Elizabeth Holmes Through the Director’s Lens
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.

Elizabeth Holmes.
"The Inventor," a chronicle of Theranos's storied downfall, premiered recently on HBO. Leapsmag reached out to director Alex Gibney, whom The New York Times has called "one of America's most successful and prolific documentary filmmakers," for his perspective on Elizabeth Holmes and the world she inhabited.
Do you think Elizabeth Holmes was a charismatic sociopath from the start — or is she someone who had good intentions, over-promised, and began the lies to keep her business afloat, a "fake it till you make it" entrepreneur like Thomas Edison?
I'm not qualified to say if EH was or is a sociopath. I don't think she started Theranos as a scam whose only purpose was to make money. If she had done so, she surely would have taken more money for herself along the way. I do think that she had good intentions and that she, as you say, "began the lies to keep her business afloat." ([Reporter John] Carreyrou's book points out that those lies began early.) I think that the Edison comparison is instructive for a lot of reasons.
First, Edison was the original "fake-it-till-you-make-it" entrepreneur. That puts this kind of behavior in the mainstream of American business. By saying that, I am NOT endorsing the ethic, just the opposite. As one Enron executive mused about the mendacity there, "Was it fraud or was it bad marketing?" That gives you a sense of how baked-in the "fake it" sensibility is.
"Having a thirst for fame and a noble cause enabled her to think it was OK to lie in service of those goals."
I think EH shares one other thing with Edison, which is a huge ego coupled with a talent for storytelling as long as she is the heroic, larger-than-life main character. It's interesting that EH calls her initial device "Edison." Edison was the world's most famous "inventor," both because of the devices that came out of his shop and and for his ability for "self-invention." As Randall Stross notes in "The Wizard of Menlo Park," he was the first celebrity businessman. In addition to her "good intentions," EH was certainly motivated by fame and glory and many of her lies were in service to those goals.
Having a thirst for fame and a noble cause enabled her to think it was OK to lie in service of those goals. That doesn't excuse the lies. But those noble goals may have allowed EH to excuse them for herself or, more perniciously, to make believe that they weren't lies at all. This is where we get into scary psychological territory.
But rather than thinking of it as freakish, I think it's more productive to think of it as an exaggeration of the way we all lie to others and to ourselves. That's the point of including the Dan Ariely experiment with the dice. In that experiment, most of the subjects cheated more when they thought they were doing it for a good cause. Even more disturbing, that "good cause" allowed them to lie much more effectively because they had come to believe they weren't doing anything wrong. As it turns out, economics isn't a rational practice; it's the practice of rationalizing.
Where EH and Edison differ is that Edison had a firm grip on reality. He knew he could find a way to make the incandescent lightbulb work. There is no evidence that EH was close to making her "Edison" work. But rather than face reality (and possibly adjust her goals) she pretended that her dream was real. That kind of "over-promising" or "bold vision" is one thing when you are making a prototype in the lab. It's a far more serious matter when you are using a deeply flawed system on real patients. EH can tell herself that she had to do that (Walgreens was ready to walk away if she hadn't "gone live") or else Theranos would have run out of money.
But look at the calculation she made: she thought it was worth putting lives at risk in order to make her dream come true. Now we're getting into the realm of the sociopath. But my experience leads me to believe that -- as in the case of the Milgram experiment -- most people don't do terrible things right away, they come to crimes gradually as they become more comfortable with bigger and bigger rationalizations. At Theranos, the more valuable the company became, the bigger grew the lies.
The two whistleblowers come across as courageous heroes, going up against the powerful and intimidating company. The contrast between their youth and lack of power and the old elite backers of Theronos is staggering, and yet justice triumphed. Were the whistleblowers hesitant or afraid to appear in the film, or were they eager to share their stories?
By the time I got to them, they were willing and eager to tell their stories, once I convinced them that I would honor their testimony. In the case of Erika and Tyler, they were nudged to participate by John Carreyrou, in whom they had enormous trust.
"It's simply crazy that no one demanded to see an objective demonstration of the magic box."
Why do you think so many elite veterans of politics and venture capitalism succumbed to Holmes' narrative in the first place, without checking into the details of its technology or financials?
The reasons are all in the film. First, Channing Robertson and many of the old men on her board were clearly charmed by her and maybe attracted to her. They may have rationalized their attraction by convincing themselves it was for a good cause! Second, as Dan Ariely tells us, we all respond to stories -- more than graphs and data -- because they stir us emotionally. EH was a great storyteller. Third, the story of her as a female inventor and entrepreneur in male-dominated Silicon Valley is a tale that they wanted to invest in.
