alzheimer’s.
BioAge Labs, a company based in California, is using genetic data to help people stay healthy for longer. CEO Kristen Fortney was inspired by the genetics of people who live long lives and resist many age-related diseases. In 2015, she started BioAge to study them and develop drug therapies based on the company’s learnings.
The team works with special biobanks that have been collecting blood samples and health data from individuals for up to 45 years. Using artificial intelligence, BioAge is able to find the distinctive molecular features that distinguish those who have healthy longevity from those who don’t.
In December 2022, BioAge published findings on a drug that worked to prevent muscular atrophy, or the loss of muscle strength and mass, in older people. Much of the research on aging has been in worms and mice, but BioAge is focused on human data, Fortney says. “This boosts our chances of developing drugs that will be safe and effective in human patients.”
How it works
With assistance from AI, BioAge measures more than 100,000 molecules in each blood sample, looking at proteins, RNA and metabolites, or small molecules that are produced through chemical processes. The company uses many techniques to identify these molecules, some of which convert the molecules into charged atoms and then separating them according to their weight and charge. The resulting data is very complex, with many thousands of data points from patients being followed over the decades.
BioAge validates its targets by examining whether a pathway going awry is actually linked to the development of diseases, based on the company’s analysis of biobank health records and blood samples. The team uses AI and machine learning to identify these pathways, and the key proteins in the unhealthy pathways become their main drug targets. “The approach taken by BioAge is an excellent example of how we can harness the power of big data and advances in AI technology to identify new drugs and therapeutic targets,” says Lorna Harries, a professor of molecular genetics at the University of Exeter Medical School.
Martin Borch Jensen is the founder of Gordian Biotechnology, a company focused on using gene therapy to treat aging. He says BioAge’s use of AI allows them to speed up the process of finding promising drug candidates. However, it remains a challenge to separate pathologies from aspects of the natural aging process that aren’t necessarily bad. “Some of the changes are likely protective responses to things going wrong,” Jensen says. “Their data doesn’t…distinguish that so they’ll need to validate and be clever.”
Developing a drug for muscle loss
BioAge decided to focus on muscular atrophy because it affects many elderly people, making it difficult to perform everyday activities and increasing the risk of falls. Using the biobank samples, the team modeled different pathways that looked like they could improve muscle health. They found that people who had faster walking speeds, better grip strength and lived longer had higher levels of a protein called apelin.
Apelin is a peptide, or a small protein, that circulates in the blood. It is involved in the process by which exercise increases and preserves muscle mass. BioAge wondered if they could prevent muscular atrophy by increasing the amount of signaling in the apelin pathway. Instead of the long process of designing a drug, they decided to repurpose an existing drug made by another biotech company. This company, called Amgen, had explored the drug as a way to treat heart failure. It didn’t end up working for that purpose, but BioAge took note that the drug did seem to activate the apelin pathway.
BioAge tested its new, repurposed drug, BGE-105, and, in a phase 1 clinical trial, it protected subjects from getting muscular atrophy compared to a placebo group that didn’t receive the drug. Healthy volunteers over age 65 received infusions of the drug during 10 days spent in bed, as if they were on bed rest while recovering from an illness or injury; the elderly are especially vulnerable to muscle loss in this situation. The 11 people taking BGE-105 showed a 100 percent improvement in thigh circumference compared to 10 people taking the placebo. Ultrasound observations also revealed that the group taking the durg had enhanced muscle quality and a 73 percent increase in muscle thickness. One volunteer taking BGE-105 did have muscle loss compared to the the placebo group.
Heather Whitson, the director of the Duke University Centre for the study of aging and human development, says that, overall, the results are encouraging. “The clinical findings so far support the premise that AI can help us sort through enormous amounts of data and identify the most promising points for beneficial interventions.”
More studies are needed to find out which patients benefit the most and whether there are side effects. “I think further studies will answer more questions,” Whitson says, noting that BGE-105 was designed to enhance only one aspect of physiology associated with exercise, muscle strength. But exercise itself has many other benefits on mood, sleep, bones and glucose metabolism. “We don’t know whether BGE-105 will impact these other outcomes,” she says.
