FDA, researchers work to make clinical trials more diverse

The U.S. population is becoming more diverse, but clinical trials don't reflect that, experts say. Some are focusing on recruiting minorities to participate in research.
Nestled in a predominately Hispanic neighborhood, a new mural outside Guadalupe Centers Middle School in Kansas City, Missouri imparts a powerful message: “Clinical Research Needs Representation.” The colorful portraits painted above those words feature four cancer survivors of different racial and ethnic backgrounds. Two individuals identify as Hispanic, one as African American and another as Native American.
One of the patients depicted in the mural is Kim Jones, a 51-year-old African American breast cancer survivor since 2012. She advocated for an African American friend who participated in several clinical trials for ovarian cancer. Her friend was diagnosed in an advanced stage at age 26 but lived nine more years, thanks to the trials testing new therapeutics. “They are definitely giving people a longer, extended life and a better quality of life,” said Jones, who owns a nail salon. And that’s the message the mural aims to send to the community: Clinical trials need diverse participants.
While racial and ethnic minority groups represent almost half of the U.S. population, the lack of diversity in clinical trials poses serious challenges. Limited awareness and access impede equitable representation, which is necessary to prove the safety and effectiveness of medical interventions across different groups.
A Yale University study on clinical trial diversity published last year in BMJ Medicine found that while 81 percent of trials testing the new cancer drugs approved by the U.S. Food and Drug Administration between 2012 and 2017 included women, only 23 percent included older adults and 5 percent fairly included racial and ethnic minorities. “It’s both a public health and social justice issue,” said Jennifer E. Miller, an associate professor of medicine at Yale School of Medicine. “We need to know how medicines and vaccines work for all clinically distinct groups, not just healthy young White males.” A recent JAMA Oncology editorial stresses out the need for legislation that would require diversity action plans for certain types of trials.
Ensuring meaningful representation of racial and ethnic minorities in clinical trials for regulated medical products is fundamental to public health.--FDA Commissioner Robert M. Califf.
But change is on the horizon. Last April, the FDA issued a new draft guidance encouraging industry to find ways to revamp recruitment into clinical trials. The announcement, which expanded on previous efforts, called for including more participants from underrepresented racial and ethnic segments of the population.
“The U.S. population has become increasingly diverse, and ensuring meaningful representation of racial and ethnic minorities in clinical trials for regulated medical products is fundamental to public health,” FDA commissioner Robert M. Califf, a physician, said in a statement. “Going forward, achieving greater diversity will be a key focus throughout the FDA to facilitate the development of better treatments and better ways to fight diseases that often disproportionately impact diverse communities. This guidance also further demonstrates how we support the Administration’s Cancer Moonshot goal of addressing inequities in cancer care, helping to ensure that every community in America has access to cutting-edge cancer diagnostics, therapeutics and clinical trials.”
Lola Fashoyin-Aje, associate director for Science and Policy to Address Disparities in the Oncology Center of Excellence at the FDA, said that the agency “has long held the view that clinical trial participants should reflect the clinical and demographic characteristics of the patients who will ultimately receive the drug once approved.” However, “numerous studies over many decades” have measured the extent of underrepresentation. One FDA analysis found that the proportion of White patients enrolled in U.S. clinical trials (88 percent) is much higher than their numbers in country's population. Meanwhile, the enrollment of African American and Native Hawaiian/American Indian and Alaskan Native patients is below their national numbers.
The FDA’s guidance is accelerating researchers’ efforts to be more inclusive of diverse groups in clinical trials, said Joyce Sackey, a clinical professor of medicine and associate dean at Stanford School of Medicine. Underrepresentation is “a huge issue,” she noted. Sackey is focusing on this in her role as the inaugural chief equity, diversity and inclusion officer at Stanford Medicine, which encompasses the medical school and two hospitals.
Until the early 1990s, Sackey pointed out, clinical trials were based on research that mainly included men, as investigators were concerned that women could become pregnant, which would affect the results. This has led to some unfortunate consequences, such as indications and dosages for drugs that cause more side effects in women due to biological differences. “We’ve made some progress in including women, but we have a long way to go in including people of different ethnic and racial groups,” she said.

