Is Red Tape Depriving Patients of Life-Altering Therapies?

Medical treatment represented on a world map.
Rich Mancuso suffered from herpes for most of his adult life. The 49-year-old New Jersey resident was miserable. He had at least two to three outbreaks every month with painful and unsightly sores on his face and in his eyes, yet the drugs he took to control the disease had terrible side effects--agonizing headaches and severe stomach disturbances.
Last week, the FDA launched a criminal investigation to determine whether the biotech behind the vaccine had violated regulations.
So in 2016, he took an unusual step: he was flown to St. Kitt's, an island in the West Indies, where he participated in a clinical trial of a herpes vaccine, and received three injections of the experimental therapeutic during separate visits to the island. Within a year, his outbreaks stopped. "Nothing else worked," says Mancuso, who feels like he's gotten his life back. "And I've tried everything on the planet."
Mancuso was one of twenty genital herpes sufferers who were given the experimental vaccine in tests conducted on the Caribbean island and in hotel rooms near the campus of Southern Illinois University in Springfield where the vaccine's developer, microbiologist William Halford, was on the faculty. But these tests were conducted under the radar, without the approval or safety oversight of the Food and Drug Administration or an institutional review board (IRB), which routinely monitor human clinical trials of experimental drugs to make sure participants are protected.
Last week, the FDA launched a criminal investigation to determine whether anyone from SIU or Rational Vaccines, the biotech behind the vaccine, had violated regulations by aiding Halford's research. The SIU scientist was a microbiologist, not a medical doctor, which means that volunteers were not only injected with an unsanctioned experimental treatment but there wasn't even routine medical oversight.
On one side are scientists and government regulators with legitimate safety concerns....On the other are desperate patients and a dying scientist willing to go rogue in a foreign country.
Halford, who was stricken with a rare form of a nasal cancer, reportedly bypassed regulatory rules because the clock was ticking and he wanted to speed this potentially life-altering therapeutic to patients. "There was no way he had enough time to raise $100 million to test the drugs in the U.S.," says Mancuso, who became friends with Halford before he died in June of 2017 at age 48. "He knew if he didn't do something, his work would just die and no one would benefit. This was the only way."
But was it the only way? Once the truth about the trial came to light, public health officials in St. Kitt's disavowed the trial, saying they had not been notified that it was happening, and Southern Illinois University's medical school launched an investigation that ultimately led to the resignation of three employees, including a faculty member, a graduate student and Halford's widow. Investors in Rational Vaccines, including maverick Silicon Valley billionaire Peter Thiel, demanded that all FDA rules must be followed in future tests.
"Trials have to yield data that can be submitted to the FDA, which means certain requirements have to be met," says Jeffrey Kahn, a bioethicist at Johns Hopkins University in Baltimore. "These were renegade researchers who exposed people to unnecessary risks, which was hugely irresponsible. I don't know what they expected to do with the research. It was a waste of money and generated data that can't be used because no regulator would accept it."
But this story illuminates both sides of a thorny issue. On one side are scientists and government regulators with legitimate safety concerns who want to protect volunteers from very real risks—people have died even in closely monitored clinical trials. On the other, are desperate patients and a dying scientist willing to go rogue in a foreign country where there is far less regulatory scrutiny. "It's a balancing act," says Jennifer Miller, a medical ethicist at New York University and president of Bioethics International. "You really need to protect participants but you also want access to safe therapies."
"Safety is important, but being too cautious kills people, too—allowing them to just die without intervention seems to be the biggest harm."
This requirement—that tests show a drug is safe and effective before it can win regulatory approval--dates back to 1962, when the sedative thalidomide was shown to have caused thousands of birth defects in Europe. But clinical trials can be costly and often proceed at a glacial pace. Typically, companies shell out more than $2.5 billion over the course of the decade it normally takes to shepherd a new treatment through the three phases of testing before it wins FDA approval, according to a 2014 study by the Tufts Center for the Study of Drug Development. Yet only 11.8 percent of experimental therapies entering clinical tests eventually cross the finish line.
