How Can We Decide If a Biomedical Advance Is Ethical?

A newborn in the maternity ward. (© rufous/Fotolia)
"All fixed, fast-frozen relations, with their train of ancient and venerable prejudices and opinions, are swept away, all new-formed ones become antiquated before they can ossify. All that is solid melts into air, all that is holy is profaned…"
On July 25, 1978, Louise Brown was born in Oldham, England, the first human born through in vitro fertilization, through the work of Patrick Steptoe, a gynecologist, and Robert Edwards, a physiologist. Her birth was greeted with strong (though not universal) expressions of ethical dismay. Yet in 2016, the latest year for which we have data, nearly two percent of the babies born in the United States – and around the same percentage throughout the developed world – were the result of IVF. Few, if any, think of these children as unnatural, monsters, or freaks or of their parents as anything other than fortunate.
How should we view Dr. He today, knowing that the world's eventual verdict on the ethics of biomedical technologies often changes?
On November 25, 2018, news broke that Chinese scientist, Dr. He Jiankui, claimed to have edited the genomes of embryos, two of whom had recently become the new babies, Lulu and Nana. The response was immediate and overwhelmingly negative.
Times change. So do views. How will Dr. He be viewed in 40 years? And, more importantly, how should we view him today, knowing that the world's eventual verdict on the ethics of biomedical technologies often changes? And when what biomedicine can do changes with vertiginous frequency?
How to determine what is and isn't ethical is above my pay grade. I'm a simple law professor – I can't claim any deeper insight into how to live a moral life than the millennia of religious leaders, philosophers, ethicists, and ordinary people trying to do the right thing. But I can point out some ways to think about these questions that may be helpful.
First, consider two different kinds of ethical commands. Some are quite specific – "thou shalt not kill," for example. Others are more general – two of them are "do unto others as you would have done to you" or "seek the greatest good for the greatest number."
Biomedicine in the last two centuries has often surprised us with new possibilities, situations that cultures, religions, and bodies of ethical thought had not previously had to consider, from vaccination to anesthesia for women in labor to genome editing. Sometimes these possibilities will violate important and deeply accepted precepts for a group or a person. The rise of blood transfusions around World War I created new problems for Jehovah's Witnesses, who believe that the Bible prohibits ingesting blood. The 20th century developments of artificial insemination and IVF both ran afoul of Catholic doctrine prohibiting methods other than "traditional" marital intercourse for conceiving children. If you subscribe to an ethical or moral code that contains prohibitions that modern biomedicine violates, the issue for you is stark – adhere to those beliefs or renounce them.
If the harms seem to outweigh the benefits, it's easy to conclude "this is worrisome."
But many biomedical changes violate no clear moral teachings. Is it ethical or not to edit the DNA of embryos? Not surprisingly, the sacred texts of various religions – few of which were created after, at the latest, the early 19th century, say nothing specific about this. There may be hints, precedents, leanings that could argue one way or another, but no "commandments." In that case, I recommend, at least as a starting point, asking "what are the likely consequences of these actions?"
Will people be, on balance, harmed or helped by them? "Consequentialist" approaches, of various types, are a vast branch of ethical theories. Personally I find a completely consequentialist approach unacceptable – I could not accept, for example, torturing an innocent child even in order to save many lives. But, in the absence of a clear rule, looking at the consequences is a great place to start. If the harms seem to outweigh the benefits, it's easy to conclude "this is worrisome."
Let's use that starting place to look at a few bioethical issues. IVF, for example, once proven (relatively) safe seems to harm no one and to help many, notably the more than 8 million children worldwide born through IVF since 1978 – and their 16 million parents. On the other hand, giving unknowing, and unconsenting, intellectually disabled children hepatitis A harmed them, for an uncertain gain for science. And freezing the heads of the dead seems unlikely to harm anyone alive (except financially) but it also seems almost certain not to benefit anyone. (Those frozen dead heads are not coming back to life.)
Now let's look at two different kinds of biomedical advances. Some are controversial just because they are new; others are controversial because they cut close to the bone – whether or not they violate pre-established ethical or moral norms, they clearly relate to them.
