CRISPR base editing gives measure of hope to people with muscular dystrophy

Next year, a neurologist will test CRISPR base editing in a trial of five people with muscular dystrophy to see if their muscles accept corrected cells and whether they multiply and take over the function of damaged cells.
When Martin Weber climbs the steps to his apartment on the fifth floor in Munich, an attentive observer might notice that he walks a little unevenly. “That’s because my calf muscles were the first to lose strength,” Weber explains.
About three years ago, the now 19-year-old university student realized that he suddenly had trouble keeping up with his track team at school. At tennis tournaments, he seemed to lose stamina after the first hour. “But it was still within the norm,” he says. “So it took a while before I noticed something was seriously wrong.” A blood test showed highly elevated liver markers. His parents feared he had liver cancer until a week-long hospital visit and scores of tests led to a diagnosis: hereditary limb-girdle muscular dystrophy, an incurable genetic illness that causes muscles to deteriorate.
As you read this text, you will surely use several muscles without being aware of them: Your heart muscle pumps blood through your arteries, your eye muscles let you follow the words in this sentence, and your hand muscles hold the tablet or cell phone. Muscles make up 40 percent of your body weight; we usually have 656 of them. Now imagine they are slowly losing their strength. No training, no protein shake can rebuild their function.
This is the reality for most people in Simone Spuler’s outpatient clinic at the Charité Hospital in Berlin, Germany: Almost all of her 2,500 patients have muscular dystrophy, a progressive illness striking mostly young people. Muscle decline leads to a wheelchair and, eventually, an early death due to a heart attack or the inability to breathe. In Germany alone, 300,000 people live with this illness, the youngest barely a year old. The CDC estimates that its most common form, Duchenne, affects 1 in every 3,500 to 6,000 male births each year in the United States.
The devastating progression of the disease is what motivates Spuler and her team of 25 scientists to find a cure. In 2019, they made a spectacular breakthrough: For the first time, they successfully used mRNA to introduce the CRISPR-Cas9 tool into human muscle stem cells to repair the dystrophy. “It’s really just one tiny molecule that doesn’t work properly,” Spuler explains.
CRISPR-Cas9 is a technology that lets scientists select and alter parts of the genome. It’s still comparatively new but has advanced quickly since its discovery in the early 2010s. “We now have the possibility to repair certain mutations with genetic editing,” Spuler says. “It’s pure magic.”
She projects a warm, motherly air and a professional calm that inspires trust from her patients. She needs these qualities because the 60-year-old neurologist has one of the toughest jobs in the world: All day long, patients with the incurable diagnosis of muscular dystrophy come to her clinic, and she watches them decline over the years. “Apart from physiotherapy, there is nothing we can recommend right now,” she says. That motivated her early in her career, when she met her first patients at the Max Planck Institute for Neurobiology near Munich in the 1990s. “I knew I had 30, 40 years to find something.”
She learned from the luminaries of her profession with postdocs at the University of California San Diego, Harvard and Johns Hopkins, before serving as a clinical fellow at the Mayo Clinic. In 2005, the Charité offered her the opportunity to establish a specialized clinic for myasthenia, or muscular weakness. An important influence on Spuler, she says, has been the French microbiologist Emmanuelle Charpentier, who received the Nobel Prize in 2020 along with Jennifer Doudna for their CRISPR research, and has worked in Berlin since 2015.
When CRISPR was first introduced, it was mainly used to cut through DNA. However, the cut can lead to undesired side effects. For the muscle stem cells, Spuler now uses a base editor to repair the damaged molecule with super fine scissors or tweezers.

“Apart from physiotherapy, there is nothing we can recommend right now,” Spuler says about her patients with limb-girdle muscular dystrophy.
Pablo Castagnola
Last year, she proved that the method works in mice. Injecting repaired cells into the rodents led to new muscle fibers and, in 2021 and 2022, she passed the first safety meetings with the Paul-Ehrlich Institute, which is responsible for approving human gene editing trials in Germany. She raised the nearly four million Euros needed to test the new method in the first clinical trial in humans with limb-girdle muscular dystrophy, beginning with one muscle that can easily be measured, such as the biceps.
This spring, Weber and his parents drove the 400 miles from Munich to Berlin. At Spuler’s lab, her team took a biopsy from muscles in his left arm. The first two steps – extraction and repair in a culture dish – went according to plan; Spuler was able to repair the mutation in Weber’s cells outside his body.
