Genetically Sequencing Healthy Babies Yielded Surprising Results

A newborn and mother in the hospital - first touch. (© martin81/Shutterstock)
Today in Melrose, Massachusetts, Cora Stetson is the picture of good health, a bubbly precocious 2-year-old. But Cora has two separate mutations in the gene that produces a critical enzyme called biotinidase and her body produces only 40 percent of the normal levels of that enzyme.
In the last few years, the dream of predicting and preventing diseases through genomics, starting in childhood, is finally within reach.
That's enough to pass conventional newborn (heelstick) screening, but may not be enough for normal brain development, putting baby Cora at risk for seizures and cognitive impairment. But thanks to an experimental study in which Cora's DNA was sequenced after birth, this condition was discovered and she is being treated with a safe and inexpensive vitamin supplement.
Stories like these are beginning to emerge from the BabySeq Project, the first clinical trial in the world to systematically sequence healthy newborn infants. This trial was led by my research group with funding from the National Institutes of Health. While still controversial, it is pointing the way to a future in which adults, or even newborns, can receive comprehensive genetic analysis in order to determine their risk of future disease and enable opportunities to prevent them.
Some believe that medicine is still not ready for genomic population screening, but others feel it is long overdue. After all, the sequencing of the Human Genome Project was completed in 2003, and with this milestone, it became feasible to sequence and interpret the genome of any human being. The costs have come down dramatically since then; an entire human genome can now be sequenced for about $800, although the costs of bioinformatic and medical interpretation can add another $200 to $2000 more, depending upon the number of genes interrogated and the sophistication of the interpretive effort.

Two-year-old Cora Stetson, whose DNA sequencing after birth identified a potentially dangerous genetic mutation in time for her to receive preventive treatment.
(Photo courtesy of Robert Green)
The ability to sequence the human genome yielded extraordinary benefits in scientific discovery, disease diagnosis, and targeted cancer treatment. But the ability of genomes to detect health risks in advance, to actually predict the medical future of an individual, has been mired in controversy and slow to manifest. In particular, the oft-cited vision that healthy infants could be genetically tested at birth in order to predict and prevent the diseases they would encounter, has proven to be far tougher to implement than anyone anticipated.
But in the last few years, the dream of predicting and preventing diseases through genomics, starting in childhood, is finally within reach. Why did it take so long? And what remains to be done?
Great Expectations
Part of the problem was the unrealistic expectations that had been building for years in advance of the genomic science itself. For example, the 1997 film Gattaca portrayed a near future in which the lifetime risk of disease was readily predicted the moment an infant is born. In the fanfare that accompanied the completion of the Human Genome Project, the notion of predicting and preventing future disease in an individual became a powerful meme that was used to inspire investment and public support for genomic research long before the tools were in place to make it happen.
Another part of the problem was the success of state-mandated newborn screening programs that began in the 1960's with biochemical tests of the "heel-stick" for babies with metabolic disorders. These programs have worked beautifully, costing only a few dollars per baby and saving thousands of infants from death and severe cognitive impairment. It seemed only logical that a new technology like genome sequencing would add power and promise to such programs. But instead of embracing the notion of newborn sequencing, newborn screening laboratories have thus far rejected the entire idea as too expensive, too ambiguous, and too threatening to the comfortable constituency that they had built within the public health framework.
"What can you find when you look as deeply as possible into the medical genomes of healthy individuals?"
Creating the Evidence Base for Preventive Genomics
Despite a number of obstacles, there are researchers who are exploring how to achieve the original vision of genomic testing as a tool for disease prediction and prevention. For example, in our NIH-funded MedSeq Project, we were the first to ask the question: "What can you find when you look as deeply as possible into the medical genomes of healthy individuals?"
Most people do not understand that genetic information comes in four separate categories: 1) dominant mutations putting the individual at risk for rare conditions like familial forms of heart disease or cancer, (2) recessive mutations putting the individual's children at risk for rare conditions like cystic fibrosis or PKU, (3) variants across the genome that can be tallied to construct polygenic risk scores for common conditions like heart disease or type 2 diabetes, and (4) variants that can influence drug metabolism or predict drug side effects such as the muscle pain that occasionally occurs with statin use.
