The Skinny on Fat and Covid-19
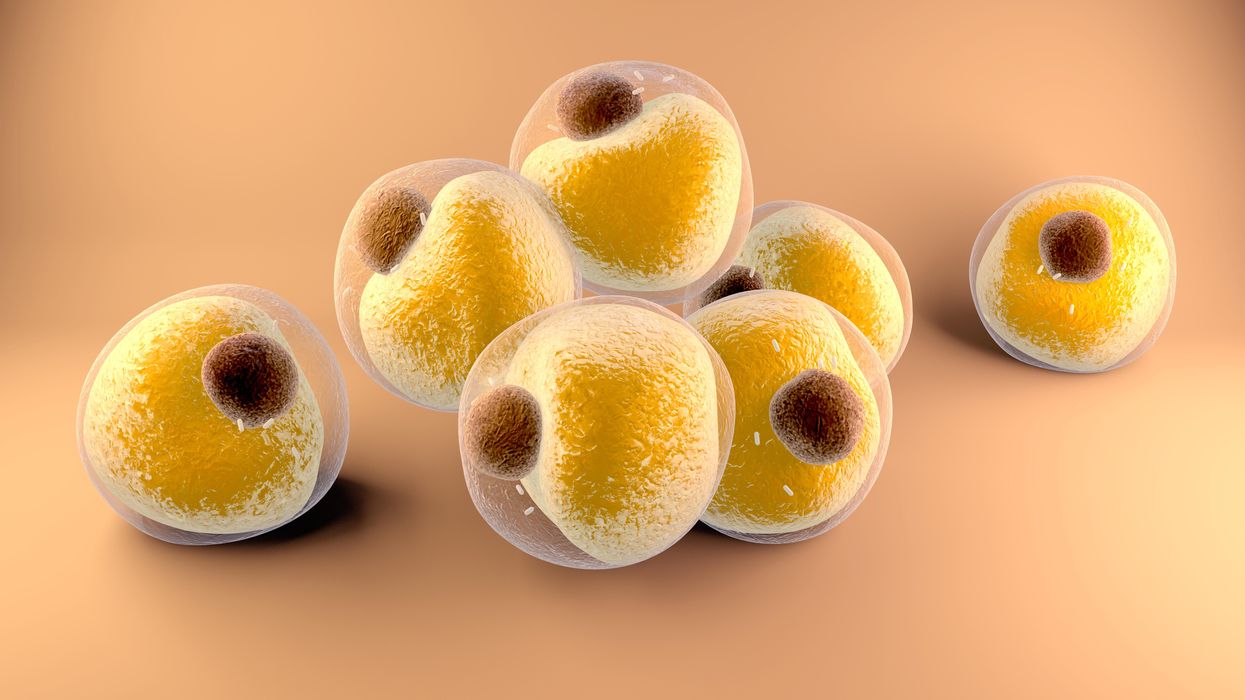
Researchers at Stanford have found that the virus that causes Covid-19 can infect fat cells, which could help explain why obesity is linked to worse outcomes for those who catch Covid-19.
Obesity is a risk factor for worse outcomes for a variety of medical conditions ranging from cancer to Covid-19. Most experts attribute it simply to underlying low-grade inflammation and added weight that make breathing more difficult.
Now researchers have found a more direct reason: SARS-CoV-2, the virus that causes Covid-19, can infect adipocytes, more commonly known as fat cells, and macrophages, immune cells that are part of the broader matrix of cells that support fat tissue. Stanford University researchers Catherine Blish and Tracey McLaughlin are senior authors of the study.
Most of us think of fat as the spare tire that can accumulate around the middle as we age, but fat also is present closer to most internal organs. McLaughlin's research has focused on epicardial fat, “which sits right on top of the heart with no physical barrier at all,” she says. So if that fat got infected and inflamed, it might directly affect the heart.” That could help explain cardiovascular problems associated with Covid-19 infections.