There may have been other factors. EH was very clever about the way she put together an ensemble of credibility. How could Channing Robertson, George Shultz, Henry Kissinger and Jim Mattis all be wrong? And when Walgreens put the Wellness Centers in stores, investors like Rupert Murdoch assumed that Walgreens must have done its due diligence. But they hadn't!
It's simply crazy that no one demanded to see an objective demonstration of the magic box. But that blind faith, as it turns out, is more a part of capitalism than we have been taught.
Do you think that Roger Parloff deserves any blame for the glowing Fortune story on Theranos, since he appears in the film to blame himself? Or was he just one more victim of Theranos's fraud?
He put her on the cover of Fortune so he deserves some blame for the fraud. He still blames himself. That willingness to hold himself to account shows how seriously he takes the job of a journalist. Unlike Elizabeth, Roger has the honesty and moral integrity to admit that he made a mistake. He owned up to it and published a mea culpa. That said, Roger was also a victim because Elizabeth lied to him.
Do you think investors in Silicon Valley, with their FOMO attitudes and deep pockets, are vulnerable to making the same mistake again with a shiny new startup, or has this saga been a sober reminder to do their due diligence first?
Many of the mistakes made with Theranos were the same mistakes made with Enron. We must learn to recognize that we are, by nature, trusting souls. Knowing that should lead us to a guiding slogan: "trust but verify."
The irony of Holmes dancing to "I Can't Touch This" is almost too perfect. How did you find that footage?
It was leaked to us.
"Elizabeth Holmes is now famous for her fraud. Who better to host the re-boot of 'The Apprentice.'"
Holmes is facing up to 20 years in prison for federal fraud charges, but Vanity Fair recently reported that she is seeking redemption, taking meetings with filmmakers for a possible documentary to share her "real" story. What do you think will become of Holmes in the long run?
It's usually a mistake to handicap a trial. My guess is that she will be convicted and do some prison time. But maybe she can convince jurors -- the way she convinced journalists, her board, and her investors -- that, on account of her noble intentions, she deserves to be found not guilty. "Somewhere, over the rainbow…"
After the trial, and possibly prison, I'm sure that EH will use her supporters (like Tim Draper) to find a way to use the virtual currency of her celebrity to rebrand herself and launch something new. Fitzgerald famously said that "there are no second acts in American lives." That may be the stupidest thing he ever said.
Donald Trump failed at virtually every business he ever embarked on. But he became a celebrity for being a fake businessman and used that celebrity -- and phony expertise -- to become president of the United States. Elizabeth Holmes is now famous for her fraud. Who better to host the re-boot of "The Apprentice." And then?
"You Can't Touch This!"
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
Everyone Should Hear My COVID Vaccine Experience
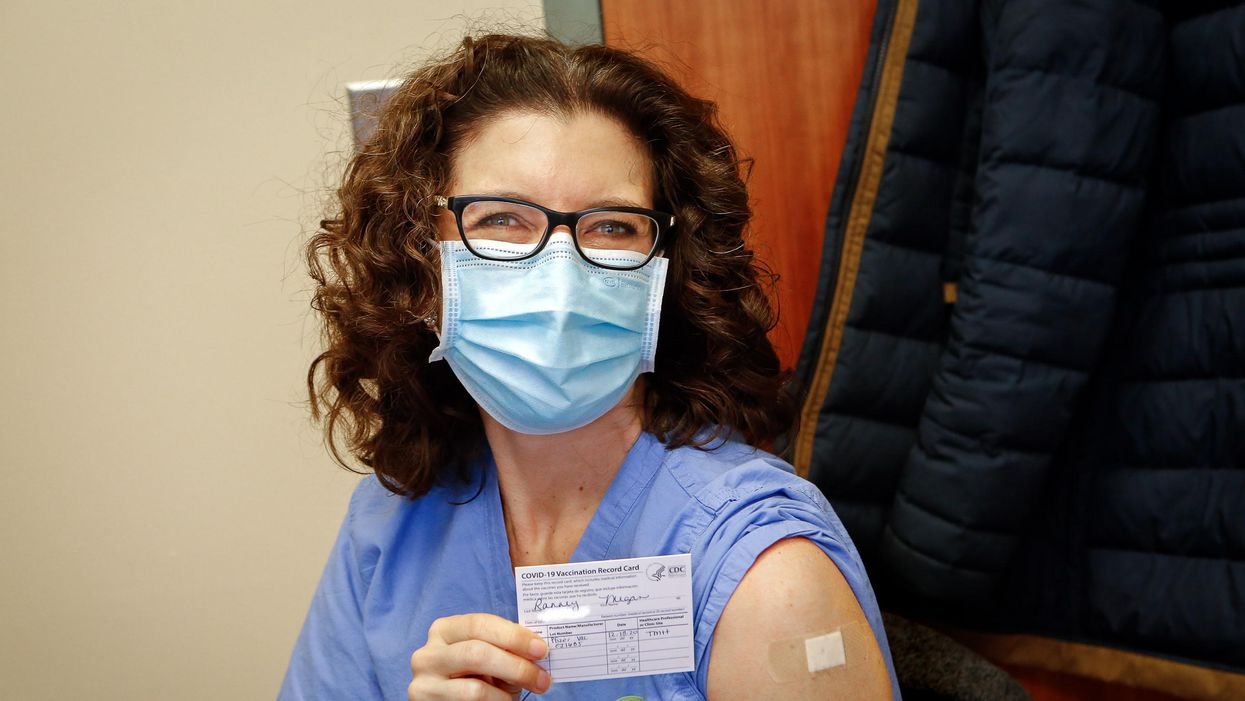
Dr. Ranney immediately after receiving her first dose of the Pfizer vaccine on December 18, 2020.
