How 30 Years of Heart Surgeries Taught My Dad How to Live

A mid-1970s photo of the author's father, and him holding a grandchild in 2012.
[Editor's Note: This piece is the winner of our 2019 essay contest, which prompted readers to reflect on the question: "How has an advance in science or medicine changed your life?"]
My father did not expect to live past the age of 50. Neither of his parents had done so. And he also knew how he would die: by heart attack, just as his father did.
In July of 1976, he had his first heart attack, days before his 40th birthday.
My dad lived the first 40 years of his life with this knowledge buried in his bones. He started smoking at the age of 12, and was drinking before he was old enough to enlist in the Navy. He had a sarcastic, often cruel, sense of humor that could drive my mother, my sister and me into tears. He was not an easy man to live with, but that was okay by him - he didn't expect to live long.
In July of 1976, he had his first heart attack, days before his 40th birthday. I was 13, and my sister was 11. He needed quadruple bypass surgery. Our small town hospital was not equipped to do this type of surgery; he would have to be transported 40 miles away to a heart center. I understood this journey to mean that my father was seriously ill, and might die in the hospital, away from anyone he knew. And my father knew a lot of people - he was a popular high school English teacher, in a town with only three high schools. He knew generations of students and their parents. Our high school football team did a blood drive in his honor.

During a trip to Disney World in 1974, Dad was suffering from angina the entire time but refused to tell me (left) and my sister, Kris.
Quadruple bypass surgery in 1976 meant that my father's breastbone was cut open by a sternal saw. His ribcage was spread wide. After the bypass surgery, his bones would be pulled back together, and tied in place with wire. The wire would later be pulled out of his body when the bones knitted back together. It would take months before he was fully healed.
Dad was in the hospital for the rest of the summer and into the start of the new school year. Going to visit him was farther than I could ride my bicycle; it meant planning a trip in the car and going onto the interstate. The first time I was allowed to visit him in the ICU, he was lying in bed, and then pushed himself to sit up. The heart monitor he was attached to spiked up and down, and I fainted. I didn't know that heartbeats change when you move; television medical dramas never showed that - I honestly thought that I had driven my father into another heart attack.
Only a few short years after that, my father returned to the big hospital to have his heart checked with a new advance in heart treatment: a CT scan. This would allow doctors to check for clogged arteries and treat them before a fatal heart attack. The procedure identified a dangerous blockage, and my father was admitted immediately. This time, however, there was no need to break bones to get to the problem; my father was home within a month.
During the late 1970's, my father changed none of his habits. He was still smoking, and he continued to drink. But now, he was also taking pills - pills to manage the pain. He would pop a nitroglycerin tablet under his tongue whenever he was experiencing angina (I have a vivid memory of him doing this during my driving lessons), but he never mentioned that he was in pain. Instead, he would snap at one of us, or joke that we were killing him.
I think he finally determined that, if he was going to have these extra decades of life, he wanted to make them count.
Being the kind of guy he was, my father never wanted to talk about his health. Any admission of pain implied that he couldn't handle pain. He would try to "muscle through" his angina, as if his willpower would be stronger than his heart muscle. His efforts would inevitably fail, leaving him angry and ready to lash out at anyone or anything. He would blame one of us as a reason he "had" to take valium or pop a nitro tablet. Dinners often ended in shouts and tears, and my father stalking to the television room with a bottle of red wine.
In the 1980's while I was in college, my father had another heart attack. But now, less than 10 years after his first, medicine had changed: our hometown hospital had the technology to run dye through my father's blood stream, identify the blockages, and do preventative care that involved statins and blood thinners. In one case, the doctors would take blood vessels from my father's legs, and suture them to replace damaged arteries around his heart. New advances in cholesterol medication and treatments for angina could extend my father's life by many years.
My father decided it was time to quit smoking. It was the first significant health step I had ever seen him take. Until then, he treated his heart issues as if they were inevitable, and there was nothing that he could do to change what was happening to him. Quitting smoking was the first sign that my father was beginning to move out of his fatalistic mindset - and the accompanying fatal behaviors that all pointed to an early death.
In 1986, my father turned 50. He had now lived longer than either of his parents. The habits he had learned from them could be changed. He had stopped smoking - what else could he do?