The technological and analytical challenges of our study were formidable, because we decided to systematically interrogate over 5000 disease-associated genes and report results in all four categories of genetic information directly to the primary care physicians for each of our volunteers. We enrolled 200 adults and found that everyone who was sequenced had medically relevant polygenic and pharmacogenomic results, over 90 percent carried recessive mutations that could have been important to reproduction, and an extraordinary 14.5 percent carried dominant mutations for rare genetic conditions.
A few years later we launched the BabySeq Project. In this study, we restricted the number of genes to include only those with child/adolescent onset that could benefit medically from early warning, and even so, we found 9.4 percent carried dominant mutations for rare conditions.
At first, our interpretation around the high proportion of apparently healthy individuals with dominant mutations for rare genetic conditions was simple – that these conditions had lower "penetrance" than anticipated; in other words, only a small proportion of those who carried the dominant mutation would get the disease. If this interpretation were to hold, then genetic risk information might be far less useful than we had hoped.
Suddenly the information available in the genome of even an apparently healthy individual is looking more robust, and the prospect of preventive genomics is looking feasible.
But then we circled back with each adult or infant in order to examine and test them for any possible features of the rare disease in question. When we did this, we were surprised to see that in over a quarter of those carrying such mutations, there were already subtle signs of the disease in question that had not even been suspected! Now our interpretation was different. We now believe that genetic risk may be responsible for subclinical disease in a much higher proportion of people than has ever been suspected!
Meanwhile, colleagues of ours have been demonstrating that detailed analysis of polygenic risk scores can identify individuals at high risk for common conditions like heart disease. So adding up the medically relevant results in any given genome, we start to see that you can learn your risks for a rare monogenic condition, a common polygenic condition, a bad effect from a drug you might take in the future, or for having a child with a devastating recessive condition. Suddenly the information available in the genome of even an apparently healthy individual is looking more robust, and the prospect of preventive genomics is looking feasible.
Preventive Genomics Arrives in Clinical Medicine
There is still considerable evidence to gather before we can recommend genomic screening for the entire population. For example, it is important to make sure that families who learn about such risks do not suffer harms or waste resources from excessive medical attention. And many doctors don't yet have guidance on how to use such information with their patients. But our research is convincing many people that preventive genomics is coming and that it will save lives.
In fact, we recently launched a Preventive Genomics Clinic at Brigham and Women's Hospital where information-seeking adults can obtain predictive genomic testing with the highest quality interpretation and medical context, and be coached over time in light of their disease risks toward a healthier outcome. Insurance doesn't yet cover such testing, so patients must pay out of pocket for now, but they can choose from a menu of genetic screening tests, all of which are more comprehensive than consumer-facing products. Genetic counseling is available but optional. So far, this service is for adults only, but sequencing for children will surely follow soon.
As the costs of sequencing and other Omics technologies continue to decline, we will see both responsible and irresponsible marketing of genetic testing, and we will need to guard against unscientific claims. But at the same time, we must be far more imaginative and fast moving in mainstream medicine than we have been to date in order to claim the emerging benefits of preventive genomics where it is now clear that suffering can be averted, and lives can be saved. The future has arrived if we are bold enough to grasp it.
Funding and Disclosures:
Dr. Green's research is supported by the National Institutes of Health, the Department of Defense and through donations to The Franca Sozzani Fund for Preventive Genomics. Dr. Green receives compensation for advising the following companies: AIA, Applied Therapeutics, Helix, Ohana, OptraHealth, Prudential, Verily and Veritas; and is co-founder and advisor to Genome Medical, Inc, a technology and services company providing genetics expertise to patients, providers, employers and care systems.
Here's What It Looks Like to Seek Therapy for Climate Change Anxiety
Treatment for climate change anxiety looks different from treating generalized anxiety in that the concerns have a legitimate basis, therapists say.
Three months after Gretchen bought a house in Grass Valley, California, the most destructive and fatal wildfire in the state's history ravaged the towns about 40 miles northwest of her.
"For a long time, I kept on having this vision of what my town will look like if one of those firestorms happens, and I felt like I needed to work on that."
The Camp Fire of November 2018 was noteworthy not just because of its damaging scale but because of what started it all: a spark from a faulty transmission line owned by the Pacific Gas & Electric Company, which services nearly two-thirds of California.