A new mural outside Guadalupe Centers Middle School in Kansas City, Missouri, advocates for increasing diversity in clinical trials. Kim Jones, 51-year-old African American breast cancer survivor, is second on the left.
Artwork by Vania Soto. Photo by Megan Peters.
Among racial and ethnic minorities, distrust of clinical trials is deeply rooted in a history of medical racism. A prime example is the Tuskegee Study, a syphilis research experiment that started in 1932 and spanned 40 years, involving hundreds of Black men with low incomes without their informed consent. They were lured with inducements of free meals, health care and burial stipends to participate in the study undertaken by the U.S. Public Health Service and the Tuskegee Institute in Alabama.
By 1947, scientists had figured out that they could provide penicillin to help patients with syphilis, but leaders of the Tuskegee research failed to offer penicillin to their participants throughout the rest of the study, which lasted until 1972.
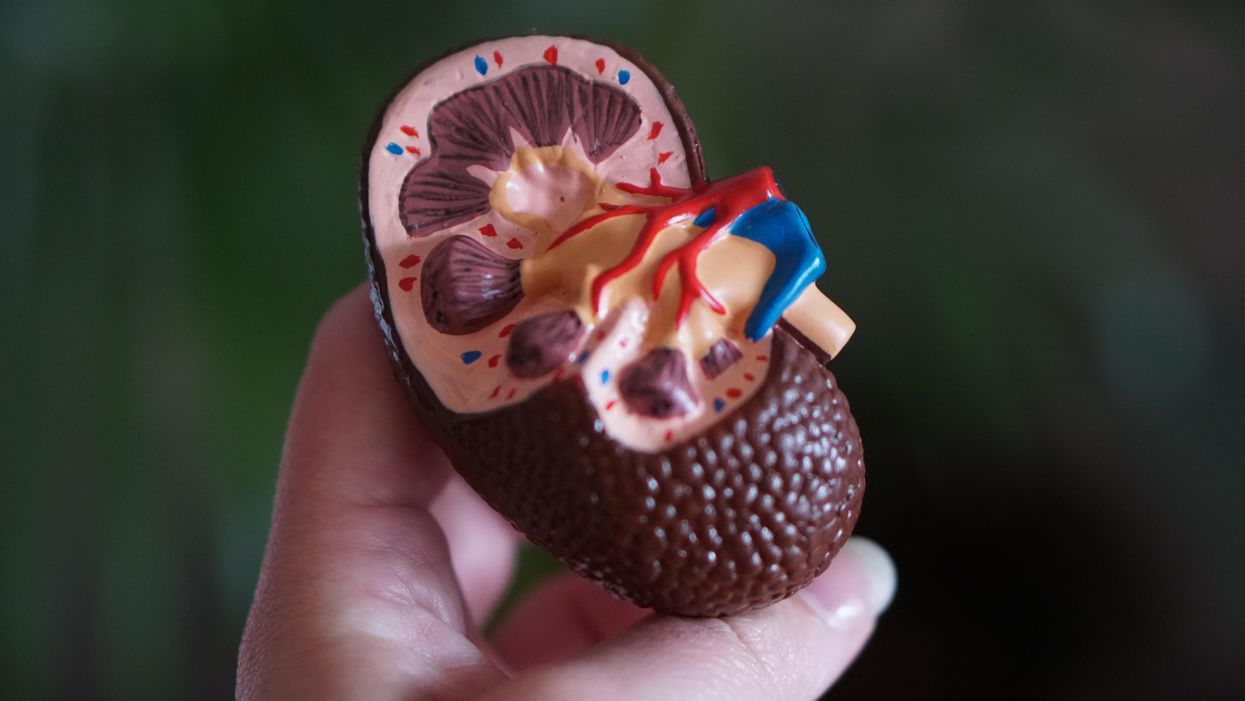
Opeyemi Olabisi, an assistant professor of medicine at Duke University Medical Center, aims to increase the participation of African Americans in clinical research. As a nephrologist and researcher, he is the principal investigator of a clinical trial focusing on the high rate of kidney disease fueled by two genetic variants of the apolipoprotein L1 (APOL1) gene in people of recent African ancestry. Individuals of this background are four times more likely to develop kidney failure than European Americans, with these two variants accounting for much of the excess risk, Olabisi noted.