Next year, Weber will be the youngest participant when Spuler starts to test the method in a trial of five people “in vivo,” inside their bodies. This will be the real moment of truth: Will the participants’ muscles accept the corrected cells? Will the cells multiply and take over the function of damaged cells, just like Spuler was able to do in her lab with the rodents?
The effort is costly and complex. “The biggest challenge is to make absolutely sure that we don’t harm the patient,” Spuler says. This means scanning their entire genomes, “so we don’t accidentally damage or knock out an important gene.”
Weber, who asked not to be identified by his real name, is looking forward to the trial and he feels confident that “the risks are comparatively small because the method will only be applied to one muscle. The worst that can happen is that it doesn’t work. But in the best case, the muscle function will improve.”
He was so impressed with the Charité scientists that he decided to study biology at his university. He’s read extensively about CRISPR, so he understands why he has three healthy siblings. “That’s the statistics,” the biologist in training explains. “You get two sets of genes from each parent, and you have to get two faulty mutations to have muscular dystrophy. So we fit the statistics exactly: One of us four kids inherited the mutation.”
It was his mother, a college teacher, and father, a physicist by training, who heard about Spuler’s research. Even though Weber does not live at home anymore, having a chronically ill son is nearly a full-time job for his mother, Annette. The Berlin visit and the trial are financed separately through private sponsors, but the fights with Weber’s health insurance are frustrating and time-consuming. “Physiotherapy is the only thing that helps a bit,” Weber says, “and yet, they fought us on approving it every step of the way.”
Spuler does not want to evoke unrealistic expectations. “Patients who are wheelchair-bound won’t suddenly get up and walk."
Her son continues to exercise as much as possible. Riding his bicycle to the university has become too difficult, so he got an e-scooter. He had to give up competitive tennis because he does not have the stamina for a two-hour match, but he can still play with his dad or his buddies for an hour. His closest friends know about the diagnosis. “They help me, for instance, to lift something heavy because I can’t do that anymore,” Weber says.
The family was elated to find medical support at the Munich Muscle Center by the German Alliance for Muscular Patients and then at Spuler’s clinic in Berlin. “When you hear that this is a progressive illness with no chance of improvement, your world collapses as a parent,” Annette Weber says. “And then all of a sudden, there is this woman who sees scientific progress as an opportunity. Even just to be able to participate in the study is fantastic.”
Spuler does not want to evoke unrealistic expectations. “Patients who are wheelchair-bound won’t suddenly get up and walk,” she says. After all, she will start by applying the gene editor to only one muscle, “but it would be a big step if even a small muscle that is essential to grip something, or to swallow, regains function.”
Weber agrees. “I understand that I won’t regain 100 percent of my muscle function but even a small improvement or at least halting the deterioration is the goal.”
And yet, Spuler and others are ultimately searching for a true solution. In a separate effort, Massachusetts-based biotech company Sarepta announced this month it will seek expedited regulators’ approval to treat Duchenne patients with its investigational gene therapy. Unlike Spuler’s methods, Sarepta focuses specifically on the Duchenne form of muscular dystrophy, and it uses an adeno-assisted virus to deliver the therapy.
Spuler’s vision is to eventually apply gene editing to the entire body of her patients. To speed up the research, she and a colleague founded a private research company, Myopax. If she is able to prove that the body accepts the edited cells, the technique could be used for other monogenetic illnesses as well. “When we speak of genetic editing, many are scared and say, oh no, this is God’s work,” says Spuler. But she sees herself as a mechanic, not a divine being. “We really just exchange a molecule, that’s it.”
If everything goes well, Weber hopes that ten years from now, he will be the one taking biopsies from the next generation of patients and repairing their genes.
New tech for prison reform spreads to 11 states
The U.S. has the highest incarceration rate in the world, costing $182 billion per year, partly because its antiquated data systems often fail to identify people who should be released. A tech nonprofit is trying to change that.
A new non-profit called Recidiviz is using data technology to reduce the size of the U.S. criminal justice system. The bi-coastal company (SF and NYC) is currently working with 11 states to improve their systems and, so far, has helped remove nearly 69,000 people — ones left floundering in jail or on parole when they should have been released.