Looking at tissue taken from autopsy, there was evidence of SARS-CoV-2 virus inside the fat cells as well as surrounding inflammation. In fat cells and immune cells harvested from health humans, infection in the laboratory drove "an inflammatory response, particularly in the macrophages…They secreted proteins that are typically seen in a cytokine storm” where the immune response runs amok with potential life-threatening consequences. This suggests to McLaughlin “that there could be a regional and even a systemic inflammatory response following infection in fat.”
It is easy to see how the airborne SARS-CoV-2 virus infects the nose and lungs, but how does it get into fat tissue? That is a mystery and the source of ample speculation.
The macrophages studied by McLaughlin and Blish were spewing out inflammatory proteins, While the the virus within them was replicating, the new viral particles were not able to replicate within those cells. It was a different story in the fat cells. “When [the virus] gets into the fat cells, it not only replicates, it's a productive infection, which means the resulting viral particles can infect another cell,” including microphages, McLaughlin explains. It seems to be a symbiotic tango of the virus between the two cell types that keeps the cycle going.
It is easy to see how the airborne SARS-CoV-2 virus infects the nose and lungs, but how does it get into fat tissue? That is a mystery and the source of ample speculation.
Macrophages are mobile; they engulf and carry invading pathogens to lymphoid tissue in the lymph nodes, tonsils and elsewhere in the body to alert T cells of the immune system to the pathogen. Perhaps some of them also carry the virus through the bloodstream to more distant tissue.
ACE2 receptors are the means by which SARS-CoV-2 latches on to and enters most cells. They are not thought to be common on fat cells, so initially most researchers thought it unlikely they would become infected.
However, while some cell receptors always sit on the surface of the cell, other receptors are expressed on the surface only under certain conditions. Philipp Scherer, a professor of internal medicine and director of the Touchstone Diabetes Center at the University of Texas Southwestern Medical Center, suggests that, in people who have obesity, “There might be higher levels of dysfunctional [fat cells] that facilitate entry of the virus,” either through transiently expressed ACE2 or other receptors. Inflammatory proteins generated by macrophages might contribute to this process.
Another hypothesis is that viral RNA might be smuggled into fat cells as cargo in small bits of material called extracellular vesicles, or EVs, that can travel between cells. Other researchers have shown that when EVs express ACE2 receptors, they can act as decoys for SARS-CoV-2, where the virus binds to them rather than a cell. These scientists are working to create drugs that mimic this decoy effect as an approach to therapy.
Do fat cells play a role in Long Covid? “Fat cells are a great place to hide. You have all the energy you need and fat cells turn over very slowly; they have a half-life of ten years,” says Scherer. Observational studies suggest that acute Covid-19 can trigger the onset of diabetes especially in people who are overweight, and that patients taking medicines to regulate their diabetes “were actually quite protective” against acute Covid-19. Scherer has funding to study the risks and benefits of those drugs in animal models of Long Covid.
McLaughlin says there are two areas of potential concern with fat tissue and Long Covid. One is that this tissue might serve as a “big reservoir where the virus continues to replicate and is sent out” to other parts of the body. The second is that inflammation due to infected fat cells and macrophages can result in fibrosis or scar tissue forming around organs, inhibiting their function. Once scar tissue forms, the tissue damage becomes more difficult to repair.
Current Covid-19 treatments work by stopping the virus from entering cells through the ACE2 receptor, so they likely would have no effect on virus that uses a different mechanism. That means another approach will have to be developed to complement the treatments we already have. So the best advice McLaughlin can offer today is to keep current on vaccinations and boosters and lose weight to reduce the risk associated with obesity.
Here's how one doctor overcame extraordinary odds to help create the birth control pill
Dr. Percy Julian had so many personal and professional obstacles throughout his life, it’s amazing he was able to accomplish anything at all. But this hidden figure not only overcame these incredible obstacles, he also laid the foundation for the creation of the birth control pill.