Consider anesthesia during childbirth. When first used, it was controversial. After all, said critics, in Genesis, the Bible says God told Eve, "I will greatly multiply Your pain in childbirth, In pain you will bring forth children." But it did not clearly prohibit pain relief and from the advent of ether on, anesthesia has been common, though not universal, in childbirth in western societies. The pre-existing ethical precepts were not clear and the consequences weighed heavily in favor of anesthesia. Similarly, vaccination seems to violate no deep moral principle. It was, and for some people, still is just strange, and unnatural. The same was true of IVF initially. Opposition to all of these has faded with time and familiarity. It has not disappeared – some people continue to find moral or philosophical problems with "unnatural" childbirth, vaccination, and IVF – but far fewer.
On the other hand, human embryonic stem cell research touches deeper issues. Human embryos are destroyed to make those stem cells. Reasonable people disagree on the moral status of the human embryo, and the moral weight of its destruction, but it does at least bring into play clear and broadly accepted moral precepts, such as "Thou shalt not kill." So, at the far side of an individual's time, does euthanasia. More exposure to, and familiarity with, these practices will not necessarily lead to broad acceptance as the objections involve more than novelty.
The first is "what would I do?" The second – what should my government, culture, religion allow or forbid?
Finally, all this ethical analysis must work at two levels. The first is "what would I do?" The second – what should my government, culture, religion allow or forbid? There are many things I would not do that I don't think should be banned – because I think other people may reasonably have different views from mine. I would not get cosmetic surgery, but I would not ban it – and will try not to think ill of those who choose it
So, how should we assess the ethics of new biomedical procedures when we know that society's views may change? More specifically, what should we think of He Jiankui's experiment with human babies?
First, look to see whether the procedure in question violates, at least fairly clearly, some rule in your ethical or moral code. If so, your choice may not be difficult. But if the procedure is unmentioned in your moral code, probably because it was inconceivable to the code's creators, examine the consequences of the act.
If the procedure is just novel, and not something that touches on important moral concerns, looking at the likely consequences may be enough for your ethical analysis –though it is always worth remembering that predicting consequences perfectly is impossible and predicting them well is never certain. If it does touch on morally significant issues, you need to think those issues through. The consequences may be important to your conclusions but they may not be determinative.
And, then, if you conclude that it is not ethical from your perspective, you need to take yet another step and consider whether it should be banned for people who do not share your perspective. Sometimes the answer will be yes – that psychopaths may not view murder as immoral does not mean we have to let them kill – but sometimes it will be no.
What does this say about He Jiankui's experiment? I have no qualms in condemning it, unequivocally. The potential risks to the babies grossly outweighed any benefits to them, and to science. And his secret work, against a near universal scientific consensus, privileged his own ethical conclusions without giving anyone else a vote, or even a voice.
But if, in ten or twenty years, genome editing of human embryos is shown to be safe (enough) and it is proposed to be used for good reasons – say, to relieve human suffering that could not be treated in other good ways – and with good consents from those directly involved as well as from the relevant society and government – my answer might well change. Yours may not. Bioethics is a process for approaching questions; it is not a set of universal answers.
This article opened with a quotation from the 1848 Communist Manifesto, referring to the dizzying pace of change from industrialization and modernity. You don't need to be a Marxist to appreciate that sentiment. Change – especially in the biosciences – keeps accelerating. How should we assess the ethics of new biotechnologies? The best we can, with what we know, at the time we inhabit. And, in the face of vast uncertainty, with humility.