“The root cause is fragmentation,” says Clementine Jacoby, 31, a software engineer who worked at Google before co-founding Recidiviz in 2019. In the 1970s and 80s, the U.S. built a series of disconnected data systems, and this patchwork is still being used by criminal justice authorities today. It requires parole officers to manually calculate release dates, leading to errors in many cases. “[They] have done everything they need to do to earn their release, but they're still stuck in the system,” Jacoby says.
Recidiviz has built a platform that connects the different databases, with the goal of identifying people who are already qualified for release but remain behind bars or on supervision. “Think of Recidiviz like Google Maps,” says Jacoby, who worked on Maps when she was at the tech giant. Google Maps takes in data from different sources – satellite images, street maps, local business data — and organizes it into one easy view. “Recidiviz does something similar with criminal justice data,” Jacoby explains, “making it easy to identify people eligible to come home or to move to less intensive levels of supervision.”
People like Jacoby’s uncle. His experience with incarceration is what inspired her passion for criminal justice reform in the first place.
The problems are vast
The U.S. has the highest incarceration rate in the world — 2 million people according to the watchdog group, Prison Policy Initiative — at a cost of $182 billion a year. The numbers could be a lot lower if not for an array of problems including inaccurate sentencing calculations, flawed algorithms and parole violations laws.
Sentencing miscalculations
To determine eligibility for release, the current system requires corrections officers to check 21 different requirements spread across five different databases for each of the 90 to 100 people under their supervision. These manual calculations are time prohibitive, says Jacoby, and fall victim to human error.
In addition, Recidiviz found that policies aimed at helping to reduce the prison population don’t always work correctly. A key example is time off for good behavior laws that allow inmates to earn one day off for every 30 days of good behavior. Some states' data systems are built to calculate time off as one day per month of good behavior, rather than per day. Over the course of a decade-long sentence, Jacoby says these miscalculations can lead to a huge discrepancy in the calculated release data and the actual release date.
Algorithms
Commercial algorithm-based software systems for risk assessment continue to be widely used in the criminal justice system, even though a 2018 study published in Science Advances exposed their limitations. After the study went viral, it took three years for the Justice Department to issue a report on their own flawed algorithms used to reduce the federal prison population as part of the 2018 First Step Act. The program, it was determined, overestimated the risk of putting inmates of color into early-release programs.
Despite its name, Recidiviz does not build these types of algorithms for predicting recidivism, or whether someone will commit another crime after being released from prison. Rather, Jacoby says the company’s "descriptive analytics” approach is specifically intended to weed out incarceration inequalities and avoid algorithmic pitfalls.
Parole violation laws
Research shows that 350,000 people a year — about a quarter of the total prison population — are sent back not because they’ve committed another crime, but because they’ve broken a specific rule of their probation. “Things that wouldn't send you or I to prison, but would send someone on parole,” such as crossing county lines or being in the presence of alcohol when they shouldn’t be, are inflating the prison population, says Jacoby.
It’s personal for the co-founder and CEO
“I grew up with an uncle who went into the prison system,” Jacoby says. At 19, he was sentenced to ten years in prison for a non-violent crime. A few months after being released from jail, he was sent back for a non-violent parole violation.
“For my family, the fact that one in four prison admissions are driven not by a crime but by someone who's broken a rule on probation and parole was really profound because that happened to my uncle,” Jacoby says. The experience led her to begin studying criminal justice in high school, then college. She continued her dive into how the criminal justice system works as part of her Passion Project while at Google, a program that allows employees to spend 20 percent of their time on pro-bono work. Two colleagues whose family members had also been stuck in the system joined her.
As part of the project, Jacoby interviewed hundreds of people involved in the criminal justice system. “Those on the right, those on the left, agreed that bad data was slowing down reform,” she says. Their research brought them to North Dakota where they began to understand the root of the problem. The corrections department is making “huge, consequential decisions every day [without] … the data,” Jacoby says. In a new video by Recidiviz not yet released, Jacoby recounts her exchange with the state’s director of corrections who told her, “‘It’s not that we have the data and we just don’t know how to make it public; we don’t have the information you think we have.'"