Julian’s first obstacle was growing up in the Jim Crow-era south in the early part of the twentieth century, where racial segregation kept many African-Americans out of schools, libraries, parks, restaurants, and more. Despite limited opportunities and education, Julian was accepted to DePauw University in Indiana, where he majored in chemistry. But in college, Julian encountered another obstacle: he wasn’t allowed to stay in DePauw’s student housing because of segregation. Julian found lodging in an off-campus boarding house that refused to serve him meals. To pay for his room, board, and food, Julian waited tables and fired furnaces while he studied chemistry full-time. Incredibly, he graduated in 1920 as valedictorian of his class.
After graduation, Julian landed a fellowship at Harvard University to study chemistry—but here, Julian ran into yet another obstacle. Harvard thought that white students would resent being taught by Julian, an African-American man, so they withdrew his teaching assistantship. Julian instead decided to complete his PhD at the University of Vienna in Austria. When he did, he became one of the first African Americans to ever receive a PhD in chemistry.
Julian received offers for professorships, fellowships, and jobs throughout the 1930s, due to his impressive qualifications—but these offers were almost always revoked when schools or potential employers found out Julian was black. In one instance, Julian was offered a job at the Institute of Paper Chemistory in Appleton, Wisconsin—but Appleton, like many cities in the United States at the time, was known as a “sundown town,” which meant that black people weren’t allowed to be there after dark. As a result, Julian lost the job.
During this time, Julian became an expert at synthesis, which is the process of turning one substance into another through a series of planned chemical reactions. Julian synthesized a plant compound called physostigmine, which would later become a treatment for an eye disease called glaucoma.
In 1936, Julian was finally able to land—and keep—a job at Glidden, and there he found a way to extract soybean protein. This was used to produce a fire-retardant foam used in fire extinguishers to smother oil and gasoline fires aboard ships and aircraft carriers, and it ended up saving the lives of thousands of soldiers during World War II.
At Glidden, Julian found a way to synthesize human sex hormones such as progesterone, estrogen, and testosterone, from plants. This was a hugely profitable discovery for his company—but it also meant that clinicians now had huge quantities of these hormones, making hormone therapy cheaper and easier to come by. His work also laid the foundation for the creation of hormonal birth control: Without the ability to synthesize these hormones, hormonal birth control would not exist.
Julian left Glidden in the 1950s and formed his own company, called Julian Laboratories, outside of Chicago, where he manufactured steroids and conducted his own research. The company turned profitable within a year, but even so Julian’s obstacles weren’t over. In 1950 and 1951, Julian’s home was firebombed and attacked with dynamite, with his family inside. Julian often had to sit out on the front porch of his home with a shotgun to protect his family from violence.
But despite years of racism and violence, Julian’s story has a happy ending. Julian’s family was eventually welcomed into the neighborhood and protected from future attacks (Julian’s daughter lives there to this day). Julian then became one of the country’s first black millionaires when he sold his company in the 1960s.
When Julian passed away at the age of 76, he had more than 130 chemical patents to his name and left behind a body of work that benefits people to this day.
Therapies for Healthy Aging with Dr. Alexandra Bause
My guest today is Dr. Alexandra Bause, a biologist who has dedicated her career to advancing health, medicine and healthier human lifespans. Dr. Bause co-founded a company called Apollo Health Ventures in 2017. Currently a venture partner at Apollo, she's immersed in the discoveries underway in Apollo’s Venture Lab while the company focuses on assembling a team of investors to support progress. Dr. Bause and Apollo Health Ventures say that biotech is at “an inflection point” and is set to become a driver of important change and economic value.
Previously, Dr. Bause worked at the Boston Consulting Group in its healthcare practice specializing in biopharma strategy, among other priorities
She did her PhD studies at Harvard Medical School focusing on molecular mechanisms that contribute to cellular aging, and she’s also a trained pharmacist
In the episode, we talk about the present and future of therapeutics that could increase people’s spans of health, the benefits of certain lifestyle practice, the best use of electronic wearables for these purposes, and much more.
Dr. Bause is at the forefront of developing interventions that target the aging process with the aim of ensuring that all of us can have healthier, more productive lifespans.