DNA- and RNA-based electronic implants may revolutionize healthcare
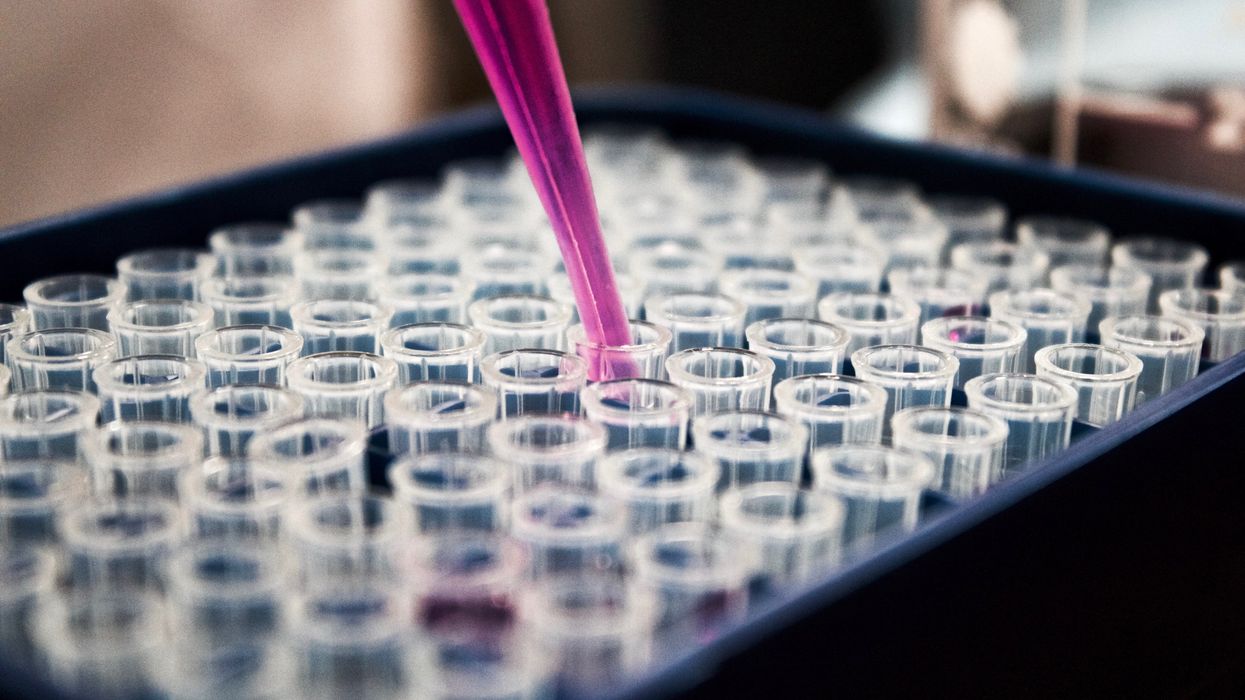
The test tubes contain tiny DNA/enzyme-based circuits, which comprise TRUMPET, a new type of electronic device, smaller than a cell.
Implantable electronic devices can significantly improve patients’ quality of life. A pacemaker can encourage the heart to beat more regularly. A neural implant, usually placed at the back of the skull, can help brain function and encourage higher neural activity. Current research on neural implants finds them helpful to patients with Parkinson’s disease, vision loss, hearing loss, and other nerve damage problems. Several of these implants, such as Elon Musk’s Neuralink, have already been approved by the FDA for human use.
Yet, pacemakers, neural implants, and other such electronic devices are not without problems. They require constant electricity, limited through batteries that need replacements. They also cause scarring. “The problem with doing this with electronics is that scar tissue forms,” explains Kate Adamala, an assistant professor of cell biology at the University of Minnesota Twin Cities. “Anytime you have something hard interacting with something soft [like muscle, skin, or tissue], the soft thing will scar. That's why there are no long-term neural implants right now.” To overcome these challenges, scientists are turning to biocomputing processes that use organic materials like DNA and RNA. Other promised benefits include “diagnostics and possibly therapeutic action, operating as nanorobots in living organisms,” writes Evgeny Katz, a professor of bioelectronics at Clarkson University, in his book DNA- And RNA-Based Computing Systems.
While a computer gives these inputs in binary code or "bits," such as a 0 or 1, biocomputing uses DNA strands as inputs, whether double or single-stranded, and often uses fluorescent RNA as an output.
Adamala’s research focuses on developing such biocomputing systems using DNA, RNA, proteins, and lipids. Using these molecules in the biocomputing systems allows the latter to be biocompatible with the human body, resulting in a natural healing process. In a recent Nature Communications study, Adamala and her team created a new biocomputing platform called TRUMPET (Transcriptional RNA Universal Multi-Purpose GatE PlaTform) which acts like a DNA-powered computer chip. “These biological systems can heal if you design them correctly,” adds Adamala. “So you can imagine a computer that will eventually heal itself.”
The basics of biocomputing
Biocomputing and regular computing have many similarities. Like regular computing, biocomputing works by running information through a series of gates, usually logic gates. A logic gate works as a fork in the road for an electronic circuit. The input will travel one way or another, giving two different outputs. An example logic gate is the AND gate, which has two inputs (A and B) and two different results. If both A and B are 1, the AND gate output will be 1. If only A is 1 and B is 0, the output will be 0 and vice versa. If both A and B are 0, the result will be 0. While a computer gives these inputs in binary code or "bits," such as a 0 or 1, biocomputing uses DNA strands as inputs, whether double or single-stranded, and often uses fluorescent RNA as an output. In this case, the DNA enters the logic gate as a single or double strand.