On December 18th, 2020, I received my first dose of the Pfizer mRNA vaccine against SARS-CoV-2. On January 9th, 2021, I received my second. I am now a CDC-card-carrying, fully vaccinated person.
The build-up to the first dose was momentous. I was scheduled for the first dose of the morning. Our vaccine clinic was abuzz with excitement and hope, and some media folks were there to capture the moment. A couple of fellow emergency physicians were in the same cohort of recipients as I; we exchanged virtual high-fives and took a picture of socially distanced hugs. It was, after all, the closest thing we'd had to a celebration in months.
I walked in the vaccine administration room with anticipation – it was tough to believe this moment was truly, finally here. I got a little video of my getting the shot, took my obligate vaccine selfie, waited in the observation area for 15 minutes to ensure I didn't have a reaction, and then proudly joined 1000s of fellow healthcare workers across the country in posting #ThisIsMyShot on social media. "Here we go, America!"
The first shot, though, didn't actually do all that much for me. It hurt less than a flu shot (which, by the way, doesn't hurt much). I had virtually no side effects. I also knew that it did not yet protect me. The Pfizer (and Moderna) data show very clearly that although the immune response starts to grow 10-12 days after the first shot, one doesn't reach full protection against COVID-19 until much later.
So when, two days after my first shot, I headed back to work in the emergency department, I kept wondering "Will this be the day that I get sick? Wouldn't that be ironic!" Although I never go without an N95 during patient care, it just takes one slip – scratching one's eyes, eating lunch in a break room that an infected colleague had just been in – to get ill. Ten months into this pandemic, it is so easy to get fatigued, to make a small error just one time.
Indeed, I had a few colleagues fall ill in between their first and second shots; one was hospitalized. This was not surprising, but still sad, given how close they had come to escaping infection.
Scientifically speaking, one doesn't need to feel bad to develop an immune response. Emotionally, though, I welcomed the symptoms as proof positive that I would be protected.
This time period felt a little like we had our learner's permit for driving: we were on our way to being safe, but not quite there yet.
I also watched, with dismay, our failures as a nation at timely distribution of the vaccine. On December 18th, despite the logistical snafus that many of us had started to highlight, it was still somewhat believable that we would at least distribute (if not actually administer) 20 million doses by the New Year. But by December 31, my worst fears about the feds' lack of planning had been realized. Only 14 million doses had gone to states, and fewer than 3 million had been administered. Within the public health and medical community, we began to debate how to handle the shortages and slow vaccination rates: should we change prioritization schemes? Get rid of the second dose, in contradiction to what our FDA had approved?
Let me be clear: I really, really, really wanted my second dose. It is what is supported by the data. After living this long at risk, it felt frankly unfair that I might not get fully protected. I waited with trepidation, afraid that policies would shift before I got it in my arm.
At last, my date for my second shot arrived.
This shot was a little less momentous on the outside. The vaccine clinic was much more crowded, as we were now administering first doses to more people, as well as providing the second dose to many. There were no high fives, no media, and I took no selfies. I finished my observation period without trouble (as did everyone else vaccinated the same day, as is typical for these vaccines). I walked out the door planning to spend a nice afternoon outdoors with my kids.
