New Options Are Emerging in the Search for Better Birth Control
A decade ago, Elizabeth Summers' options for birth control suddenly narrowed. Doctors diagnosed her with Factor V Leiden, a rare genetic disorder, after discovering blood clots in her lungs. The condition increases the risk of clotting, so physicians told Summers to stay away from the pill and other hormone-laden contraceptives. "Modern medicine has generally failed to provide me with an effective and convenient option," she says.
But new birth control options are emerging for women like Summers. These alternatives promise to provide more choices to women who can't ingest hormones or don't want to suffer their unpleasant side effects.
These new products have their own pros and cons. Still, doctors are welcoming new contraceptives following a long drought in innovation. "It's been a long time since we've had something new in the world of contraception," says Heather Irobunda, an obstetrician and gynecologist at NYC Health and Hospitals.
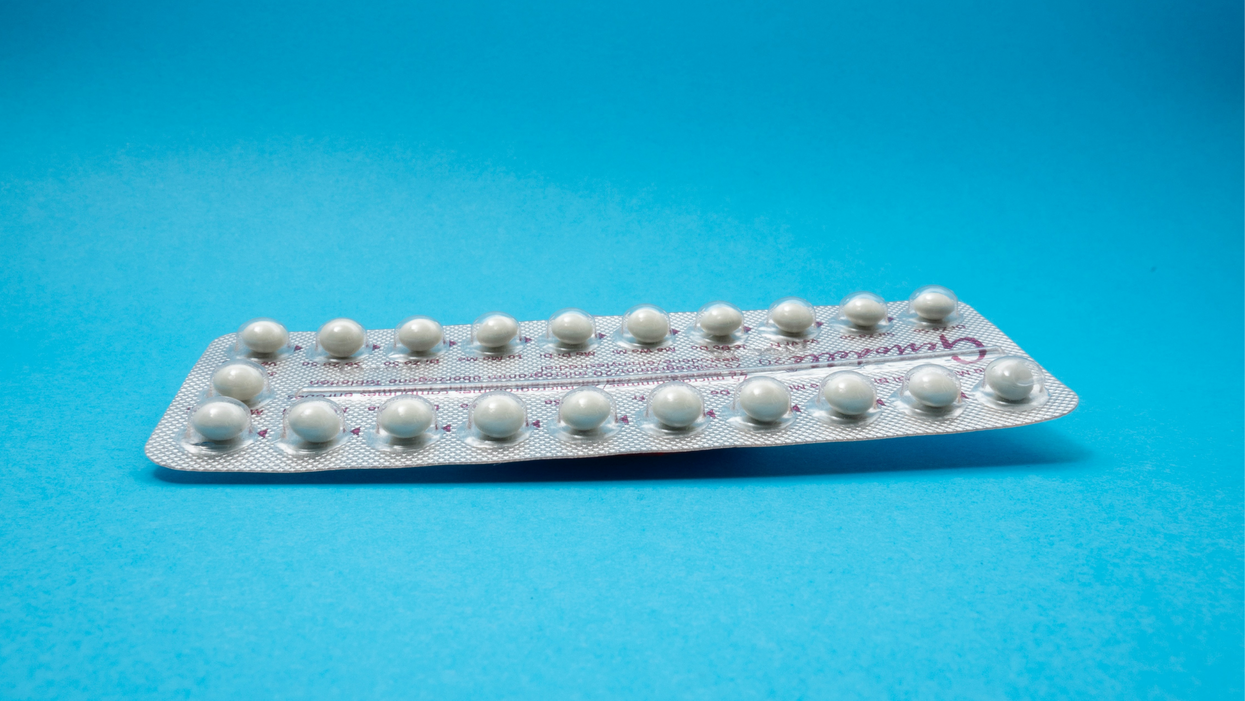
On social media, Irobunda often fields questions about one of these new options, a lubricating gel called Phexxi. San Diego-based Evofem, the company behind Phexxi, has been advertising the product on Hulu and Instagram after the gel was approved by the Food and Drug Administration in May 2020. The company's trendy ads target women who feel like condoms diminish the mood, but who also don't want to mess with an IUD or hormones.