The trial is part of an initiative, CARE and JUSTICE for APOL1-Mediated Kidney Disease, through which Olabisi hopes to diversify study participants. “We seek ways to engage African Americans by meeting folks in the community, providing accessible information and addressing structural hindrances that prevent them from participating in clinical trials,” Olabisi said. The researchers go to churches and community organizations to enroll people who do not visit academic medical centers, which typically lead clinical trials. Since last fall, the initiative has screened more than 250 African Americans in North Carolina for the genetic variants, he said.
Other key efforts are underway. “Breaking down barriers, including addressing access, awareness, discrimination and racism, and workforce diversity, are pivotal to increasing clinical trial participation in racial and ethnic minority groups,” said Joshua J. Joseph, assistant professor of medicine at the Ohio State University Wexner Medical Center. Along with the university’s colleges of medicine and nursing, researchers at the medical center partnered with the African American Male Wellness Agency, Genentech and Pfizer to host webinars soliciting solutions from almost 450 community members, civic representatives, health care providers, government organizations and biotechnology professionals in 25 states and five countries.
Their findings, published in February in the journal PLOS One, suggested that including incentives or compensation as part of the research budget at the institutional level may help resolve some issues that hinder racial and ethnic minorities from participating in clinical trials. Compared to other groups, more Blacks and Hispanics have jobs in service, production and transportation, the authors note. It can be difficult to get paid leave in these sectors, so employees often can’t join clinical trials during regular business hours. If more leaders of trials offer money for participating, that could make a difference.
Obstacles include geographic access, language and other communications issues, limited awareness of research options, cost and lack of trust.
Christopher Corsico, senior vice president of development at GSK, formerly GlaxoSmithKline, said the pharmaceutical company conducted a 17-year retrospective study on U.S. clinical trial diversity. “We are using epidemiology and patients most impacted by a particular disease as the foundation for all our enrollment guidance, including study diversity plans,” Corsico said. “We are also sharing our results and ideas across the pharmaceutical industry.”
Judy Sewards, vice president and head of clinical trial experience at Pfizer’s headquarters in New York, said the company has committed to achieving racially and ethnically diverse participation at or above U.S. census or disease prevalence levels (as appropriate) in all trials. “Today, barriers to clinical trial participation persist,” Sewards said. She noted that these obstacles include geographic access, language and other communications issues, limited awareness of research options, cost and lack of trust. “Addressing these challenges takes a village. All stakeholders must come together and work collaboratively to increase diversity in clinical trials.”
It takes a village indeed. Hope Krebill, executive director of the Masonic Cancer Alliance, the outreach network of the University of Kansas Cancer Center in Kansas City, which commissioned the mural, understood that well. So her team actively worked with their metaphorical “village.” “We partnered with the community to understand their concerns, knowledge and attitudes toward clinical trials and research,” said Krebill. “With that information, we created a clinical trials video and a social media campaign, and finally, the mural to encourage people to consider clinical trials as an option for care.”
Besides its encouraging imagery, the mural will also be informational. It will include a QR code that viewers can scan to find relevant clinical trials in their location, said Vania Soto, a Mexican artist who completed the rendition in late February. “I’m so honored to paint people that are survivors and are living proof that clinical trials worked for them,” she said.
Jones, the cancer survivor depicted in the mural, hopes the image will prompt people to feel more open to partaking in clinical trials. “Hopefully, it will encourage people to inquire about what they can do — how they can participate,” she said.
When Willem Johan Kolff invented dialysis, the "father" of artificial organs was just getting started.
