Fetuses can save their mothers' lives
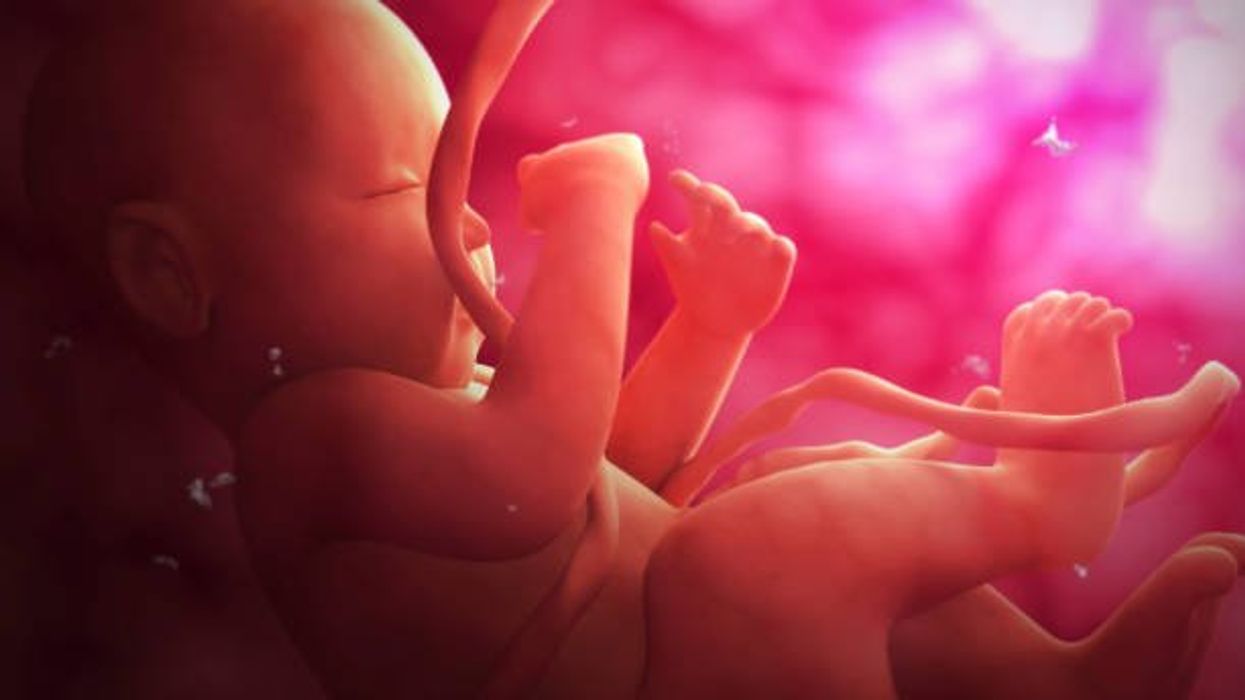
Stem cells from a fetus can travel to the heart and regenerate the muscle, essentially saving a mother’s life.
Story by Big Think
In rare cases, a woman’s heart can start to fail in the months before or after giving birth. The all-important muscle weakens as its chambers enlarge, reducing the amount of blood pumped with each beat. Peripartum cardiomyopathy can threaten the lives of both mother and child. Viral illness, nutritional deficiency, the bodily stress of pregnancy, or an abnormal immune response could all play a role, but the causes aren’t concretely known.
If there is a silver lining to peripartum cardiomyopathy, it’s that it is perhaps the most survivable form of heart failure. A remarkable 50% of women recover spontaneously. And there’s an even more remarkable explanation for that glowing statistic: The fetus‘ stem cells migrate to the heart and regenerate the beleaguered muscle. In essence, the developing or recently born child saves its mother’s life.
Saving mama
While this process has not been observed directly in humans, it has been witnessed in mice. In a 2015 study, researchers tracked stem cells from fetal mice as they traveled to mothers’ damaged cardiac cells and integrated themselves into hearts.
Evolutionarily, this function makes sense: It is in the fetus’ best interest that its mother remains healthy.