If the DNA is double-stranded, the system “digests” the DNA or destroys it, which results in non-fluorescence or “0” output. Conversely, if the DNA is single-stranded, it won’t be digested and instead will be copied by several enzymes in the biocomputing system, resulting in fluorescent RNA or a “1” output. And the output for this type of binary system can be expanded beyond fluorescence or not. For example, a “1” output might be the production of the enzyme insulin, while a “0” may be that no insulin is produced. “This kind of synergy between biology and computation is the essence of biocomputing,” says Stephanie Forrest, a professor and the director of the Biodesign Center for Biocomputing, Security and Society at Arizona State University.
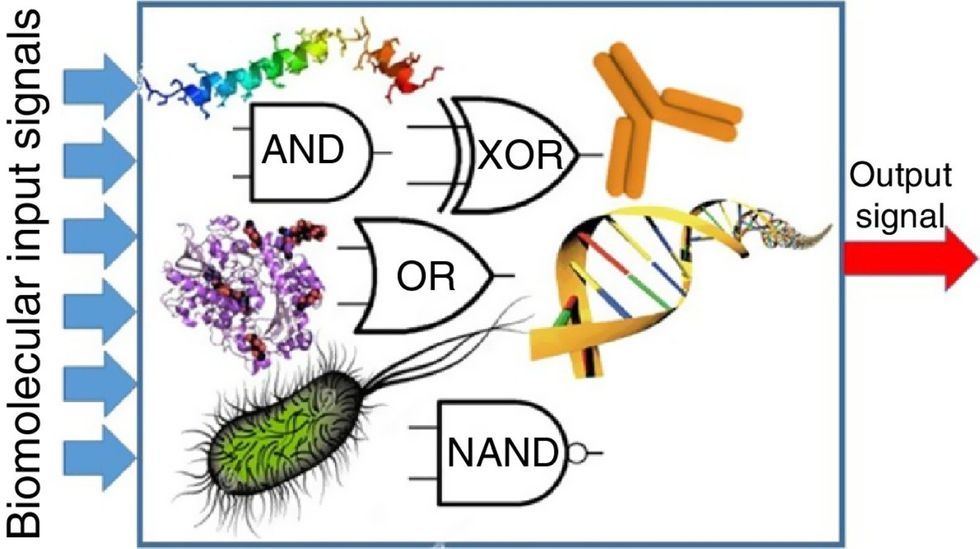
Biocomputing circles are made of DNA, RNA, proteins and even bacteria.
Evgeny Katz
The TRUMPET’s promise
Depending on whether the biocomputing system is placed directly inside a cell within the human body, or run in a test-tube, different environmental factors play a role. When an output is produced inside a cell, the cell's natural processes can amplify this output (for example, a specific protein or DNA strand), creating a solid signal. However, these cells can also be very leaky. “You want the cells to do the thing you ask them to do before they finish whatever their businesses, which is to grow, replicate, metabolize,” Adamala explains. “However, often the gate may be triggered without the right inputs, creating a false positive signal. So that's why natural logic gates are often leaky." While biocomputing outside a cell in a test tube can allow for tighter control over the logic gates, the outputs or signals cannot be amplified by a cell and are less potent.
TRUMPET, which is smaller than a cell, taps into both cellular and non-cellular biocomputing benefits. “At its core, it is a nonliving logic gate system,” Adamala states, “It's a DNA-based logic gate system. But because we use enzymes, and the readout is enzymatic [where an enzyme replicates the fluorescent RNA], we end up with signal amplification." This readout means that the output from the TRUMPET system, a fluorescent RNA strand, can be replicated by nearby enzymes in the platform, making the light signal stronger. "So it combines the best of both worlds,” Adamala adds.
These organic-based systems could detect cancer cells or low insulin levels inside a patient’s body.