It was a painful decade for all of us. My parents divorced. My sister quit college. I moved to the other side of the country and stopped speaking to my father for almost 10 years. My father remarried, and divorced a second time. I stopped counting the number of times he was in and out of the hospital with heart-related issues.
In the early 1990's, my father reached out to me. I think he finally determined that, if he was going to have these extra decades of life, he wanted to make them count. He traveled across the country to spend a week with me, to meet my friends, and to rebuild his relationship with me. He did the same with my sister. He stopped drinking. He was more forthcoming about his health, and admitted that he was taking an antidepressant. His humor became less cruel and sadistic. He took an active interest in the world. He became part of my life again.
The 1990's was also the decade of angioplasty. My father explained it to me like this: during his next surgery, the doctors would place balloons in his arteries, and inflate them. The balloons would then be removed (or dissolve), leaving the artery open again for blood. He had several of these surgeries over the next decade.
When my father was in his 60's, he danced at with me at my wedding. It was now 10 years past the time he had expected to live, and his life was transformed. He was living with a woman I had known since I was a child, and my wife and I would make regular visits to their home. My father retired from teaching, became an avid gardener, and always had a home project underway. He was a happy man.

Dancing with my father at my wedding in 1998.
Then, in the mid 2000's, my father faced another serious surgery. Years of arterial surgery, angioplasty, and damaged heart muscle were taking their toll. He opted to undergo a life-saving surgery at Cleveland Clinic. By this time, I was living in New York and my sister was living in Arizona. We both traveled to the Midwest to be with him. Dad was unconscious most of the time. We took turns holding his hand in the ICU, encouraging him to regain his will to live, and making outrageous threats if he didn't listen to us.
The nursing staff were wonderful. I remember telling them that my father had never expected to live this long. One of the nurses pointed out that most of the patients in their ward were in their 70's and 80's, and a few were in their 90's. She reminded me that just a decade earlier, most hospitals were unwilling to do the kind of surgery my father had received on patients his age. In the first decade of the 21st century, however, things were different: 90-year-olds could now undergo heart surgery and live another decade. My father was on the "young" side of their patients.
The Cleveland Clinic visit would be the last major heart surgery my father would have. Not that he didn't return to his local hospital a few times after that: he broke his neck -- not once, but twice! -- slipping on ice. And in the 2010's, he began to show signs of dementia, and needed more home care. His partner, who had her own health issues, was not able to provide the level of care my father needed. My sister invited him to move in with her, and in 2015, I traveled with him to Arizona to get him settled in.
After a few months, he accepted home hospice. We turned off his pacemaker when the hospice nurse explained to us that the job of a pacemaker is to literally jolt a patient's heart back into beating. The jolts were happening more and more frequently, causing my Dad additional, unwanted pain.

My father in 2015, a few months before his death.
My father died in February 2016. His body carried the scars and implants of 30 years of cardiac surgeries, from the ugly breastbone scar from the 1970's to scars on his arms and legs from borrowed blood vessels, to the tiny red circles of robotic incisions from the 21st century. The arteries and veins feeding his heart were a patchwork of transplanted leg veins and fragile arterial walls pressed thinner by balloons.
And my father died with no regrets or unfinished business. He died in my sister's home, with his long-time partner by his side. Medical advancements had given him the opportunity to live 30 years longer than he expected. But he was the one who decided how to live those extra years. He was the one who made the years matter.
7 Reasons Why We Should Not Need Boosters for COVID-19
A top infectious disease physician explains why immunity derived from natural infection and the vaccines should be long-lasting.
There are at least 7 reasons why immunity after vaccination or infection with COVID-19 should likely be long-lived. If durable, I do not think boosters will be necessary in the future, despite CEOs of pharmaceutical companies (who stand to profit from boosters) messaging that they may and readying such boosters. To explain these reasons, let's orient ourselves to the main components of the immune system.