Within 15 minutes, though, the very common side effects – reported by 80% of people my age after the second dose – began to appear. First I got a headache (like 52% of people my age), then body aches (37%), fatigue (59%), and chills (35%). I felt "foggy", like I was fighting something. Like 45% of trial participants who had received the actual vaccine, I took acetaminophen and ibuprofen to stave off the symptoms. There is some minimal evidence from other vaccines that pre-treatment with these anti-inflammatories may reduce antibodies, but given that half of trial participants took these medications, there's no reason to make yourself suffer if you develop side effects. Forty-eight hours later, just in time for my next shift, the side effects magically cleared. Scientifically speaking, one doesn't need to feel bad to develop an immune response. Emotionally, though, I welcomed the symptoms as proof positive that I would be protected.
My reaction was truly typical. Although the media hype focuses on major negative reactions, they are – statistically speaking – tremendously rare: fewer than 11/million people who received the Pfizer vaccine, and 3/million who received the Moderna vaccine, developed anaphylaxis; of these, all were treated, and all are fine. Compare this with the fact that approximately 1200/million Americans have died of this virus. I'll choose the minor, temporary, utterly treatable side effects any day.
Now, more than 14 days after my second dose, the data says that my chance of getting really sick is, truly, infinitesimally low. I don't have to worry that each shift will put me into the hospital. I feel emotionally lighter, and a little bit like I have a secret super-power.
But I also know that we are not yet home free.
I may have my personal equivalent of Harry Potter's invisibility cloak – but we don't yet know whether it protects those around me, at all. As Dr. Fauci himself has written, while community spread is high, there is still a chance that I could be a carrier of infection to others. So I still wear my N95 at work, I still mask in public, and I still shower as soon as I get home from a shift and put my scrubs right in the washing machine to protect my husband and children. I also won't see my parents indoors until they, too, have been vaccinated.
At the end of the day, these vaccines are both amazing and life-changing, and not. My colleagues are getting sick less often, now that many of us are a week or more out from our second dose. I can do things (albeit still masked) that would simply not have been safe a month ago. These are small miracles, for which I am thankful. But like so many things in life, they would be better if shared with others. Only when my community is mostly vaccinated, will I breathe easy again.
My deepest hope is that we all have – and take - the chance to get our shots, soon. Because although the symbolism and effect of the vaccine is high, the experience itself was … not that big a deal.
New Hope for Organ Transplantation: Life Without Anti-Rejection Drugs
Kidney transplant patient Robert Waddell, center, with his wife and children after being off immunosuppresants; photo aken last summer in Perdido Key, FL. Left to right: Christian, Bailey, Rob, Karen (wife), Robby and Casey.
Rob Waddell dreaded getting a kidney transplant. He suffers from a genetic condition called polycystic kidney disease that causes the uncontrolled growth of cysts that gradually choke off kidney function. The inherited defect has haunted his family for generations, killing his great grandmother, grandmother, and numerous cousins, aunts and uncles.
But he saw how difficult it was for his mother and sister, who also suffer from this condition, to live with the side effects of the drugs they needed to take to prevent organ rejection, which can cause diabetes, high blood pressure and cancer, and even kidney failure because of their toxicity. Many of his relatives followed the same course, says Waddell: "They were all on dialysis, then a transplant and ended up usually dying from cancers caused by the medications."
When the Louisville native and father of four hit 40, his kidneys barely functioned and the only alternative was either a transplant or the slow death of dialysis. But in 2009, when Waddell heard about an experimental procedure that could eliminate the need for taking antirejection drugs, he jumped at the chance to be their first patient. Devised by scientists at the University of Louisville and Northwestern University, the innovative approach entails mixing stem cells from the live kidney donor with that of the recipient to create a hybrid immune system, known as a chimera, that would trick the immune system and prevent it from attacking the implanted kidney.
The procedure itself was done at Northwestern Memorial Hospital in Chicago, using a live kidney donated by a neighbor of Waddell's, who camped out in Chicago during his recovery. Prior to surgery, Waddell underwent a conditioning treatment that consisted of low dose radiation and chemotherapy to weaken his own immune system and make room for the infusion of stem cells.
"The low intensity chemo and radiation conditioning regimen create just enough space for the donor stem cells to gain a foothold in the bone marrow and the donor's immune system takes over," says Dr. Joseph Levanthal, the transplant surgeon who performed the operation and director of kidney and pancreas transplantation at Northwestern University Feinberg School of Medicine. "That way the recipient develops an immune system that doesn't see the donor organ as foreign."