The TRUMPET biocomputing process is relatively straightforward. “If the DNA [input] shows up as single-stranded, it will not be digested [by the logic gate], and you get this nice fluorescent output as the RNA is made from the single-stranded DNA, and that's a 1,” Adamala explains. "And if the DNA input is double-stranded, it gets digested by the enzymes in the logic gate, and there is no RNA created from the DNA, so there is no fluorescence, and the output is 0." On the story's leading image above, if the tube is "lit" with a purple color, that is a binary 1 signal for computing. If it's "off" it is a 0.
While still in research, TRUMPET and other biocomputing systems promise significant benefits to personalized healthcare and medicine. These organic-based systems could detect cancer cells or low insulin levels inside a patient’s body. The study’s lead author and graduate student Judee Sharon is already beginning to research TRUMPET's ability for earlier cancer diagnoses. Because the inputs for TRUMPET are single or double-stranded DNA, any mutated or cancerous DNA could theoretically be detected from the platform through the biocomputing process. Theoretically, devices like TRUMPET could be used to detect cancer and other diseases earlier.
Adamala sees TRUMPET not only as a detection system but also as a potential cancer drug delivery system. “Ideally, you would like the drug only to turn on when it senses the presence of a cancer cell. And that's how we use the logic gates, which work in response to inputs like cancerous DNA. Then the output can be the production of a small molecule or the release of a small molecule that can then go and kill what needs killing, in this case, a cancer cell. So we would like to develop applications that use this technology to control the logic gate response of a drug’s delivery to a cell.”
Although platforms like TRUMPET are making progress, a lot more work must be done before they can be used commercially. “The process of translating mechanisms and architecture from biology to computing and vice versa is still an art rather than a science,” says Forrest. “It requires deep computer science and biology knowledge,” she adds. “Some people have compared interdisciplinary science to fusion restaurants—not all combinations are successful, but when they are, the results are remarkable.”
Crickets are low on fat, high on protein, and can be farmed sustainably. They are also crunchy.
In today’s podcast episode, Leaps.org Deputy Editor Lina Zeldovich speaks about the health and ecological benefits of farming crickets for human consumption with Bicky Nguyen, who joins Lina from Vietnam. Bicky and her business partner Nam Dang operate an insect farm named CricketOne. Motivated by the idea of sustainable and healthy protein production, they started their unconventional endeavor a few years ago, despite numerous naysayers who didn’t believe that humans would ever consider munching on bugs.
Yet, making creepy crawlers part of our diet offers many health and planetary advantages. Food production needs to match the rise in global population, estimated to reach 10 billion by 2050. One challenge is that some of our current practices are inefficient, polluting and wasteful. According to nonprofit EarthSave.org, it takes 2,500 gallons of water, 12 pounds of grain, 35 pounds of topsoil and the energy equivalent of one gallon of gasoline to produce one pound of feedlot beef, although exact statistics vary between sources.
Meanwhile, insects are easy to grow, high on protein and low on fat. When roasted with salt, they make crunchy snacks. When chopped up, they transform into delicious pâtes, says Bicky, who invents her own cricket recipes and serves them at industry and public events. Maybe that’s why some research predicts that edible insects market may grow to almost $10 billion by 2030. Tune in for a delectable chat on this alternative and sustainable protein.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
Further reading:
More info on Bicky Nguyen
https://yseali.fulbright.edu.vn/en/faculty/bicky-n...
The environmental footprint of beef production
https://www.earthsave.org/environment.htm
https://www.watercalculator.org/news/articles/beef-king-big-water-footprints/
https://www.frontiersin.org/articles/10.3389/fsufs.2019.00005/full
https://ourworldindata.org/carbon-footprint-food-methane
Insect farming as a source of sustainable protein
https://www.insectgourmet.com/insect-farming-growing-bugs-for-protein/
https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/insect-farming
Cricket flour is taking the world by storm
https://www.cricketflours.com/
https://talk-commerce.com/blog/what-brands-use-cricket-flour-and-why/

Lina Zeldovich has written about science, medicine and technology for Popular Science, Smithsonian, National Geographic, Scientific American, Reader’s Digest, the New York Times and other major national and international publications. A Columbia J-School alumna, she has won several awards for her stories, including the ASJA Crisis Coverage Award for Covid reporting, and has been a contributing editor at Nautilus Magazine. In 2021, Zeldovich released her first book, The Other Dark Matter, published by the University of Chicago Press, about the science and business of turning waste into wealth and health. You can find her on http://linazeldovich.com/ and @linazeldovich.