Here's how it works: Phexxi is inserted via a tampon-like device up to an hour before sex. The gel regulates vaginal pH — essentially, the acidity levels — in a range that's inhospitable to sperm. It sounds a lot like spermicide, which is also placed in the vagina prior to sex to prevent pregnancy. But spermicide can damage the vagina's cell walls, which can increase the risk of contracting sexually transmitted diseases.
"Not only is innovation needed, but women want a non-hormonal option."
Phexxi isn't without side effects either. The most common one is vaginal burning, according to a late-stage trial. It's also possible to develop a urinary tract infection while using the product. That same study found that during typical use, Phexxi is about 86 percent effective at preventing pregnancy. The efficacy rate is comparable to condoms but lower than birth control pills (91 percent) and significantly lower than an IUD (99 percent).
Phexxi – which comes in a pack of 12 – represents a tiny but growing part of the birth control market. Pharmacies dispensed more than 14,800 packs from April through June this year, a 65 percent increase over the previous quarter, according to data from Evofem.
"We've been able to demonstrate that not only is innovation needed, but women want a non-hormonal option," says Saundra Pelletier, Evofem's CEO.
Beyond contraception, the company is carrying out late-stage tests to gauge Phexxi's effectiveness at preventing the sexually transmitted infections chlamydia and gonorrhea.

Phexxi is inserted via a tampon-like device up to an hour before sex.
Phexxi
A New Pill
The first birth control pill arrived in 1960, combining the hormones estrogen and progestin to stop sperm from joining with an egg, giving women control over their fertility. Subsequent formulations sought to ease side effects, by way of lower amounts of estrogen. But some women still experience headaches and nausea – or more serious complications like blood clots. On social media, women recently noted that birth control pills are much more likely to cause blood clots than Johnson & Johnson's COVID-19 vaccine that was briefly paused to evaluate the risk of clots in women under age 50. What will it take, they wondered, for safer birth control?
Mithra Pharmaceuticals of Belgium sought to create a gentler pill. In April, the FDA approved Mithra's Nextstellis, which includes a naturally occurring estrogen, the first new estrogen in the U.S. in 50 years. Nextstellis selectively acts on tissues lining the uterus, while other birth control pills have a broader target.
A Phase 3 trial showed a 98 percent efficacy rate. Andrew London, an obstetrician and gynecologist, who practices at several Maryland hospitals, says the results are in line with some other birth control pills. But, he added, early studies indicate that Nextstellis has a lower risk of blood clotting, along with other potential benefits, which additional clinical testing must confirm.
"It's not going to be worse than any other pill. We're hoping it's going to be significantly better," says London.
The estrogen in Nexstellis, called estetrol, was skipped over by the pharmaceutical industry after its discovery in the 1960s. Estetrol circulates between the mother and fetus during pregnancy. Decades later, researchers took a new look, after figuring out how to synthesize estetrol in a lab, as well as produce estetrol from plants.
"That allowed us to really start to investigate the properties and do all this stuff you have to do for any new drug," says Michele Gordon, vice president of marketing in women's health at Mayne Pharma, which licensed Nextstellis.
Bonnie Douglas, who followed the development of Nextstellis as part of a search for better birth control, recently switched to the product. "So far, it's much more tolerable," says Douglas. Previously, the Midwesterner was so desperate to find a contraceptive with fewer side effects that she turned to an online pharmacy to obtain a different birth control pill that had been approved in Canada but not in the U.S.
Contraceptive Access
Even if a contraceptive lands FDA approval, access poses a barrier. Getting insurers to cover new contraceptives can be difficult. For the uninsured, state and federal programs can help, and companies should keep prices in a reasonable range, while offering assistance programs. So says Kelly Blanchard, president of the nonprofit Ibis Reproductive Health. "For innovation to have impact, you want to reach as many folks as possible," she says.
In addition, companies developing new contraceptives have struggled to attract venture capital. That's changing, though.
In 2015, Sabrina Johnson founded DARÉ Bioscience around the idea of women's health. She estimated the company would be fully funded in six months, based on her track record in biotech and the demand for novel products.
But it's been difficult to get male investors interested in backing new contraceptives. It took Johnson two and a half years to raise the needed funds, via a reverse merger that took the company public. "There was so much education that was necessary," Johnson says, adding: "The landscape has changed considerably."