One of the Netherlands’ most famous pieces of pop culture is “Soldier of Orange.” It’s the title of the country’s most celebrated war memoir, movie and epic stage musical, all of which detail the exploits of the nation’s resistance fighters during World War II.
Willem Johan Kolff was a member of the Dutch resistance, but he doesn’t rate a mention in the “Solider of Orange” canon. Yet his wartime toils in a rural backwater not only changed medicine, but the world.
Kolff had been a physician less than two years before Germany invaded the Netherlands in May 1940. He had been engaged in post-graduate studies at the University of Gronigen but withdrew because he refused to accommodate the demands of the Nazi occupiers. Kolff’s Jewish supervisor made an even starker choice: He committed suicide.
After his departure from the university, Kolff took a job managing a small hospital in Kampen. Located 50 miles from the heavily populated coastal region, the facility was far enough away from the prying eyes of Germans that not only could Kolff care for patients, he could hide fellow resistance fighters and even Jewish refugees in relative safety. Kolff coached many of them to feign convincing terminal illnesses so the Nazis would allow them to remain in the hospital.
Despite the demands of practicing medicine and resistance work, Kolff still found time to conduct research. He had been haunted and inspired when, not long before the Nazi invasion, one of his patients died in agony from kidney disease. Kolff wanted to find a way to save future patients.
He broke his problem down to a simple task: If he could remove 20 grams of urea from a patient’s blood in 24 hours, they would survive. He began experimenting with ways to filter blood and return it to a patient’s body. Since the war had ground all non-military manufacturing to a halt, he was mostly forced to make do with material he could find at the hospital and around Kampen. Kolff eventually built a device from a washing machine parts, juice cans, sausage casings, a valve from an old Ford automobile radiator, and even scrap from a downed German aircraft.
The world’s first dialysis machine was hardly imposing; it resembled a rotating drum for a bingo game or raffle. Yet it carried on the highly sophisticated task of moving a patient’s blood through a semi-permeable membrane (about a 50-foot length of sausage casings) into a saline solution that drew out urea while leaving the blood cells untouched.
In emigrating to the U.S. to practice medicine, Kolff's intent was twofold: Advocate for a wider adoption of dialysis, and work on new projects. He wildly succeeded at both.
Kolff began using the machine to treat patients in 1943, most of whom had lapsed into comas due to their kidney failure. But like most groundbreaking medical devices, it was not an immediate success. By the end of the war, Kolff had dialyzed more than a dozen patients, but all had died. He briefly suspended use of the device after the Allied invasion of Europe, but he continued to refine its operation and the administration of blood thinners to patients.
In September 1945, Kolff dialyzed another comatose patient, 67-year-old Sofia Maria Schafstadt. She regained consciousness after 11 hours, and would live well into the 1950s with Kolff’s assistance. Yet this triumph contained a dark irony: At the time of her treatment, Schafstadt had been imprisoned for collaborating with the Germans.
With a tattered Europe struggling to overcome the destruction of the war, Kolff and his family emigrated to the U.S. in 1950, where he began working for the Cleveland Clinic while undergoing the naturalization process so he could practice medicine in the U.S. His intent was twofold: Advocate for a wider adoption of dialysis, and work on new projects. He wildly succeeded at both.
By the mid-1950s, dialysis machines had become reliable and life-saving medical devices, and Kolff had become a U.S. citizen. About that time he invented a membrane oxygenator that could be used in heart bypass surgeries. This was a critical component of the heart-lung machine, which would make heart transplants possible and bypass surgeries routine. He also invented among the very first practical artificial hearts, which in 1957 kept a dog alive for 90 minutes.
Kolff moved to the University of Utah in 1967 to become director of its Institute for Biomedical Engineering. It was a promising time for such a move, as the first successful transplant of a donor heart to a human occurred that year. But he was interested in going a step further and creating an artificial heart for human use.