Scientists also have spotted cells from the fetus within the hearts of human mothers, as well as countless other places inside the body, including the skin, spleen, liver, brain, lung, kidney, thyroid, lymph nodes, salivary glands, gallbladder, and intestine. These cells essentially get everywhere. While most are eliminated by the immune system during pregnancy, some can persist for an incredibly long time — up to three decades after childbirth.
This integration of the fetus’ cells into the mother’s body has been given a name: fetal microchimerism. The process appears to start between the fourth and sixth week of gestation in humans. Scientists are actively trying to suss out its purpose. Fetal stem cells, which can differentiate into all sorts of specialized cells, appear to target areas of injury. So their role in healing seems apparent. Evolutionarily, this function makes sense: It is in the fetus’ best interest that its mother remains healthy.
Sending cells into the mother’s body may also prime her immune system to grow more tolerant of the developing fetus. Successful pregnancy requires that the immune system not see the fetus as an interloper and thus dispatch cells to attack it.
Fetal microchimerism
But fetal microchimerism might not be entirely beneficial. Greater concentrations of the cells have been associated with various autoimmune diseases such as lupus, Sjogren’s syndrome, and even multiple sclerosis. After all, they are foreign cells living in the mother’s body, so it’s possible that they might trigger subtle, yet constant inflammation. Fetal cells also have been linked to cancer, although it isn’t clear whether they abet or hinder the disease.
A team of Spanish scientists summarized the apparent give and take of fetal microchimerism in a 2022 review article. “On the one hand, fetal microchimerism could be a source of progenitor cells with a beneficial effect on the mother’s health by intervening in tissue repair, angiogenesis, or neurogenesis. On the other hand, fetal microchimerism might have a detrimental function by activating the immune response and contributing to autoimmune diseases,” they wrote.
Regardless of a fetus’ cells net effect, their existence alone is intriguing. In a paper published earlier this year, University of London biologist Francisco Úbeda and University of Western Ontario mathematical biologist Geoff Wild noted that these cells might very well persist within mothers for life.
“Therefore, throughout their reproductive lives, mothers accumulate fetal cells from each of their past pregnancies including those resulting in miscarriages. Furthermore, mothers inherit, from their own mothers, a pool of cells contributed by all fetuses carried by their mothers, often referred to as grandmaternal microchimerism.”
So every mother may carry within her literal pieces of her ancestors.

This article originally appeared on Big Think, home of the brightest minds and biggest ideas of all time.
A robot server, controlled remotely by a disabled worker, delivers drinks to patrons at the DAWN cafe in Tokyo.
A sleek, four-foot tall white robot glides across a cafe storefront in Tokyo’s Nihonbashi district, holding a two-tiered serving tray full of tea sandwiches and pastries. The cafe’s patrons smile and say thanks as they take the tray—but it’s not the robot they’re thanking. Instead, the patrons are talking to the person controlling the robot—a restaurant employee who operates the avatar from the comfort of their home.

It’s a typical scene at DAWN, short for Diverse Avatar Working Network—a cafe that launched in Tokyo six years ago as an experimental pop-up and quickly became an overnight success. Today, the cafe is a permanent fixture in Nihonbashi, staffing roughly 60 remote workers who control the robots remotely and communicate to customers via a built-in microphone.
More than just a creative idea, however, DAWN is being hailed as a life-changing opportunity. The workers who control the robots remotely (known as “pilots”) all have disabilities that limit their ability to move around freely and travel outside their homes. Worldwide, an estimated 16 percent of the global population lives with a significant disability—and according to the World Health Organization, these disabilities give rise to other problems, such as exclusion from education, unemployment, and poverty.
These are all problems that Kentaro Yoshifuji, founder and CEO of Ory Laboratory, which supplies the robot servers at DAWN, is looking to correct. Yoshifuji, who was bedridden for several years in high school due to an undisclosed health problem, launched the company to help enable people who are house-bound or bedridden to more fully participate in society, as well as end the loneliness, isolation, and feelings of worthlessness that can sometimes go hand-in-hand with being disabled.
“It’s heartbreaking to think that [people with disabilities] feel they are a burden to society, or that they fear their families suffer by caring for them,” said Yoshifuji in an interview in 2020. “We are dedicating ourselves to providing workable, technology-based solutions. That is our purpose.”