Johnson says she would like to think DARÉ had something to do with the shift, along with companies like Organon, a spinout of pharma company Merck that's focused on reproductive health. In surveying the fertility landscape, DARÉ saw limited non-hormonal options. On-demand options – like condoms – can detract from the moment. Copper IUDs must be inserted by a doctor and removed if a woman wants to return to fertility, and this method can have onerous side effects.
So, DARÉ created Ovaprene, a hormone-free device that's designed to be inserted into the vagina monthly by the user. The mesh product acts as a barrier, while releasing a chemical that immobilizes sperm. In an early study, the company reported that Ovaprene prevented almost all sperm from entering the cervical canal. The results, DARÉ believes, indicate high efficacy.
A late-stage study, slated to kick off next year, will be the true judge. Should Ovaprene eventually win regulatory approval, drug giant Bayer will handle commercializing the device.
Other new forms of birth control in development are further out, and that's assuming they perform well in clinical trials. Among them: a once-a-month birth control pill, along with a male version of the birth control pill. The latter is often brought up among women who say it's high time that men take a more proactive role in birth control.
For Summers, her search for a safe and convenient birth control continues. She tried Phexxi, which caused irritation. Still, she's excited that a non-hormonal option now exists. "I'm sure it will work for others," she says.
A Futuristic Suicide Machine Aims to End the Stigma of Assisted Dying
A prototype of the Sarco is currently on display at Venice Design.
Bob Dent ended his life in Perth, Australia in 1996 after multiple surgeries to treat terminal prostate cancer had left him mostly bedridden and in agony.
Although Dent and his immediate family believed it was the right thing to do, the physician who assisted in his suicide – and had pushed for Australia's Northern Territory to legalize the practice the prior year – was deeply shaken.
"You climb in, you are going somewhere, you are leaving, and you are saying goodbye."
"When you get to know someone pretty well, and they set a date to have lunch with you and then have them die at 2 p.m., it's hard to forget," recalls Philip Nitschke.
Nitschke remembers being highly anxious that the device he designed – which released a fatal dose of Nembutal into a patient's bloodstream after they answered a series of questions on a laptop computer to confirm consent – wouldn't work. He was so alarmed by the prospect he recalls his shirt being soaked through with perspiration.
Known as a "Deliverance Machine," it was comprised of the computer, attached by a sheet of wiring to an attache case containing an apparatus for delivering the Nembutal. Although gray, squat and grimly businesslike, it was vastly more sophisticated than Jack Kevorkian's Thanatron – a tangle of tubes, hooks and vials redolent of frontier dentistry.
The Deliverance Machine did work – for Dent and three other patients of Nitschke. However, it remained far from reassuring. "It's not a very comfortable feeling, having a little suitcase and going around to people," he says. "I felt a little like an executioner."
The furor caused in part by Nitschke's work led to Australia's federal government banning physician-assisted suicide in 1997. Nitschke went on to co-found Exit International, one of the foremost assisted suicide advocacy groups, and relocated to the Netherlands.
Exit International recently introduced its most ambitious initiative to date. It's called the Sarco — essentially the Eames lounger of suicide machines. A prototype is currently on display at Venice Design, an adjunct to the Biennale.
Sheathed in a soothing blue coating, the Sarco prototype contains a window and pivots on a pedestal to allow viewing by friends and family. Its close quarters means the opening of a small canister of liquid nitrogen would cause quick and painless asphyxiation. Patrons with second thoughts can press a button to cancel the process.
"The sleek and colorful death-pod looks like it is about to whisk you away to a new territory, or that it just landed after being launched from a Star Trek federation ship," says Charles C. Camosy, associate professor of theological and social ethics at Fordham University in New York City, in an email. Camosy, who has profound misgivings about such a device, was not being complimentary.
Nitschke's goal is to de-medicalize assisted suicide, as liquid nitrogen is readily available. But he suggests employing a futuristic design will also move debate on the issue forward.
"You pick the time...have the party and people come around. You climb in, you are going somewhere, you are leaving, and you are saying goodbye," he says. "It lends itself to a sense of occasion."
Assisted suicide is spreading in developed countries, but very slowly. It was legalized again in Australia just last June, but only in one of its six states. It is legal throughout Canada and in nine U.S. states.
Although the process is outlawed throughout much of Europe, nations permitting it have taken a liberal approach. Euthanasia — where death may be instigated by an assenting physician at a patient's request — is legal in both Belgium and the Netherlands. A terminal illness is not required; a severe disability or a condition causing profound misery may suffice.