PG&E reacted by announcing almost a year later that in advance of days with a high fire risk, it would proactively institute power outages in 17 counties throughout the northern part of the state, including the one where Gretchen lives. The binary options seemed to be: cause another fire or intermittently plunge tens of thousands of people into literal and figurative darkness, impacting emergency services, health, food, internet, gas, and any other electrified necessity or convenience of modern life.
This summer, in between the end of the Camp Fire and the beginning of the blackouts, Gretchen, who asked to keep her last name private, decided it was time to seek counseling for climate-related anxiety.
"That was a very traumatic experience to go through," Gretchen, 39, says, describing what it was like to have recently settled in this increasingly fire-prone part of her home state, and later witnessing a colleague flee California altogether after his own home burned down and he couldn't afford to stay. "For a long time, I kept on having this vision of what my town will look like if one of those firestorms happens, and I felt like I needed to work on that."
While research on climate anxiety—or, more broadly, the effects of climate change on mental health—has been slowly but surely piling up, the actual experience of diagnosing and treating it is less well-documented in both media and academia. An ongoing Yale University study of American perceptions of climate change shows an increasing proportion of concern: In 2018, 29 percent of 1,114 survey respondents said they were "very worried" about climate change, up from 16 percent in 2008. But there are no parallel large-scale studies of whether a similar proportion of people are in therapy for climate change-related mental health issues.
That might be because many would-be clients don't yet realize that this is a valid concern for which to seek out professional support. It could also be because there are no definitive or unifying resources for therapists who are counseling people on the topic. Climate anxiety is notably absent by name from the Diagnostic and Statistical Manual of Mental Disorders (DSM), the psychological gospel for everyone from clinicians to lawmakers. The manual was last updated in 2012 (and published in 2013), just when the first documents of climate anxiety were beginning to crop up.
A small 2013 study surveyed college students in the U.S. and Europe to try and answer the question: Is habitually worrying about the environment a mental health concern if it's a response to a real threat? The study concluded: "...those who habitually worry about the ecology are not only lacking in any psychopathology, but demonstrate a constructive and adaptive response to a serious problem." In other words, worrying about a concrete external concern like the state of the environment is on a different plane than habitually worrying about an internal concern, like feelings of inadequacy. Therapy may still help with the former, but the diagnostic framework could ultimately look different than what is typically used in generalized anxiety.
For now, the best resource for therapists counseling patients battling what is sometimes dubbed "ecoanxiety" is a 70-page booklet called "Mental Health and Our Changing Climate: Impacts, Implications, and Guidance," whose publication was co-sponsored by the American Psychological Association, which publishes the DSM. It's been through two editions already, the first in 2014 and the second in 2017.
"It's not clear to me that [climate anxiety] would merit its own diagnosis, at least at this point," says Susan Clayton, who was the lead author on the 2017 edition and who studies this area at The College of Wooster, but doesn't counsel people directly. However, she says, "I do think that there are some differences [from generalized anxiety], and one of the important differences is, of course, that there's some realism here."
Clayton says that group therapy may be a particularly useful way to affirm for people that they're not the only one experiencing climate anxiety, especially in communities where it might be taboo to not only affirm the existence of climate change but to be openly affected by it.
On drawing therapeutic inspiration from historical examples of other global dangers—such as the widespread fear of nuclear threat during the Cold War—Clayton says: "That was such a different time and they were thinking differently about mental health, but I think in many ways the fear is very similar. It's not like worrying about your finances, it's worrying about the end of the world. So that sort of existential component, and the fact that it's shared, both are very similar here."
There are precedents that therapists can refer to for guidance on helping clients managing climate anxiety, like the approaches used to support people dealing with a terminal illness or battling systemic racism. Such treatments need to stay rooted in the reality of the trigger.
"You don't want to say to them, 'That's not a real thing,'" Clayton explains. "So I think of [climate anxiety] like that. It does mean that the therapeutic focus is not going to be on trying to get people to be reasonable," which is to say that their anxiety is not inherently unreasonable.
"I think it is important to recognize that the anxieties have a legitimate basis," she adds.
"I feel more comfortable now being prepared, being prudent, but not dwelling on it all the time."