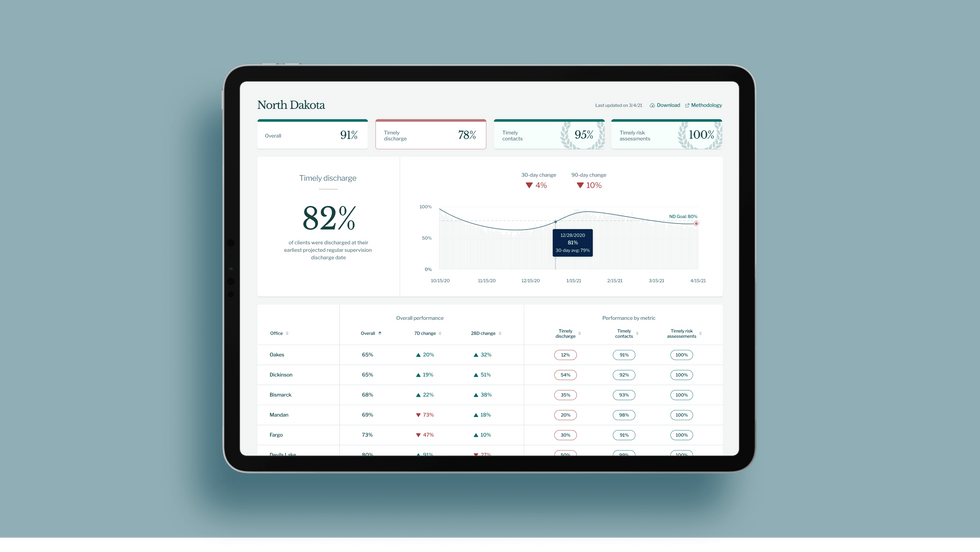
A mock-up (with fake data) of the types of dashboards and insights that Recidiviz provides to state governments.
Recidiviz
As a software engineer, Jacoby says the comment made no sense to her — until she witnessed it first-hand. “We spent a lot of time driving around in cars with corrections directors and parole officers watching them use these incredibly taxing, frankly terrible, old data systems,” Jacoby says.
As they weeded through thousands of files — some computerized, some on paper — they unearthed the consequences of bad data: Hundreds of people in prison well past their release date and thousands more whose release from parole was delayed because of minor paperwork issues. They found individuals stuck in parole because they hadn’t checked one last item off their eligibility list — like simply failing to provide their parole officer with a paystub. And, even when parolees advocated for themselves, the archaic system made it difficult for their parole officers to confirm their eligibility, so they remained in the system. Jacoby and her team also unpacked specific policies that drive racial disparities — such as fines and fees.
The Solution
It’s more than a trivial technical challenge to bring the incomplete, fragmented data onto a 21st century data platform. It takes months for Recidiviz to sift through a state’s information systems to connect databases “with the goal of tracking a person all the way through their journey and find out what’s working for 18- to 25-year-old men, what’s working for new mothers,” explains Jacoby in the video.
TED Talk: How bad data traps people in the U.S. justice system

TED Fellow Clementine Jacoby's TED Talk went live on Jan. 13. It describes how we can fix bad data in the criminal justice system, "bringing thousands of people home, reducing costs and improving public safety along the way."
Clementine Jacoby • TED2022
Ojmarrh Mitchell, an associate professor in the School of Criminology and Criminal Justice at Arizona State University, who is not involved with the company, says what Recidiviz is doing is “remarkable.” His perspective goes beyond academic analysis. In his pre-academic years, Mitchell was a probation officer, working within the framework of the “well known, but invisible” information sharing issues that plague criminal justice departments. The flexibility of Recidiviz’s approach is what makes it especially innovative, he says. “They identify the specific gaps in each jurisdiction and tailor a solution for that jurisdiction.”
On the downside, the process used by Recidiviz is “a bit opaque,” Mitchell says, with few details available on how Recidiviz designs its tools and tracks outcomes. By sharing more information about how its actions lead to progress in a given jurisdiction, Recidiviz could help reformers in other places figure out which programs have the best potential to work well.
The eleven states in which Recidiviz is working include California, Colorado, Maine, Michigan, Missouri, Pennsylvania and Tennessee. And a pilot program launched last year in Idaho, if scaled nationally, with could reduce the number of people in the criminal justice system by a quarter of a million people, Jacoby says. As part of the pilot, rather than relying on manual calculations, Recidiviz is equipping leaders and the probation officers with actionable information with a few clicks of an app that Recidiviz built.
Mitchell is disappointed that there’s even the need for Recidiviz. “This is a problem that government agencies have a responsibility to address,” he says. “But they haven’t.” For one company to come along and fill such a large gap is “remarkable.”