Only Switzerland permits suicide with non-physician assistance regardless of an individual's medical condition. David Goodall, a 104-year Australian scientist, traveled 8,000 miles to Basel last year to die with Exit International's assistance. Goodall was in good health for his age and his mind was needle sharp; at a news conference the day before he passed, he thoughtfully answered questions and sang Beethoven's "Ode to Joy" from memory. He simply believed he had lived long enough and wanted to avoid a diminishing quality of life.
"Dying is not a medical process, and if you've decided to do this through rational [decision-making], you should not have to get permission from the medical profession," Nitschke says.
However, the deathstyle aspirations of the Sarco bely the fact obtaining one will not be as simple as swiping a credit card. To create a legal firewall, anyone wishing to obtain a Sarco would have to purchase the plans, print the device themselves — it requires a high-end industrial printer to do so — then assemble it. As with the Deliverance device, the end user must be able to answer computer-generated questions designed by a Swiss psychiatrist to determine if they are making a rational decision. The process concludes with the transmission of a four-digit code to make the Sarco operational.
As with many cutting-edge designs, the path to a working prototype has been nettlesome. Plans for a printed window have been abandoned. How it will be obtained by end users remains unclear. There have also been complications in creating an AI-based algorithm underlying the user questions to reliably determine if the individual is of sound mind.
While Nitschke believes the Sarco will be deployed in Switzerland for the first time sometime next year, it will almost certainly be a subject of immense controversy. The Hastings Center, one of the world's major bioethics organizations and a leader on end-of-life decision-making, flatly refused to comment on the Sarco.
Camosy strongly condemns it. He notes since U.S. life expectancy is actually shortening — with despair-driven suicide playing a role — efforts must be marshaled to mitigate the trend. To him, the Sarco sends an utterly wrong message.
"It is diabolical that we would create machines to make it easier for people to kill themselves."
"Most people who request help in killing themselves don't do so because they are in intense, unbearable pain," he observes. "They do it because the culture in which they live has made them feel like a burden. This culture has told them they only have value if they are able to be 'productive' and 'contribute to society.'" He adds that the large majority of disability activists have been against assisted suicide and euthanasia because it is imperative to their movement that a stigma remain in place.
"It is diabolical that we would create machines to make it easier for people to kill themselves," Camosy concludes. "And anyone with even a single progressive bone in their body should resist this disturbingly morbid profit-making venture with everything they have."
Biologists are Growing Mini-Brains. What If They Become Conscious?
Brain coral metaphorically represents the concept of growing organoids in a dish that may eventually attain consciousness--if scientists can first determine what that means.
Few images are more uncanny than that of a brain without a body, fully sentient but afloat in sterile isolation. Such specters have spooked the speculatively-minded since the seventeenth century, when René Descartes declared, "I think, therefore I am."
Since August 29, 2019, the prospect of a bodiless but functional brain has begun to seem far less fantastical.
In Meditations on First Philosophy (1641), the French penseur spins a chilling thought experiment: he imagines "having no hands or eyes, or flesh, or blood or senses," but being tricked by a demon into believing he has all these things, and a world to go with them. A disembodied brain itself becomes a demon in the classic young-adult novel A Wrinkle in Time (1962), using mind control to subjugate a planet called Camazotz. In the sci-fi blockbuster The Matrix (1999), most of humanity endures something like Descartes' nightmare—kept in womblike pods by their computer overlords, who fill the captives' brains with a synthetized reality while tapping their metabolic energy as a power source.
Since August 29, 2019, however, the prospect of a bodiless but functional brain has begun to seem far less fantastical. On that date, researchers at the University of California, San Diego published a study in the journal Cell Stem Cell, reporting the detection of brainwaves in cerebral organoids—pea-size "mini-brains" grown in the lab. Such organoids had emitted random electrical impulses in the past, but not these complex, synchronized oscillations. "There are some of my colleagues who say, 'No, these things will never be conscious,'" lead researcher Alysson Muotri, a Brazilian-born biologist, told The New York Times. "Now I'm not so sure."

Alysson Muotri has no qualms about his creations attaining consciousness as a side effect of advancing medical breakthroughs.