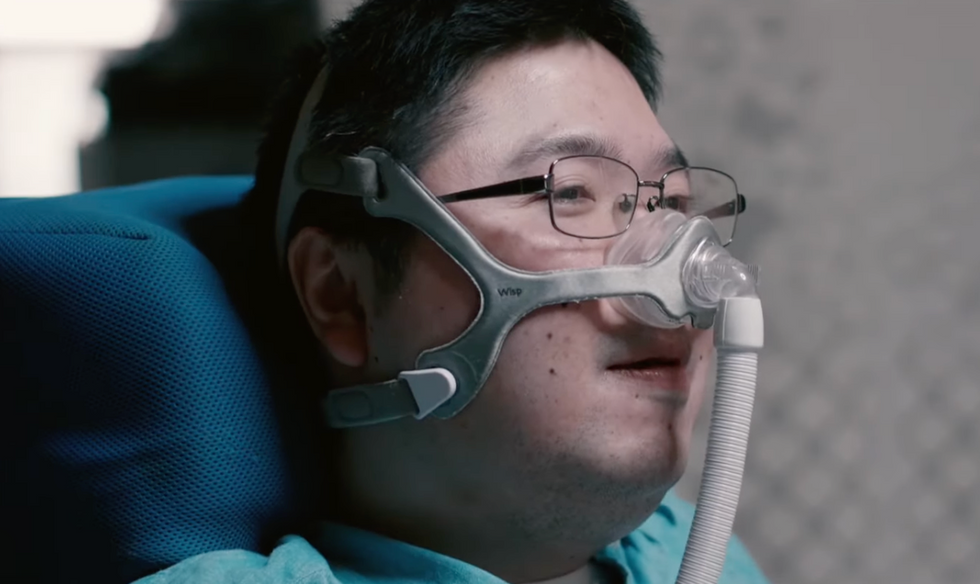
Shota, Kuwahara, a DAWN employee with muscular dystrophy, agrees. "There are many difficulties in my daily life, but I believe my life has a purpose and is not being wasted," he says. "Being useful, able to help other people, even feeling needed by others, is so motivational."
A woman receives a mammogram, which can detect the presence of tumors in a patient's breast.
When a patient is diagnosed with early-stage breast cancer, having surgery to remove the tumor is considered the standard of care. But what happens when a patient can’t have surgery?
Whether it’s due to high blood pressure, advanced age, heart issues, or other reasons, some breast cancer patients don’t qualify for a lumpectomy—one of the most common treatment options for early-stage breast cancer. A lumpectomy surgically removes the tumor while keeping the patient’s breast intact, while a mastectomy removes the entire breast and nearby lymph nodes.
Fortunately, a new technique called cryoablation is now available for breast cancer patients who either aren’t candidates for surgery or don’t feel comfortable undergoing a surgical procedure. With cryoablation, doctors use an ultrasound or CT scan to locate any tumors inside the patient’s breast. They then insert small, needle-like probes into the patient's breast which create an “ice ball” that surrounds the tumor and kills the cancer cells.
Cryoablation has been used for decades to treat cancers of the kidneys and liver—but only in the past few years have doctors been able to use the procedure to treat breast cancer patients. And while clinical trials have shown that cryoablation works for tumors smaller than 1.5 centimeters, a recent clinical trial at Memorial Sloan Kettering Cancer Center in New York has shown that it can work for larger tumors, too.
In this study, doctors performed cryoablation on patients whose tumors were, on average, 2.5 centimeters. The cryoablation procedure lasted for about 30 minutes, and patients were able to go home on the same day following treatment. Doctors then followed up with the patients after 16 months. In the follow-up, doctors found the recurrence rate for tumors after using cryoablation was only 10 percent.
For patients who don’t qualify for surgery, radiation and hormonal therapy is typically used to treat tumors. However, said Yolanda Brice, M.D., an interventional radiologist at Memorial Sloan Kettering Cancer Center, “when treated with only radiation and hormonal therapy, the tumors will eventually return.” Cryotherapy, Brice said, could be a more effective way to treat cancer for patients who can’t have surgery.
“The fact that we only saw a 10 percent recurrence rate in our study is incredibly promising,” she said.