The future
BioAge is planning phase 2 trials for muscular atrophy in patients with obesity and those who have been hospitalized in an intensive care unit. Using the data from biobanks, they’ve also developed another drug, BGE-100, to treat chronic inflammation in the brain, a condition that can worsen with age and contributes to neurodegenerative diseases. The team is currently testing the drug in animals to assess its effects and find the right dose.
BioAge envisions that its drugs will have broader implications for health than treating any one specific disease. “Ultimately, we hope to pioneer a paradigm shift in healthcare, from treatment to prevention, by targeting the root causes of aging itself,” Fortney says. “We foresee a future where healthy longevity is within reach for all.”
How old fishing nets turn into chairs, car mats and Prada bags
A company called Aquafil is turning the fibers from fishing nets and old carpets into new threads for chairs, car mats, bikinis and Prada bags.
Discarded nylon fishing nets in the oceans are among the most harmful forms of plastic pollution. Every year, about 640,000 tons of fishing gear are left in our oceans and other water bodies to turn into death traps for marine life. London-based non-profit World Animal Protection estimates that entanglement in this “ghost gear” kills at least 136,000 seals, sea lions and large whales every year. Experts are challenged to estimate how many birds, turtles, fish and other species meet the same fate because the numbers are so high.
Since 2009, Giulio Bonazzi, the son of a small textile producer in northern Italy, has been working on a solution: an efficient recycling process for nylon. As CEO and chairman of a company called Aquafil, Bonazzi is turning the fibers from fishing nets – and old carpets – into new threads for car mats, Adidas bikinis, environmentally friendly carpets and Prada bags.
For Bonazzi, shifting to recycled nylon was a question of survival for the family business. His parents founded a textile company in 1959 in a garage in Verona, Italy. Fifteen years later, they started Aquafil to produce nylon for making raincoats, an enterprise that led to factories on three continents. But before the turn of the century, cheap products from Asia flooded the market and destroyed Europe’s textile production. When Bonazzi had finished his business studies and prepared to take over the family company, he wondered how he could produce nylon, which is usually produced from petrochemicals, in a way that was both successful and ecologically sustainable.
The question led him on an intellectual journey as he read influential books by activists such as world-renowned marine biologist Sylvia Earle and got to know Michael Braungart, who helped develop the Cradle-to-Cradle ethos of a circular economy. But the challenges of applying these ideologies to his family business were steep. Although fishing nets have become a mainstay of environmental fashion ads—and giants like Dupont and BASF have made breakthroughs in recycling nylon—no one had been able to scale up these efforts.
For ten years, Bonazzi tinkered with ideas for a proprietary recycling process. “It’s incredibly difficult because these products are not made to be recycled,” Bonazzi says. One complication is the variety of materials used in older carpets. “They are made to be beautiful, to last, to be useful. We vastly underestimated the difficulty when we started.”
Soon it became clear to Bonazzi that he needed to change the entire production process. He found a way to disintegrate old fibers with heat and pull new strings from the discarded fishing nets and carpets. In 2022, his company Aquafil produced more than 45,000 tons of Econyl, which is 100% recycled nylon, from discarded waste.
More than half of Aquafil’s recyclate is from used goods. According to the company, the recycling saves 90 percent of the CO2 emissions compared to the production of conventional nylon. That amounts to saving 57,100 tons of CO2 equivalents for every 10,000 tons of Econyl produced.
Bonazzi collects fishing nets from all over the world, including Norway and Chile—which have the world’s largest salmon productions—in addition to the Mediterranean, Turkey, India, Japan, Thailand, the Philippines, Pakistan, and New Zealand. He counts the government leadership of Seychelles as his most recent client; the island has prohibited ships from throwing away their fishing nets, creating the demand for a reliable recycler. With nearly 3,000 employees, Aquafil operates almost 40 collection and production sites in a dozen countries, including four collection sites for old carpets in the U.S., located in California and Arizona.
First, the dirty nets are gathered, washed and dried. Bonazzi explains that nets often have been treated with antifouling agents such as copper oxide. “We recycle the coating separately,” he says via Zoom from his home near Verona. “Copper oxide is a useful substance, why throw it away?”