It took more than a decade of tinkering and research, but in 1982, a team of physicians and engineers led by Kolff succeeded in implanting the first artificial heart in dentist Barney Clark, whose failing health disqualified him from a heart transplant. Although Clark died in March 1983 after 112 days tethered to the device, that it kept him alive generated international headlines. While graduate student Robert Jarvik received the named credit for the heart, he was directly supervised by Kolff, whose various endeavors into artificial organ research at the University of Utah were segmented into numerous teams.
Forty years later, several artificial hearts have been approved for use by the Food and Drug Administration, although all are a “bridge” that allow patients to wait for a transplant.
Kolff continued researching and tinkering with biomedical devices – including artificial eyes and ears – until he retired in 1997 at the age of 86. When he died in 2009, the medical community acknowledged that he was not only a pioneer in biotechnology, but the “father” of artificial organs.
An image from one of TRIPP's virtual reality experiences for mental wellness. Nanea Reeves is TRIPP's CEO.
The "Making Sense of Science" podcast features interviews with leading experts about health innovations and the ethical questions they raise. The podcast is hosted by Matt Fuchs, editor of Leaps.org, the award-winning science outlet.
My guest today is Nanea Reeves, the CEO of TRIPP, a wellness platform with some big differences from meditation apps you may have tried like Calm and Headspace. TRIPP's experiences happen in virtual reality, and its realms are designed based on scientific findings about states of mindfulness. Users report feelings of awe and wonder and even mystical experiences. Nanea brings over 15 years of leadership in digital distribution, apps and video game technologies. Before co-founding TRIPP, she had several other leadership roles in tech with successful companies like textPlus and Machinima. Read her full bio below in the links section.

Nanea Reeves, CEO of TRIPP.
TRIPP
Listen to the Episode
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
This conversation coincided with National Brain Awareness Week. The topic is a little different from the Making Sense of Science podcast’s usual focus on breakthroughs in treating and preventing disease, but there’s a big overlap when it comes to breakthroughs in optimal health. Nanea’s work is at the leading edge of health, technology and the science of wellness.
With TRIPP, you might find yourself deep underwater, looking up at the sunlight shimmering on the ocean surface, or in the cosmos staring down at a planet glowing with an arresting diversity of colors. Using TRIPP for the past six months has been a window for me into the future of science-informed wellness and an overall fascinating experience, as was my conversation with Nanea.
Show notes:
Nanea and I discuss her close family members' substance addictions and her own struggle with mental illness as a teen, which led to her first meditation experiences, and much more:
- The common perception that technology is an obstacle for mental well-being, a narrative that overlooks how tech can also increase wellness when it’s designed right.
- Emerging ways of measuring meditation experiences by recording brain waves - and the shortcomings of the ‘measured self’ movement.
- Why TRIPP’s users multiplied during the stress and anxiety of the pandemic, and how TRIPP can can be used to enhance emotional states.
- Ways in which TRIPP’s visuals and targeted sound frequencies have been informed by innovative research from psychologists like Johns Hopkins’ Matthew Johnson.
- Ways to design apps and other technologies to better fulfill the true purpose of mindfulness meditation. (Hint: not simply relaxation.)
- And of course, because the topic is mental wellness and tech, I had to get Nanea's thoughts on Elon Musk, Neuralink and brain machine interfaces.
Here are links for learning more about TRIPP:
- TRIPP website: https://www.tripp.com/about/
- Nanea Reeves bio: https://www.tripp.com/team/nanea-reeves/
- Study of data collected by UK's Office for National Statistics on behavior during the pandemic, which suggests that TRIPP enhanced users' psychological and emotional mindsets: https://link.springer.com/chapter/10.1007/978-3-03...
- Research that's informed TRIPP: https://www.tripp.com/research/
- Washington Post Top Pick at CES: https://www.washingtonpost.com/technology/2019/01/...
- TRIPP's new offering, PsyAssist, to provide support for ketamine-assisted therapy: https://www.mobihealthnews.com/news/tripp-acquires...
- Randomized pilot trial involving TRIPP: https://bmjopen.bmj.com/content/bmjopen/11/4/e0441...