Gretchen's reality is now one of adapting to living an off-the-grid lifestyle that she didn't intentionally sign up for. She puts gas in her car in advance of blackouts, and waits to see week-by-week if the school where she teaches second and third grade, in the foothills of Tahoe National Park, will be closed. Her union has yet to figure out how this stop-and-go schedule will affect her salary; she has to keep rescheduling parent-teacher conferences; and she no longer knows when the last day of school will be—existing summer plans for her personal life be damned. Even her interview for this story was affected by this instability.
While trying to schedule a time to talk, she wrote, "Speaking of climate change, I may not have work the rest of the week due to PG&E power outages. If so I will have a very flexible schedule." Later, she suddenly had to decline. "As it turns out, the power's not going out. I will be at work."
In therapy sessions, she works with her counselor to focus on preparedness, where possible, and to specifically frame that preparedness as a source of regaining some of the stability she's lost rather than a sign of imminent trouble. That nuance became necessary after a training at work had the opposite effect.
"We've gone through scenarios [where] if a firestorm happens and we don't have time to evacuate, we have to gather all the children into the cafeteria and fend off the flames ourselves with help from the fire department, and keep them alive if we can't get out in time," she says. "After that day, or that training, that really scared me."
Her therapist uses a type of psychotherapy called eye movement desensitization and reprocessing (EMDR) to help Gretchen move away from traumatizing images, such as picturing her town on fire, while emphasizing what it is that she can control, such as making sure her car has a full tank, in case she needs to evacuate. EMDR has been shown to help people with post-traumatic stress disorder (PTSD) and the World Health Organization offers practice guidelines around it.
"I feel more comfortable now being prepared, being prudent, but not dwelling on it all the time," she says. "I feel a little less heightened anxiety and have stopped replaying [those images] in my mind."
Overall, the type of support Gretchen receives is based on pre-existing tools for managing other well-established mental health burdens like PTSD and generalized anxiety. Although no definitive, new practices have specifically emerged around climate anxiety on a comprehensive scale yet, Gretchen says she was nonetheless met with compassion when she first approached a therapist about the topical source of her anxiety, and doesn't feel that her care is lacking in any way.
"I don't know enough to know whether or how it should become its own diagnosis, but I feel like it's something that is still evolving. Down the road, as we see more populations having to move, more refugees, more real effects, that might change," she says. "For me, using the old tools in a new way has been effective at this point."
Gretchen hasn't yet explored with her therapist the more existential worries that climate change dredges up for her—worries about whether or not to have children, and if it was a mistake to settle down in Grass Valley. She's only been in therapy for her climate anxiety since the summer (although she has intermittently sought out professional mental health support for other reasons over the last eight years), and it will take time to get to these bigger issues, she says. She's not sure yet whether that part of her counseling will look different than what's she's done so far.
But she does wonder about the overall usefulness of pathologizing what, as Clayton said, are legitimate anxieties. She has the same question when it comes to providing mental health support for her students, many of whom live in poverty.
"Is it just putting a bandaid on something that is unfixable, or is unfair?" she ponders. But de-escalating the psychological toll that climate change can have on people is crucial to giving them back the energy to deal with the problem itself, not just their reaction to the problem. Clayton believes that engaging in climate activism can provide solace for the people who do have that energy.
"This is a social issue, and there's obviously lots and lots of climate activism," she says. "You might not be comfortable being politically active, but I think getting involved in some way, and addressing the issue, would help people feel much more empowered, and would help with the experience of climate anxiety."
"Remember that nature is not just a source of anxiety, it's also a source of replenishment and restoration."
As far as what shape this personal involvement takes, an increasingly vocal movement of people is calling for a refocus. They say the onus of reversing, or at least stymying, the situation should fall on the big businesses and governments that have been too slow to act, not on individual consumer actions, like buying sustainably made clothes, divesting from the meat and dairy industry, or driving an electric car.
But outside of formal therapy and even activism, however that looks, Clayton has another suggestion for combating climate anxiety, and it's one that is surprising in its simplicity: Go outside, and take stock of that which boldly continues to exist.
"People who are anxious about climate change, it's partly about the survival of the species, but it's partly about the sense that, 'Something I care about is being destroyed,'" she says. "Remember that nature is not just a source of anxiety, it's also a source of replenishment and restoration."