The upshot is that millions can suffer and thousands of people may die awaiting approvals for life saving drugs, according to Elizabeth Parrish, the founder and CEO of BioViva, a Seattle-based biotech that aims to provide data collection platforms to scientists doing overseas tests. "Going offshore to places where it's legal to take a therapeutic can created expedited routes for patients to get therapies for which there is a high level of need," she says. "Safety is important, but being too cautious kills people, too—allowing them to just die without intervention seems to be the biggest harm."
Parrish herself was frustrated with the slow pace of gene therapy trials; scientists worried about the risks associated with fixing mutant DNA. To prove a point, she traveled to a clinic in Colombia in 2015 where she was injected with two gene therapies that aim to improve muscle function and lengthen telomeres, the caps on the end of chromosomes that are linked to aging and genetic diseases. Six months later, the therapy seemed to have worked—her muscle mass had increased and her telomeres had grown by 9 percent, the equivalent of turning back 20 years of aging, according to her own account. Yet the treatments are still unavailable here in the U.S.
In the past decade, Latin American countries like Columbia, and Mexico in particular, have become an increasingly attractive test destination for multi-national drug companies and biotechs because of less red tape.
In the past decade, Latin American countries like Columbia, and Mexico in particular, have become an increasingly attractive test destination for multi-national drug companies and biotechs because of less red tape around testing emerging new science, like gene therapies or stem cells. Plus, clinical trials are cheaper to conduct, it's easier to recruit volunteers, especially ones who are treatment naïve, and these human tests can reveal whether local populations actually respond to a particular therapy. "We do have an exhaustive framework for running clinical trials that are aligned with international requirements," says Ernesto Albaga, an attorney with Hogan Lovells in Mexico City who specializes in the life sciences. "But our environment is still not as stringent as it is in other places, like the U.S."
The fact is American researchers are increasingly testing experimental drugs outside of the U.S., although virtually all of them are monitored by local scientists who serve as co-investigators. In 2017 alone, more than 86 percent of experimental drugs seeking FDA approval have been tested, at least in part, in foreign countries, like Mexico, China, Russia, Poland and South Africa, according to an analysis by STAT. However, in places without strict oversight, such as Russia and Georgia, results may be fraudulent, according to one 2017 report in the New England Journal of Medicine. And in developing countries, the poor can become guinea pigs. In the early 2000s, for example, a test in Uganda of an AIDS drug resulted in thousands of unreported serious adverse reactions and 14 deaths; in India, eight volunteers died during a test of the anti-clotting drug, Streptokinase—and test subjects didn't even know they were part of a clinical trials.
Still, "the world is changing," concludes Dr. Jennifer Miller of NYU. "We need to figure out how to get safe and effective drugs to patients more quickly without sacrificing too much protection."
Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
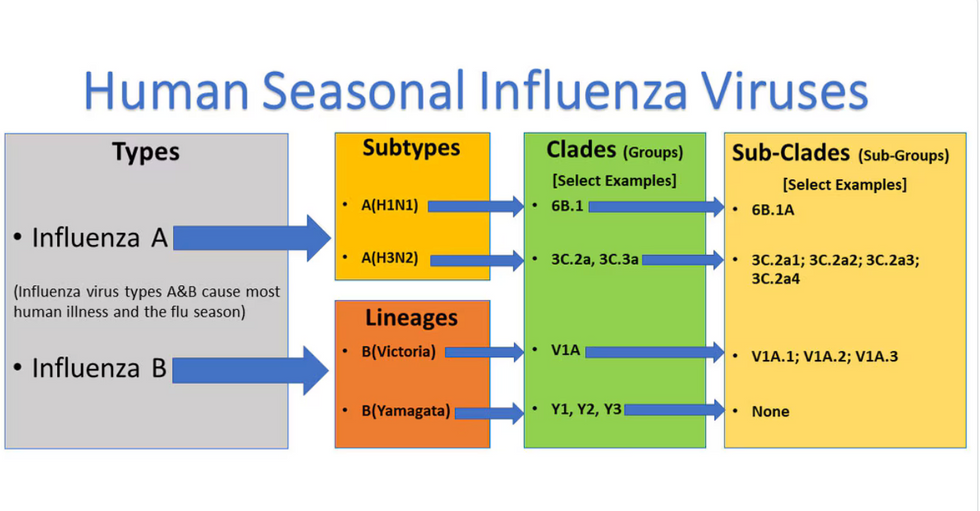
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.

