Breakthrough in Creating Fuel from Sunlight Puts Us Closer to Carbon-Neutral Energy

Recent leaps in technology represent an important step forward in unlocking artificial photosynthesis.
Since the beginning of life on Earth, plants have been naturally converting sunlight into energy. This photosynthesis process that's effortless for them has been anything but for scientists who have been trying to achieve artificial photosynthesis for the last half a century with the goal of creating a carbon-neutral fuel. Such a fuel could be a gamechanger — rather than putting CO2 back into the atmosphere like traditional fuels do, it would take CO2 out of the atmosphere and convert it into usable energy.
If given the option between a carbon-neutral fuel at the gas station and a fuel that produces carbon dioxide in spades -- and if costs and effectiveness were equal --who wouldn't choose the one best for the planet? That's the endgame scientists are after. A consumer switch to clean fuel could have a huge impact on our global CO2 emissions.
Up until this point, the methods used to make liquid fuel from atmospheric CO2 have been expensive, not efficient enough to really get off the ground, and often resulted in unwanted byproducts. But now, a new technology may be the key to unlocking the full potential of artificial photosynthesis. At the very least, it's a step forward and could help make a dent in atmospheric CO2 reduction.
"It's an important breakthrough in artificial photosynthesis," says Qian Wang, a researcher in the Department of Chemistry at Cambridge University and lead author on a recent study published in Nature about an innovation she calls "photosheets."
The latest version of the artificial leaf directly produces liquid fuel, which is easier to transport and use commercially.
These photosheets convert CO2, sunlight, and water into a carbon-neutral liquid fuel called formic acid without the aid of electricity. They're made of semiconductor powders that absorb sunlight. When in the presence of water and CO2, the electrons in the powders become excited and join with the CO2 and protons from the water molecules, reducing the CO2 in the process. The chemical reaction results in the production of formic acid, which can be used directly or converted to hydrogen, another clean energy fuel.
In the past, it's been difficult to reduce CO2 without creating a lot of unwanted byproducts. According to Wang, this new conversion process achieves the reduction and fuel creation with almost no byproducts.
The Cambridge team's new technology is a first and certainly momentous, but they're far from the only team to have produced fuel from CO2 using some form of artificial photosynthesis. More and more scientists are aiming to perfect the method in hopes of producing a truly sustainable, photosynthetic fuel capable of lowering carbon emissions.
Thanks to advancements in nanoscience, which has led to better control of materials, more successes are emerging. A team at the University of Illinois at Urbana-Champaign, for example, used gold nanoparticles as the photocatalysts in their process.
"My group demonstrated that you could actually use gold nanoparticles both as a light absorber and a catalyst in the process of converting carbon dioxide to hydrocarbons such as methane, ethane and propane fuels," says professor Prashant Jain, co-author of the study. Not only are gold nanoparticles great at absorbing light, they don't degrade as quickly as other metals, which makes them more sustainable.
That said, Jain's team, like every other research team working on artificial photosynthesis including the Cambridge team, is grappling with efficiency issues. Jain says that all parts of the process need to be optimized so the reaction can happen as quickly as possible.
"You can't just improve one [aspect], because that can lead to a decrease in performance in some other aspects," Jain explains.
The Cambridge team is currently experimenting with a range of catalysts to improve their device's stability and efficiency. Virgil Andrei, who is working on an artificial leaf design that was developed at Cambridge in 2019, was recently able to improve the performance and selectivity of the device. Now the leaf's solar-to-CO2 energy conversion efficiency is 0.2%, twice its previous efficiency.
The latest version also directly produces liquid fuel, which is easier to transport and use commercially.
In determining a method of fuel production's efficiency, one must consider how sustainable it is at every stage. That involves calculating whenever excess energy is needed to complete a step. According to Jain, in order to use CO2 for fuel production, you have to condense the CO2, which takes energy. And on the fuel production side, once the chemical reaction has created your byproducts, they need to be separated, which also takes energy.
To be truly sustainable, each part of the conversion system also needs to be durable. If parts need to be replaced often, or regularly maintained, that counts against it. Then you have to account for the system's reuse cycle. If you extract CO2 from the environment and convert it into fuel that's then put into a fuel cell, it's going to release CO2 at the other end. In order to create a fully green, carbon-neutral fuel source, that same amount of CO2 needs to be trapped and reintroduced back into the fuel conversion system.
"The cycle continues, and at each point, you will see a loss in efficiency, and depending on how much you [may also] see a loss in yield," says Jain. "And depending on what those efficiencies are at each one of those points will determine whether or not this process can be sustainable."
The science is at least a decade away from offering a competitive sustainable fuel option at scale. Streamlining a process to mimic what plants have perfected over billions of years is no small feat, but an ever-growing community of researchers using rapidly advancing technology is driving progress forward.
Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
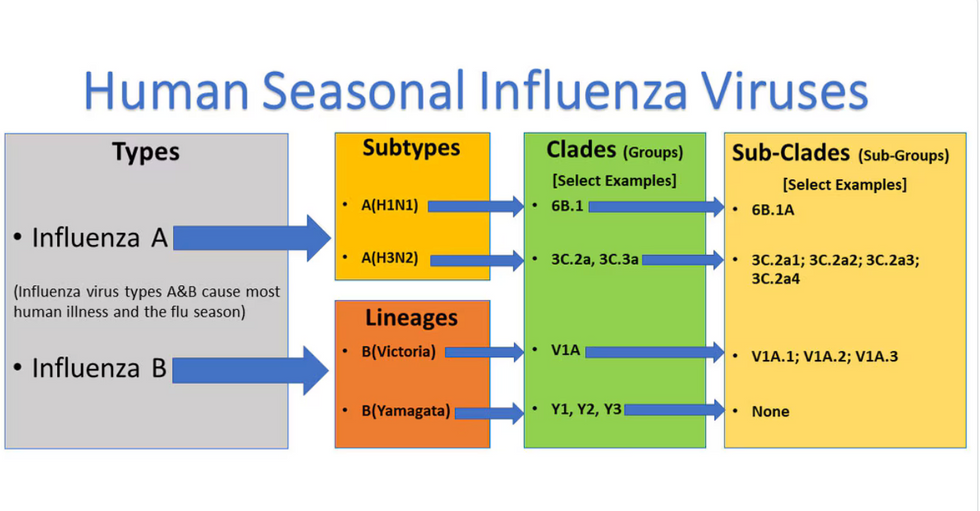
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.