Three Big Biotech Ideas to Watch in 2020—And Beyond
Body-on-a-chip, prime editing, and gut microbes all are poised to make a big impact in 2020.
1. Happening Now: Body-on-a-Chip Technology Is Enabling Safer Drug Trials and Better Cancer Research
Researchers have increasingly used the technology known as "lab-on-a-chip" or "organ-on-a-chip" to test the effects of pharmaceuticals, toxins, and chemicals on humans. Rather than testing on animals, which raises ethical concerns and can sometimes be inaccurate, and human-based clinical trials, which can be expensive and difficult to iterate, scientists turn to tiny, micro-engineered chips—about the size of a thumb drive.
It's possible that doctors could one day take individual cell samples and create personalized treatments, testing out any medications on the chip.
The chips are lined with living samples of human cells, which mimic the physiology and mechanical forces experienced by cells inside the human body, down to blood flow and breathing motions; the functions of organs ranging from kidneys and lungs to skin, eyes, and the blood-brain barrier.
A more recent—and potentially even more useful—development takes organ-on-a-chip technology to the next level by integrating several chips into a "body-on-a-chip." Since human organs don't work in isolation, seeing how they all react—and interact—once a foreign element has been introduced can be crucial to understanding how a certain treatment will or won't perform. Dr. Shyni Varghese, a MEDx investigator at the Duke University School of Medicine, is one of the researchers working with these systems in order to gain a more nuanced understanding of how multiple different organs react to the same stimuli.
Her lab is working on "tumor-on-a-chip" models, which can not only show the progression and treatment of cancer, but also model how other organs would react to immunotherapy and other drugs. "The effect of drugs on different organs can be tested to identify potential side effects," Varghese says. In addition, these models can help the researchers figure out how cancers grow and spread, as well as how to effectively encourage immune cells to move in and attack a tumor.
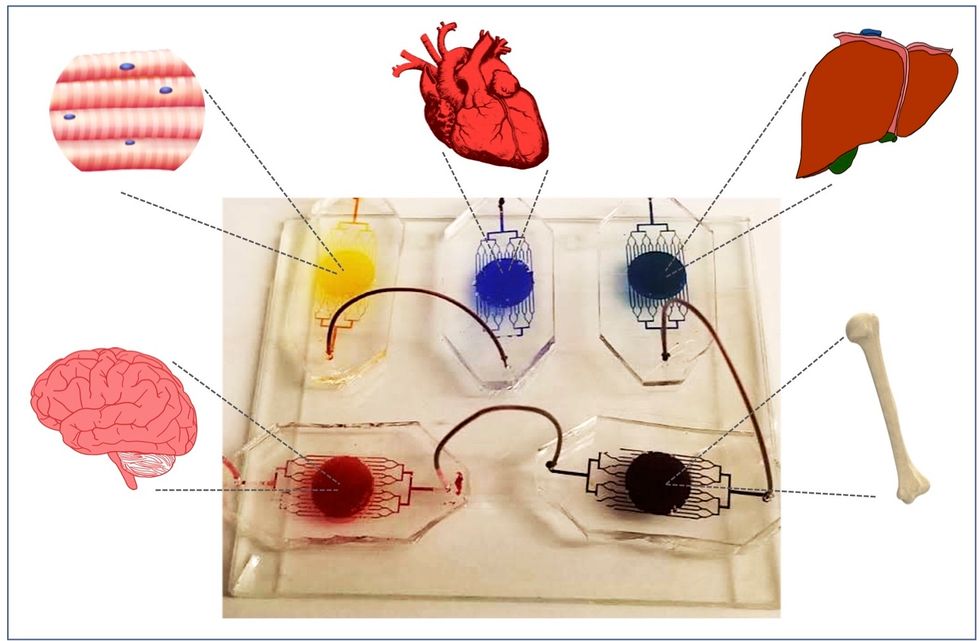
One body-on-a-chip used by Dr. Varghese's lab tracks the interactions of five organs—brain, heart, liver, muscle, and bone.
As their research progresses, Varghese and her team are looking for ways to maintain the long-term function of the engineered organs. In addition, she notes that this kind of research is not just useful for generalized testing; "organ-on-chip technologies allow patient-specific analyses, which can be used towards a fundamental understanding of disease progression," Varghese says. It's possible that doctors could one day take individual cell samples and create personalized treatments, testing out any medications on the chip for safety, efficacy, and potential side effects before writing a prescription.