How Leqembi became the biggest news in Alzheimer’s disease in 40 years, and what comes next
Betsy Groves, 73, with her granddaughter. Groves learned in 2021 that she has Alzheimer's disease. She hopes to take Leqembi, a drug approved by the FDA last week.
A few months ago, Betsy Groves traveled less than a mile from her home in Cambridge, Mass. to give a talk to a bunch of scientists. The scientists, who worked for the pharmaceutical companies Biogen and Eisai, wanted to know how she lived her life, how she thought about her future, and what it was like when a doctor’s appointment in 2021 gave her the worst possible news. Groves, 73, has Alzheimer’s disease. She caught it early, through a lumbar puncture that showed evidence of amyloid, an Alzheimer’s hallmark, in her cerebrospinal fluid. As a way of dealing with her diagnosis, she joined the Alzheimer’s Association’s National Early-Stage Advisory Board, which helped her shift into seeing her diagnosis as something she could use to help others.
After her talk, Groves stayed for lunch with the scientists, who were eager to put a face to their work. Biogen and Eisai were about to release the first drug to successfully combat Alzheimer’s in 40 years of experimental disaster. Their drug, which is known by the scientific name lecanemab and the marketing name Leqembi, was granted accelerated approval by the U.S. Food and Drug Administration last Friday, Jan. 6, after a study in 1,800 people showed that it reduced cognitive decline by 27 percent over 18 months.
It is no exaggeration to say that this result is a huge deal. The field of Alzheimer’s drug development has been absolutely littered with failures. Almost everything researchers have tried has tanked in clinical trials. “Most of the things that we've done have proven not to be effective, and it's not because we haven’t been taking a ton of shots at goal,” says Anton Porsteinsson, director of the University of Rochester Alzheimer's Disease Care, Research, and Education Program, who worked on the lecanemab trial. “I think it's fair to say you don't survive in this field unless you're an eternal optimist.”
As far back as 1984, a cure looked like it was within reach: Scientists discovered that the sticky plaques that develop in the brains of those who have Alzheimer’s are made up of a protein fragment called beta-amyloid. Buildup of beta-amyloid seemed to be sufficient to disrupt communication between, and eventually kill, memory cells. If that was true, then the cure should be straightforward: Stop the buildup of beta-amyloid; stop the Alzheimer’s disease.
It wasn’t so simple. Over the next 38 years, hundreds of drugs designed either to interfere with the production of abnormal amyloid or to clear it from the brain flamed out in trials. It got so bad that neuroscience drug divisions at major pharmaceutical companies (AstraZeneca, Pfizer, Bristol-Myers, GSK, Amgen) closed one by one, leaving the field to smaller, scrappier companies, like Cambridge-based Biogen and Tokyo-based Eisai. Some scientists began to dismiss the amyloid hypothesis altogether: If this protein fragment was so important to the disease, why didn’t ridding the brain of it do anything for patients? There was another abnormal protein that showed up in the brains of Alzheimer’s patients, called tau. Some researchers defected to the tau camp, or came to believe the proteins caused damage in combination.
The situation came to a head in 2021, when the FDA granted provisional approval to a drug called aducanumab, marketed as Aduhelm, against the advice of its own advisory council. The approval was based on proof that Aduhelm reduced beta-amyloid in the brain, even though one research trial showed it had no effect on people’s symptoms or daily life. Aduhelm could also cause serious side effects, like brain swelling and amyloid related imaging abnormalities (known as ARIA, these are basically micro-bleeds that appear on MRI scans). Without a clear benefit to memory loss that would make these risks worth it, Medicare refused to pay for Aduhelm among the general population. Two congressional committees launched an investigation into the drug’s approval, citing corporate greed, lapses in protocol, and an unjustifiably high price. (Aduhelm was also produced by the pharmaceutical company Biogen.)
To be clear, Leqembi is not the cure Alzheimer’s researchers hope for. While the drug is the first to show clear signs of a clinical benefit, the scientific establishment is split on how much of a difference Leqembi will make in the real world.
So far, Leqembi is like Aduhelm in that it has been given accelerated approval only for its ability to remove amyloid from the brain. Both are monoclonal antibodies that direct the immune system to attack and clear dysfunctional beta-amyloid. The difference is that, while that’s all Aduhelm was ever shown to do, Leqembi’s makers have already asked the FDA to give it full approval – a decision that would increase the likelihood that Medicare will cover it – based on data that show it also improves Alzheimer’s sufferer’s lives. Leqembi targets a different type of amyloid, a soluble version called “protofibrils,” and that appears to change the effect. “It can give individuals and their families three, six months longer to be participating in daily life and living independently,” says Claire Sexton, PhD, senior director of scientific programs & outreach for the Alzheimer's Association. “These types of changes matter for individuals and for their families.”
