Where Are the Lab-Grown Replacement Organs?
A futuristic rendering of a scientist holding a pair of lungs in a glass dish.
The headline blared from newspapers all the way back in 2006: "First Lab-Grown Organs Implanted in Humans!" A team from Wake Forest University had biopsied cells from the bladders of patients with spina bifida and used them to create brand new full-size bladders, which they then implanted. Although the bladders had to be emptied via catheter, they were still functioning a few years after implantation, and the public grew confident that doctors had climbed an intermediary step on the way to the medicine of science fiction. Ten years later, though, more than 20 people a day are still dying while waiting for an organ transplant, which leads to a simple question: Where are our fake organs?
"We can make small organs and tissues but we can't make larger ones."
Not coming anytime soon, unfortunately. The company that was created to transition Wake Forest's bladders to the market failed. And while there are a few simple bioengineered skins and cartilages already on the market, they are hardly identical to the real thing. Something like a liver could take another 20 to 25 years, says Shay Soker, professor at Wake Forest's Institute for Regenerative Medicine. "The first barrier is the technology: We can make small organs and tissues but we can't make larger ones," he says. "Also there are several cell types or functions that you can reliably make from stem cells, but not all of them, so the technology of stem cells has to catch up with what the body can do." Finally, he says, you have support the new organ inside the body, providing it with a circulatory and nervous system and integrating it with the immune system.
While these are all challenging problems, circulation appears to be the most intractable. "Tissue's not able to survive if the cells don't have oxygen, and the bigger it gets, the more complex vasculature you need to keep that alive," says Chiara Ghezzi, research professor in the Tufts University Department of Biomedical Engineering. "Vasculature is highly organized in the body. It has a hierarchical structure, with different branches that have different roles depending on where they are." So far, she says, researchers have had trouble scaling up from capillaries to larger vessels that could be grafted onto blood vessels in a patient's body.
"The FDA is still getting its hands and minds around the field of tissue engineering."
Last, but hardly least, is the question of FDA approval. Lab-grown organs are neither drugs nor medical devices, and the agency is not set up to quickly or easily approve new technologies that don't fit into current categories. "The FDA is still getting its hands and minds around the field of tissue engineering," says Soker. "They were not used to that… so it requires the regulatory and financial federal agencies to really help and support these initiatives."
A pencil eraser-size model of the human brain is now being used for drug development and research.
If all of this sounds discouraging, it's worth mentioning some of the incredible progress the field has made since the first strides toward lab-grown organs began nearly 30 years ago: Though full-size replacement organs are still decades away, many labs have diverted their resources into what they consider an intermediate step, developing miniature organs and systems that can be used for drug development and research. This platform will yield more relevant results (Imagine! Testing cardiovascular drugs on an actual human heart!) and require the deaths of far fewer animals. And it's already here: Two years ago, scientists at Ohio State University developed a pencil eraser-size model of the human brain they intend to use for this exact purpose.
Perhaps the most exciting line of research these days is one that at first doesn't seem to have anything to do with bioengineered organs at all. Along with his colleagues, Chandan Sen, Director of the Center for Regenerative Medicine and Cell-based Therapies at Ohio State University, has developed a nanoscale chip that can turn any cell in the body into any other kind of cell—reverting fully differentiated adult cells into, essentially, stem cells, which can then grow into any tissue you want. Sen has used his chip to reprogram skin cells in the bodies of mice into neurons to help them recover from strokes, and blood vessels to save severe leg injuries. "There's this concept of a bioreactor, where you convince an organ to grow outside the body. They're getting more and more sophisticated over time. But to my mind it will never match the sophistication or complexity of the human body," Sen says. "I believe that in order to have an organ that behaves the way you want it to in the live body, you must use the body itself as a bioreactor, not a bunch of electronic gadgetry." There you have it, the next step in artificial organ manufacture is as crazy as it is intuitive: Grow it back where it was in the first place.
Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
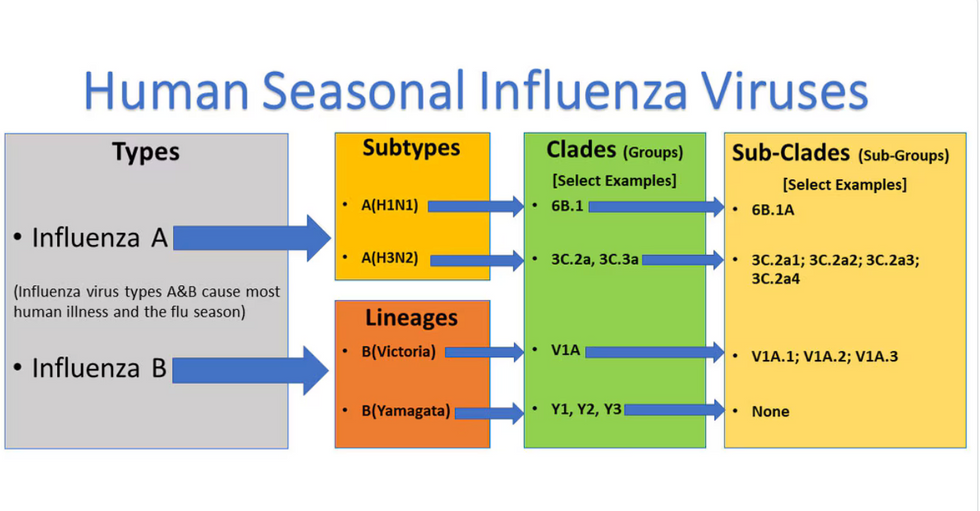
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.