2. Happening Soon: Prime Editing Will Have the Power to "Find and Replace" Disease-Causing Genes
Biochemist David Liu made industry-wide news last fall when he and his lab at MIT's Broad Institute, led by Andrew Anzalone, published a paper on prime editing: a new, more focused technology for editing genes. Prime editing is a descendant of the CRISPR-Cas9 system that researchers have been working with for years, and a cousin to Liu's previous innovation—base editing, which can make a limited number of changes to a single DNA letter at a time.
By contrast, prime editing has the potential to make much larger insertions and deletions; it also doesn't require the tweaked cells to divide in order to write the changes into the DNA, which could make it especially suitable for central nervous system diseases, like Parkinson's.
Crucially, the prime editing technique has a much higher efficiency rate than the older CRISPR system, and a much lower incidence of accidental insertions or deletions, which can make dangerous changes for a patient.
It also has a very broad potential range: according to Liu, 89% of the pathogenic mutations that have been collected in ClinVar (a public archive of human variations) could, in principle, be treated with prime editing—although he is careful to note that correcting a single genetic mutation may not be sufficient to fully treat a genetic disease.
Figuring out just how prime editing can be used most effectively and safely will be a long process, but it's already underway. The same day that Liu and his team posted their paper, they also made the basic prime editing constructs available for researchers around the world through Addgene, a plasmid repository, so that others in the scientific community can test out the technique for themselves. It might be years before human patients will see the results, and in the meantime, significant bioethical questions remain about the limits and sociological effects of such a powerful gene-editing tool. But in the long fight against genetic diseases, it's a huge step forward.
3. Happening When We Fund It: Focusing on Microbiome Health Could Help Us Tackle Social Inequality—And Vice Versa
The past decade has seen a growing awareness of the major role that the microbiome, the microbes present in our digestive tract, play in human health. Having a less-healthy microbiome is correlated with health risks like diabetes and depression, and interventions that target gut health, ranging from kombucha to fecal transplants, have cropped up with increasing frequency.
New research from the University of Maine's Dr. Suzanne Ishaq takes an even broader view, arguing that low-income and disadvantaged populations are less likely to have healthy, diverse gut bacteria, and that increasing access to beneficial microorganisms is an important juncture of social justice and public health.
"Basically, allowing people to lead healthy lives allows them to access and recruit microbes."
"Typically, having a more diverse bacterial community is associated with health, and having fewer different species is associated with illness and may leave you open to infection from bacteria that are good at exploiting opportunities," Ishaq says.
Having a healthy biome doesn't mean meeting one fixed ratio of gut bacteria, since different combinations of microbes can generate roughly similar results when they work in concert. Generally, "good" microbes are the ones that break down fiber and create the byproducts that we use for energy, or ones like lactic acid bacteria that work to make microbials and keep other bacteria in check. The microbial universe in your gut is chaotic, Ishaq says. "Microbes in your gut interact with each other, with you, with your food, or maybe they don't interact at all and pass right through you." Overall, it's tricky to name specific microbial communities that will make or break someone's health.
There are important corollaries between environment and biome health, though, which Ishaq points out: Living in urban environments reduces microbial exposure, and losing the microorganisms that humans typically source from soil and plants can reduce our adaptive immunity and ability to fight off conditions like allergies and asthma. Access to green space within cities can counteract those effects, but in the U.S. that access varies along income, education, and racial lines. Likewise, lower-income communities are more likely to live in food deserts or areas where the cheapest, most convenient food options are monotonous and low in fiber, further reducing microbial diversity.
Ishaq also suggests other areas that would benefit from further study, like the correlation between paid family leave, breastfeeding, and gut microbiota. There are technical and ethical challenges to direct experimentation with human populations—but that's not what Ishaq sees as the main impediment to future research.
"The biggest roadblock is money, and the solution is also money," she says. "Basically, allowing people to lead healthy lives allows them to access and recruit microbes."
That means investment in things we already understand to improve public health, like better education and healthcare, green space, and nutritious food. It also means funding ambitious, interdisciplinary research that will investigate the connections between urban infrastructure, housing policy, social equity, and the millions of microbes keeping us company day in and day out.