(Credit: ZELMAN STUDIOS)
Muotri's findings—and his avowed ambition to push them further—brought new urgency to simmering concerns over the implications of brain organoid research. "The closer we come to his goal," said Christof Koch, chief scientist and president of the Allen Brain Institute in Seattle, "the more likely we will get a brain that is capable of sentience and feeling pain, agony, and distress." At the annual meeting of the Society for Neuroscience, researchers from the Green Neuroscience Laboratory in San Diego called for a partial moratorium, warning that the field was "perilously close to crossing this ethical Rubicon and may have already done so."
Yet experts are far from a consensus on whether brain organoids can become conscious, whether that development would necessarily be dreadful—or even how to tell if it has occurred.
So how worried do we need to be?
***
An organoid is a miniaturized, simplified version of an organ, cultured from various types of stem cells. Scientists first learned to make them in the 1980s, and have since turned out mini-hearts, lungs, kidneys, intestines, thyroids, and retinas, among other wonders. These creations can be used for everything from observation of basic biological processes to testing the effects of gene variants, pathogens, or medications. They enable researchers to run experiments that might be less accurate using animal models and unethical or impractical using actual humans. And because organoids are three-dimensional, they can yield insights into structural, developmental, and other matters that an ordinary cell culture could never provide.
In 2006, Japanese biologist Shinya Yamanaka developed a mix of proteins that turned skin cells into "pluripotent" stem cells, which could subsequently be transformed into neurons, muscle cells, or blood cells. (He later won a Nobel Prize for his efforts.) Developmental biologist Madeline Lancaster, then a post-doctoral student at the Institute of Molecular Biotechnology in Vienna, adapted that technique to grow the first brain organoids in 2013. Other researchers soon followed suit, cultivating specialized mini-brains to study disorders ranging from microcephaly to schizophrenia.
Muotri, now a youthful 45-year-old, was among the boldest of these pioneers. His team revealed the process by which Zika virus causes brain damage, and showed that sofosbuvir, a drug previously approved for hepatitis C, protected organoids from infection. He persuaded NASA to fly his organoids to the International Space Station, where they're being used to trace the impact of microgravity on neurodevelopment. He grew brain organoids using cells implanted with Neanderthal genes, and found that their wiring differed from organoids with modern DNA.
Like the latter experiment, Muotri's brainwave breakthrough emerged from a longtime obsession with neuroarchaeology. "I wanted to figure out how the human brain became unique," he told me in a phone interview. "Compared to other species, we are very social. So I looked for conditions where the social brain doesn't function well, and that led me to autism." He began investigating how gene variants associated with severe forms of the disorder affected neural networks in brain organoids.
Tinkering with chemical cocktails, Muotri and his colleagues were able to keep their organoids alive far longer than earlier versions, and to culture more diverse types of brain cells. One team member, Priscilla Negraes, devised a way to measure the mini-brains' electrical activity, by planting them in a tray lined with electrodes. By four months, the researchers found to their astonishment, normal organoids (but not those with an autism gene) emitted bursts of synchronized firing, separated by 20-second silences. At nine months, the organoids were producing up to 300,000 spikes per minute, across a range of frequencies.
He shared his vision for "brain farms," which would grow organoids en masse for drug development or tissue transplants.
When the team used an artificial intelligence system to compare these patterns with EEGs of gestating fetuses, the program found them to be nearly identical at each stage of development. As many scientists noted when the news broke, that didn't mean the organoids were conscious. (Their chaotic bursts bore little resemblance to the orderly rhythms of waking adult brains.) But to some observers, it suggested that they might be approaching the borderline.
***
Shortly after Muotri's team published their findings, I attended a conference at UCSD on the ethical questions they raised. The scientist, in jeans and a sky-blue shirt, spoke rhapsodically of brain organoids' potential to solve scientific mysteries and lead to new medical treatments. He showed video of a spider-like robot connected to an organoid through a computer interface. The machine responded to different brainwave patterns by walking or stopping—the first stage, Muotri hoped, in teaching organoids to communicate with the outside world. He described his plans to develop organoids with multiple brain regions, and to hook them up to retinal organoids so they could "see." He shared his vision for "brain farms," which would grow organoids en masse for drug development or tissue transplants.

Muotri holds a spider-like robot that can connect to an organoid through a computer interface.