Still, only a small percentage of Aquafil’s products are made from nets fished out of the ocean, so your new bikini may not have saved a strangled baby dolphin. “Generally, nylon recycling is a good idea,” says Christian Schiller, the CEO of Cirplus, the largest global marketplace for recyclates and plastic waste. “But contrary to what consumers think, people rarely go out to the ocean to collect ghost nets. Most are old, discarded nets collected on land. There’s nothing wrong with this, but I find it a tad misleading to label the final products as made from ‘ocean plastic,’ prompting consumers to think they’re helping to clean the oceans by buying these products.”
Aquafil gets most of its nets from aqua farms. Surprisingly, one of Aquafil’s biggest problems is finding enough waste. “I know, it’s hard to believe because waste is everywhere,” Bonazzi says. “But we need to find it in reliable quantity and quality.” He has invested millions in establishing reliable logistics to source the fishing nets. Then the nets get shredded into granules that can be turned into car mats for the new Hyundai Ioniq 5 or a Gucci swimsuit.
The process works similarly with carpets. In the U.S. alone, 3.5 billion pounds of carpet are discarded in landfills every year, and less than 3 percent are currently recycled. Aquafil has built a recycling plant in Phoenix to help divert 12,500 tons of carpets from the landfill every year. The carpets are shredded and deconstructed into three components: fillers such as calcium carbonate will be reused in the cement industry, synthetic fibers like polypropylene can be used for engineering plastics, and nylon. Only the pelletized nylon gets shipped back to Europe for the production of Econyl. “We ship only what’s necessary,” Bonazzi says. Nearly 50 percent of his nylon in Italy and Slovenia is produced from recyclate, and he hopes to increase the percentage to two-thirds in the next two years.
His clients include Interface, the leading world pioneer for sustainable flooring, and many other carpet producers plus more than 2500 fashion labels, including Gucci, Prada, Patagonia, Louis Vuitton, Adidas and Stella McCartney. “Stella McCartney just introduced a parka that’s made 100 percent from Econyl,” Bonazzi says. “We’re also in a lot of sportswear because Nylon is a good fabric for swimwear and for yoga clothes.” Next, he’s looking into sunglasses and chairs made with Econyl - for instance, the flexible ergonomic noho chair, designed by New Zealand company Formway.
“When I look at a landfill, I see a gold mine," Bonazzi says.
“Bonazzi decided many years ago to invest in the production of recycled nylon though industry giants halted similar plans after losing large investments,” says Anika Herrmann, vice president of the German Greentech-competitor Camm Solutions, which creates bio-based polymers from cane sugar and other ag waste. “We need role models like Bonazzi who create sustainable solutions with courage and a pioneering spirit. Like Aquafil, we count on strategic partnerships to enable fast upscaling along the entire production chain.”
Bonazzi’s recycled nylon is still five to 10 percent more expensive than conventionally produced material. However, brands are increasingly bending to the pressure of eco-conscious consumers who demand sustainable fashion. What helped Bonazzi was the recent rise of oil prices and the pressure on industries to reduce their carbon footprint. Now Bonazzi says, “When I look at a landfill, I see a gold mine.”
Ideally, the manufacturers take the products back when the client is done with it, and because the nylon can theoretically be reused nearly infinitely, the chair or bikini could be made into another chair or bikini. “But honestly,” Bonazzi half-jokes, “if someone returns a McCartney parka to me, I’ll just resell it because it’s so expensive.”
The next step: Bonazzi wants to reshape the entire nylon industry by pivoting from post-consumer nylon to plant-based nylon. In 2017, he began producing “nylon-6,” together with Genomatica in San Diego. The process uses sugar instead of petroleum. “The idea is to make the very same molecule from sugar, not from oil,” he says. The demonstration plant in Ljubljana, Slovenia, has already produced several hundred tons of nylon, and Genomatica is collaborating with Lululemon to produce plant-based yoga wear.
Bonazzi acknowledges that his company needs a few more years before the technology is ready to meet his ultimate goal, producing only recyclable products with no petrochemicals, low emissions and zero waste on an industrial scale. “Recycling is not enough,” he says. “You also need to produce the primary material in a sustainable way, with a low carbon footprint.”