To be clear, Leqembi is not the cure Alzheimer’s researchers hope for. It does not halt or reverse the disease, and people do not get better. While the drug is the first to show clear signs of a clinical benefit, the scientific establishment is split on how much of a difference Leqembi will make in the real world. It has “a rather small effect,” wrote NIH Alzheimer’s researcher Madhav Thambisetty, MD, PhD, in an email to Leaps.org. “It is unclear how meaningful this difference will be to patients, and it is unlikely that this level of difference will be obvious to a patient (or their caregivers).” Another issue is cost: Leqembi will become available to patients later this month, but Eisai is setting the price at $26,500 per year, meaning that very few patients will be able to afford it unless Medicare chooses to reimburse them for it.
The same side effects that plagued Aduhelm are common in Leqembi treatment as well. In many patients, amyloid doesn’t just accumulate around neurons, it also forms deposits in the walls of blood vessels. Blood vessels that are shot through with amyloid are more brittle. If you infuse a drug that targets amyloid, brittle blood vessels in the brain can develop leakage that results in swelling or bleeds. Most of these come with no symptoms, and are only seen during testing, which is why they are called “imaging abnormalities.” But in situations where patients have multiple diseases or are prescribed incompatible drugs, they can be serious enough to cause death. The three deaths reported from Leqembi treatment (so far) are enough to make Thambisetty wonder “how well the drug may be tolerated in real world clinical practice where patients are likely to be sicker and have multiple other medical conditions in contrast to carefully selected patients in clinical trials.”
Porsteinsson believes that earlier detection of Alzheimer’s disease will be the next great advance in treatment, a more important step forward than Leqembi’s approval.
Still, there are reasons to be excited. A successful Alzheimer’s drug can pave the way for combination studies, in which patients try a known effective drug alongside newer, more experimental ones; or preventative studies, which take place years before symptoms occur. It also represents enormous strides in researchers’ understanding of the disease. For example, drug dosages have increased massively—in some cases quadrupling—from the early days of Alzheimer’s research. And patient selection for studies has changed drastically as well. Doctors now know that you’ve got to catch the disease early, through PET-scans or CSF tests for amyloid, if you want any chance of changing its course.
Porsteinsson believes that earlier detection of Alzheimer’s disease will be the next great advance in treatment, a more important step forward than Leqembi’s approval. His lab already uses blood tests for different types of amyloid, for different types of tau, and for measures of neuroinflammation, neural damage, and synaptic health, but commercially available versions from companies like C2N, Quest, and Fuji Rebio are likely to hit the market in the next couple of years. “[They are] going to transform the diagnosis of Alzheimer's disease,” Porsteinsson says. “If someone is experiencing memory problems, their physicians will be able to order a blood test that will tell us if this is the result of changes in your brain due to Alzheimer's disease. It will ultimately make it much easier to identify people at a very early stage of the disease, where they are most likely to benefit from treatment.”
Learn more about new blood tests to detect Alzheimer's
Early detection can help patients for more philosophical reasons as well. Betsy Groves credits finding her Alzheimer’s early with giving her the space to understand and process the changes that were happening to her before they got so bad that she couldn’t. She has been able to update her legal documents and, through her role on the Advisory Group, help the Alzheimer’s Association with developing its programs and support services for people in the early stages of the disease. She still drives, and because she and her husband love to travel, they are hoping to get out of grey, rainy Cambridge and off to Texas or Arizona this spring.
Because her Alzheimer’s disease involves amyloid deposits (a “substantial portion” do not, says Claire Sexton, which is an additional complication for research), and has not yet reached an advanced stage, Groves may be a good candidate to try Leqembi. She says she’d welcome the opportunity to take it. If she can get access, Groves hopes the drug will give her more days to be fully functioning with her husband, daughters, and three grandchildren. Mostly, she avoids thinking about what the latter stages of Alzheimer’s might be like, but she knows the time will come when it will be her reality. “So whatever lecanemab can do to extend my more productive ways of engaging with relationships in the world,” she says. “I'll take that in a minute.”