"As a surgeon, I saw what my patients had to go through—taking 25 pills a day, dying at an early age from heart disease, or having a 35% chance of dying every year on dialysis."
A week later, Waddell had the kidney transplant. The following day, he was infused with a complex cellular cocktail that included blood-forming stem cells derived from his donor's bone marrow mixed what are called tolerance inducing facilitator cells (FCs); these cells help the foreign stem cells get established in the recipient's bone marrow.
Over the course of the following year, he was slowly weaned off of antirejection medications—a precaution in case the procedure didn't work—and remarkably, hasn't needed them since. "I felt better than I had in decades because my kidneys [had been] degrading," recalls Waddell, now 54 and a CPA for a global beverage company. And what's even better is that this new approach offers hope for one of his sons who has also inherited the disorder.
Kidney transplants are the most frequent organ transplants in the world and more than 23,000 of these procedures were done in the United States in 2019, according to the United Network for Organ Sharing. Of this, about 7,000 operations are done annually using live organ donors; the remainder use organs from people who are deceased. Right now, this revolutionary new approach—as well as a similar strategy formulated by Stanford University scientists--is in the final phase of clinical trials. Ultimately, this research may pave the way towards realizing the holy grail of organ transplantation: preventing organ rejection by creating a tolerant state in which the recipient's immune system is compatible with the donor, which would eliminate the need for a lifetime of medications.
"As a surgeon, I saw what my patients had to go through—taking 25 pills a day, dying at an early age from heart disease, or having a 35% chance of dying every year on dialysis," says Dr. Suzanne Ildstad, a transplant surgeon and director of the Institute for Cellular Therapeutics at the University of Louisville, whose discovery of facilitator cells were the basis for this therapeutic platform. Ildstad, who has spent more than two decades searching for a better way, says, "This is something I have worked for my entire life."
The Louisville group uses a combination of chemo and radiation to replace the recipient's immune and blood forming cells with that of the donor. In contrast, the Stanford protocol involves harvesting the donor's blood stem cells and T-cells, which are the foot soldiers of the immune system that fight off infections and would normally orchestrate the rejection of the transplanted organ. Their transplant recipients undergo a milder form of "conditioning" that only radiates discrete parts of the body and selectively targets the recipient's T-cells, creating room for both sets of T-cells, a strategy these researchers believe has a better safety profile and less of a chance of rejection.
"We try to achieve immune tolerance by a true chimerism," says Dr. Samuel Strober, a professor of medicine for immunology and rheumatology at Stanford University and a leader of this research team. "The recipients immune system cells are maintained but mixed in the blood with that of the donor."
Studies suggest both approaches work. In a 2018 clinical trial conducted by Talaris Therapeutics, a Louisville-based biotech founded by Ildstad, 26 of 37 (70%) of the live donor kidney transplant recipients no longer need immunosuppressants. Last fall, Talaris began the final phase of clinical tests that will eventually encompass more than 120 such patients.
The Stanford group's cell-based immunotherapy, which is called MDR-101 and is sponsored by the South San Francisco biotech, Medeor Therapeutics, has had similar results in patients who received organs from live donors who were either well matched, such as one from siblings, meaning they were immunologically identical, or partially matched; Talaris uses unrelated donors where there is only a partial match.
In their 2020 clinical trial of 51 patients, 29 were fully matched and 22 were a partial match; 22 of the fully matched recipients didn't need antirejection drugs and ten of the partial matches were able to stop taking some of these medications without rejection. "With our fully matched, roughly 80% have been completely off drugs up to 14 years later," says Strober, "and reducing the number of drugs from three to one [in the partial matches] means you have far fewer side effects. The goal is to get them off of all drugs."
But these protocols are limited to a small number of patients—living donor kidney recipients. As a consequence, both teams are experimenting with ways to broaden their approach so they can use cadaver organs from deceased donors, with human tests planned in the coming year. Here's how that would work: after the other organs are removed from a deceased donor, stem cells are harvested from the donor's vertebrae in the spinal column and then frozen for storage.
"We do the transplant and give the patient a chance to recover and maintain them on drugs," says Ildstad. "Then we do the tolerance conditioning at a later stage."
If this strategy is successful, it would be a genuine game changer, and open the door to using these protocols for transplanting other cadaver organs, including the heart, lungs and liver. While the overall procedure is complex and costly, in the long run it's less expensive than repeated transplant surgeries, the cost of medications and hospitalizations for complications caused by the drugs, or thrice weekly dialysis treatments, says Ildstad.
And she adds, you can't put a price tag on the vast improvement in quality of life.