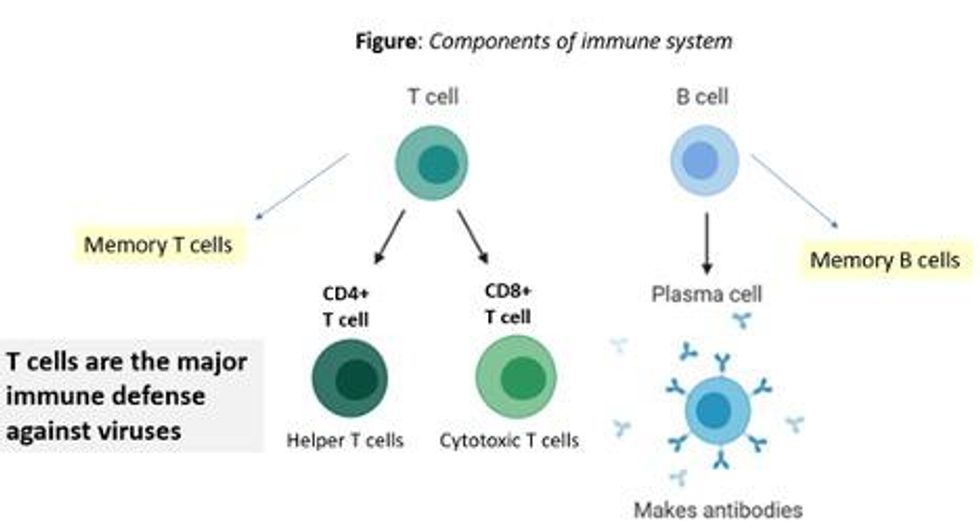
There are two major arms of the immune system: B cells (which produce antibodies) and T cells (which are formed specifically to attack and kill pathogens). T cells are divided into two types, CD4 cells ("helper" T cells) and CD8 cells ("cytotoxic" T cells).
Each arm, once stimulated by infection or vaccine, should hopefully make "memory" banks. So if the body sees the pathogen in the future, these defenses should come roaring back to attack the virus and protect you from getting sick. Plenty of research in COVID-19 indicates a likely long-lasting response to the vaccine or infection. Here are seven of the most compelling reasons:
REASON 1: Memory B Cells Are Produced By Vaccines and Natural Infection
In one study, 12 volunteers who had never had Covid-19--and were fully vaccinated with two Pfizer/BioNTech shots-- underwent biopsies of their lymph nodes. This is where memory B cells are stored in places called "germinal centers". The biopsies were performed three, four, six, and seven weeks after the first mRNA vaccine shot, and were stained to reveal that germinal center memory B cells in the lymph nodes increased in concentration over time.
Natural infection also generates memory B cells. Even after antibody levels wane over time, strong memory B cells were detected in the blood of individuals six and eight months after infection in different studies. Indeed, the half-lives of the memory B cells seen in the study examining patients 8 months after COVID-19 led the authors to conclude that "B cell memory to SARS-CoV-2 was robust and is likely long-lasting." Reason #2 tells us that memory B cells can be active for a very long time indeed.
REASON #2: Memory B Cells Can Produce Neutralizing Antibodies If They See Infection Again Decades Later
Demonstrated production of memory B cells after vaccination or natural infection with COVID-19 is so important because memory B cells, once generated, can be activated to produce high levels of neutralizing antibodies against the pathogen even if encountered many years after the initial exposure. In one amazing study (published in 2008), researchers isolated memory B cells against the 1918 flu strain from the blood of 32 individuals aged 91-101 years. These people had been born on or before 1915 and had survived that pandemic.
Their memory B cells, when exposed to the 1918 flu strain in a test tube, generated high levels of neutralizing antibodies against the virus -- antibodies that then protected mice from lethal infection with this deadly strain. The ability of memory B cells to produce complex antibody responses against an infection nine decades after exposure speaks to their durability.
REASON #3: Vaccines or Natural Infection Trigger Strong Memory T Cell Immunity
All of the trials of the major COVID-19 vaccine candidates measured strong T cell immunity following vaccination, most often assessed by measuring SARS-CoV-2 specific T cells in the phase I/II safety and immunogenicity studies. There are a number of studies that demonstrate the production of strong T cell immunity to COVID-19 after natural infection as well, even when the infection was mild or asymptomatic.
The same study that showed us robust memory B cell production 8 months after natural infection also demonstrated strong and sustained memory T cell production. In fact, the half-lives of the memory T cells in this cohort were long (~125-225 days for CD8+ and ~94-153 days for CD4+ T cells), comparable to the 123-day half-life observed for memory CD8+ T cells after yellow fever immunization (a vaccine usually given once over a lifetime).