(Credit: ROLAND LIZARONDO/KPBS)
Yet Muotri also stressed the current limitations of the technology. His organoids contain approximately 2 million neurons, compared to about 200 million in a rat's brain and 86 billion in an adult human's. They consist only of a cerebral cortex, and lack many of a real brain's cell types. Because researchers haven't yet found a way to give organoids blood vessels, moreover, nutrients can't penetrate their inner recesses—a severe constraint on their growth.
Another panelist strongly downplayed the imminence of any Rubicon. Patricia Churchland, an eminent philosopher of neuroscience, cited research suggesting that in mammals, networked connections between the cortex and the thalamus are a minimum requirement for consciousness. "It may be a blessing that you don't have the enabling conditions," she said, "because then you don't have the ethical issues."
Christof Koch, for his part, sounded much less apprehensive than the Times had made him seem. He noted that science lacks a definition of consciousness, beyond an organism's sense of its own existence—"the fact that it feels like something to be you or me." As to the competing notions of how the phenomenon arises, he explained, he prefers one known as Integrated Information Theory, developed by neuroscientist Giulio Tononi. IIT considers consciousness to be a quality intrinsic to systems that reach a certain level of complexity, integration, and causal power (the ability for present actions to determine future states). By that standard, Koch doubted that brain organoids had stepped over the threshold.
One way to tell, he said, might be to use the "zap and zip" test invented by Tononi and his colleague Marcello Massimini in the early 2000s to determine whether patients are conscious in the medical sense. This technique zaps the brain with a pulse of magnetic energy, using a coil held to the scalp. As loops of neural impulses cascade through the cerebral circuitry, an EEG records the firing patterns. In a waking brain, the feedback is highly complex—neither totally predictable nor totally random. In other states, such as sleep, coma, or anesthesia, the rhythms are simpler. Applying an algorithm commonly used for computer "zip" files, the researchers devised a scale that allowed them to correctly diagnose most patients who were minimally conscious or in a vegetative state.
If scientists could find a way to apply "zap and zip" to brain organoids, Koch ventured, it should be possible to rank their degree of awareness on a similar scale. And if it turned out that an organoid was conscious, he added, our ethical calculations should strive to minimize suffering, and avoid it where possible—just as we now do, or ought to, with animal subjects. (Muotri, I later learned, was already contemplating sensors that would signal when organoids were likely in distress.)
During the question-and-answer period, an audience member pressed Churchland about how her views might change if the "enabling conditions" for consciousness in brain organoids were to arise. "My feeling is, we'll answer that when we get there," she said. "That's an unsatisfying answer, but it's because I don't know. Maybe they're totally happy hanging out in a dish! Maybe that's the way to be."
***
Muotri himself admits to no qualms about his creations attaining consciousness, whether sooner or later. "I think we should try to replicate the model as close as possible to the human brain," he told me after the conference. "And if that involves having a human consciousness, we should go in that direction." Still, he said, if strong evidence of sentience does arise, "we should pause and discuss among ourselves what to do."
"The field is moving so rapidly, you blink your eyes and another advance has occurred."
Churchland figures it will be at least a decade before anyone reaches the crossroads. "That's partly because the thalamus has a very complex architecture," she said. It might be possible to mimic that architecture in the lab, she added, "but I tend to think it's not going to be a piece of cake."
If anything worries Churchland about brain organoids, in fact, it's that Muotri's visionary claims for their potential could set off a backlash among those who find them unacceptably spooky. "Alysson has done brilliant work, and he's wonderfully charismatic and charming," she said. "But then there's that guy back there who doesn't think it's exciting; he thinks you're the Devil incarnate. You're playing into the hands of people who are going to shut you down."
Koch, however, is more willing to indulge Muotri's dreams. "Ten years ago," he said, "nobody would have believed you can take a stem cell and get an entire retina out of it. It's absolutely frigging amazing. So who am I to say the same thing can't be true for the thalamus or the cortex? The field is moving so rapidly, you blink your eyes and another advance has occurred."
The point, he went on, is not to build a Cartesian thought experiment—or a Matrix-style dystopia—but to vanquish some of humankind's most terrifying foes. "You know, my dad passed away of Parkinson's. I had a twin daughter; she passed away of sudden death syndrome. One of my best friends killed herself; she was schizophrenic. We want to eliminate all these terrible things, and that requires experimentation. We just have to go into it with open eyes."