A recent study of individuals recovered from COVID-19 show that the initial T cells generated by natural infection mature and differentiate over time into memory T cells that will be "put in the bank" for sustained periods.
REASON #4: T Cell Immunity Following Vaccinations for Other Infections Is Long-Lasting
Last year, we were fortunate to be able to measure how T cell immunity is generated by COVID-19 vaccines, which was not possible in earlier eras when vaccine trials were done for other infections (such as measles, mumps, rubella, pertussis, diphtheria). Antibodies are just the "tip of the iceberg" when assessing the response to vaccination, but were the only arm of the immune response that could be measured following vaccination in the past.
Measuring pathogen-specific T cell responses takes sophisticated technology. However, T cell responses, when assessed years after vaccination for other pathogens, has been shown to be long-lasting. For example, in one study of 56 volunteers who had undergone measles vaccination when they were much younger, strong CD8 and CD4 cell responses to vaccination could be detected up to 34 years later.
REASON #5: T Cell Immunity to Related Coronaviruses That Caused Severe Disease is Long-Lasting
SARS-CoV-2 is a coronavirus that causes severe disease, unlike coronaviruses that cause the common cold. Two other coronaviruses in the recent past caused severe disease, specifically Severely Acute Respiratory Distress Syndrome (SARS) in late 2002-2003 and Middle East Respiratory Syndrome (MERS) in 2011.
A study performed in 2020 demonstrated that the blood of 23 recovered SARS patients possess long-lasting memory T cells that were still reactive to SARS 17 years after the outbreak in 2003. Many scientists expect that T cell immunity to SARS-CoV-2 will be equally durable to that of its cousin.
REASON #6: T Cell Responses from Vaccination and Natural Infection With the Ancestral Strain of COVID-19 Are Robust Against Variants
Even though antibody responses from vaccination may be slightly lower against various COVID-19 variants of concern that have emerged in recent months, T cell immunity after vaccination has been shown to be unperturbed by mutations in the spike protein (in the variants). For instance, T cell responses after mRNA vaccines maintained strong activity against different variants (including P.1 Brazil variant, B.1.1.7 UK variant, B.1.351 South Africa variant and the CA.20.C California variant) in a recent study.
Another study showed that the vaccines generated robust T cell immunity that was unfazed by different variants, including B.1.351 and B.1.1.7. The CD4 and CD8 responses generated after natural infection are equally robust, showing activity against multiple "epitopes" (little segments) of the spike protein of the virus. For instance, CD8 cells responds to 52 epitopes and CD4 cells respond to 57 epitopes across the spike protein, so that a few mutations in the variants cannot knock out such a robust and in-breadth T cell response. Indeed, a recent paper showed that mRNA vaccines were 97.4 percent effective against severe COVID-19 disease in Qatar, even when the majority of circulating virus there was from variants of concern (B.1.351 and B.1.1.7).
REASON #7: Coronaviruses Don't Mutate Quickly Like Influenza, Which Requires Annual Booster Shots
Coronaviruses are RNA viruses, like influenza and HIV (which is actually a retrovirus), but do not mutate as quickly as either one. The reason that coronaviruses don't mutate very rapidly is that their replicating mechanism (polymerase) has a strong proofreading mechanism: If the virus mutates, it usually goes back and self-corrects. Mutations can arise with high rates of replication when transmission is very frequent -- as has been seen in recent months with the emergence of SARS-CoV-2 variants during surges. However, the COVID-19 virus will not be mutating like this when we tamp down transmission with mass vaccination.
In conclusion, I and many of my infectious disease colleagues expect the immunity from natural infection or vaccination to COVID-19 to be durable. Let's put discussion of boosters aside and work hard on global vaccine equity and distribution since the pandemic is not over until it is over for us all.
Professor Tim Caulfield stops by the "Making Sense of Science" podcast this month to discuss the infodemic around COVID-19 and how to deal with it.
The "Making Sense of Science" podcast features interviews with leading medical and scientific experts about the latest developments and the big ethical and societal questions they raise. This monthly podcast is hosted by journalist Kira Peikoff, founding editor of the award-winning science outlet Leaps.org.
Hear the 30-second trailer:
Listen to the whole episode:
.
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.

