Epigenetic therapeutics could revolutionize medicine in the coming decades. (© kentoh/Adobe)
How exactly does your DNA make you who you are?
It's because of epigenetics that identical twins can actually look different and develop different diseases.
Just as software developers don't write apps out of ones and zeros, the interesting parts of the human genome aren't written merely in As, Ts, Cs and Gs. Yes, these are the fundamental letters that make up our DNA and encode the proteins that make our cells function, but the story doesn't end there.
Our cells possess amazing abilities, like eating invading bacteria or patching over a wound, and these abilities require the coordinated action of hundreds, if not thousands, of proteins. Epigenetics, the study of gene expression, examines how multiple genes work at once to make these biological processes happen.
It's because of epigenetics that identical twins – who possess identical DNA -- can actually look different and develop different diseases. Their environments may influence the expression of their genes in unique ways. For example, a research study in mice found that maternal exposure to a chemical called bisphenol A (BPA) resulted in drastic differences between genetically identical offspring. BPA exposure increased the likelihood that a certain gene was turned on, which led to the birth of yellow mice who were prone to obesity. Their genetically identical siblings who were not exposed to BPA were thinner and born with brown fur.

These three mice are genetically identical. Epigenetic differences, however, result in vastly different phenotypes.
(© 1994 Nature Publishing Group, Duhl, D.)
This famous mouse experiment is just one example of how epigenetics may transform medicine in the coming years. By studying the way genes are turned on and off, and maybe even making those changes ourselves, scientists are beginning to approach diseases like cancer in a completely new way.
With few exceptions, most of the 1 trillion cells that make up your body contain the same DNA instructions as all the others. How does each cell in your body know what it is and what it has to do? One of the answers appears to lie in epigenetic regulation. Just as everyone at a company may have access to all the same files on the office Dropbox, the accountants will put different files on their desktop than the lawyers do.
Our cells prioritize DNA sequences in the same way, even storing entire chromosomes that aren't needed along the wall of the nucleus, while keeping important pieces of DNA in the center, where it is most accessible to be read and used. One of the ways our cells prioritize certain DNA sequences is through methylation, a process that inactivates large regions of genes without editing the underlying "file" itself.
As we learn more about epigenetics, we gain more opportunities to develop therapeutics for a broad range of human conditions, from cancer to metabolic disorders. Though there have not been any clinical applications of epigenetics to immune or metabolic diseases yet, cancer is one of the leading areas, with promising initial successes.
One of the challenges of cancer treatments is that different patients may respond positively or negatively to the same treatment. With knowledge of epigenetics, however, doctors could conduct diagnostic tests to identify a patient's specific epigenetic profile and determine the best treatment for him or her. Already, commercial kits are available that help doctors screen glioma patients for an epigenetic biomarker called MGMT, because patients with this biomarker have shown high rates of success with certain kinds of treatments.
Other epigenetic advances go beyond personalized screening to treatments targeting the mechanism of disease. Some epigenetic drugs turn on genes that help suppress tumors, while others turn on genes that reveal the identity of tumor cells to the immune system, allowing it to attack cancerous cells.
Direct, targeted control of your epigenome could allow doctors to reprogram cancerous or aging cells.
The study of epigenetics has also been fundamental to the field of aging research. The older you get, the more methylation marks your DNA carries, and this has led to the distinction between biological aging, or the state of your cells, and chronological aging, or how old you actually are.
Just as our DNA can get miscopied and accumulate mutations, errors in DNA methylation can lead to so-called "epimutations". One of the big hypotheses in aging research today is that the accumulation of these random epimutations over time is responsible for what we perceive as aging.
Studies thus far have been correlative - looking at several hundred sites of epigenetic modifications in a person's cell, scientists can now roughly discern the age of that person. The next set of advances in the field will come from learning what these epigenetic changes individually do by themselves, and if certain methylations are correlated with cellular aging. General diagnostic terms like "aging" could be replaced with "abnormal methylation at these specific locations," which would also open the door to new therapeutic targets.
Direct, targeted control of your epigenome could allow doctors to reprogram cancerous or aging cells. While this type of genetic surgery is not feasible just yet, current research is bringing that possibility closer. The Cas9 protein of genome-editing CRISPR/Cas9 fame has been fused with epigenome modifying enzymes to target epigenetic modifications to specific DNA sequences.
A therapeutic of this type could theoretically undo a harmful DNA methylation, but would also be competing with the cell's native machinery responsible for controlling this process. One potential approach around this problem involves making beneficial synthetic changes to the epigenome that our cells do not have the capacity to undo.
Also fueling this frontier is a new approach to understanding disease itself. Scientists and doctors are now moving beyond the "one defective gene = one disease" paradigm. Because lots of diseases are caused by multiple genes going haywire, epigenetic therapies could hold the key to new types of treatments by targeting multiple defective genes at once.
Scientists are still discovering which epigenetic modifications are responsible for particular diseases, and engineers are building new tools for epigenome editing. Given the proliferation of work in these fields within the last 10 years, we may see epigenetic therapeutics emerging within the next couple of decades.
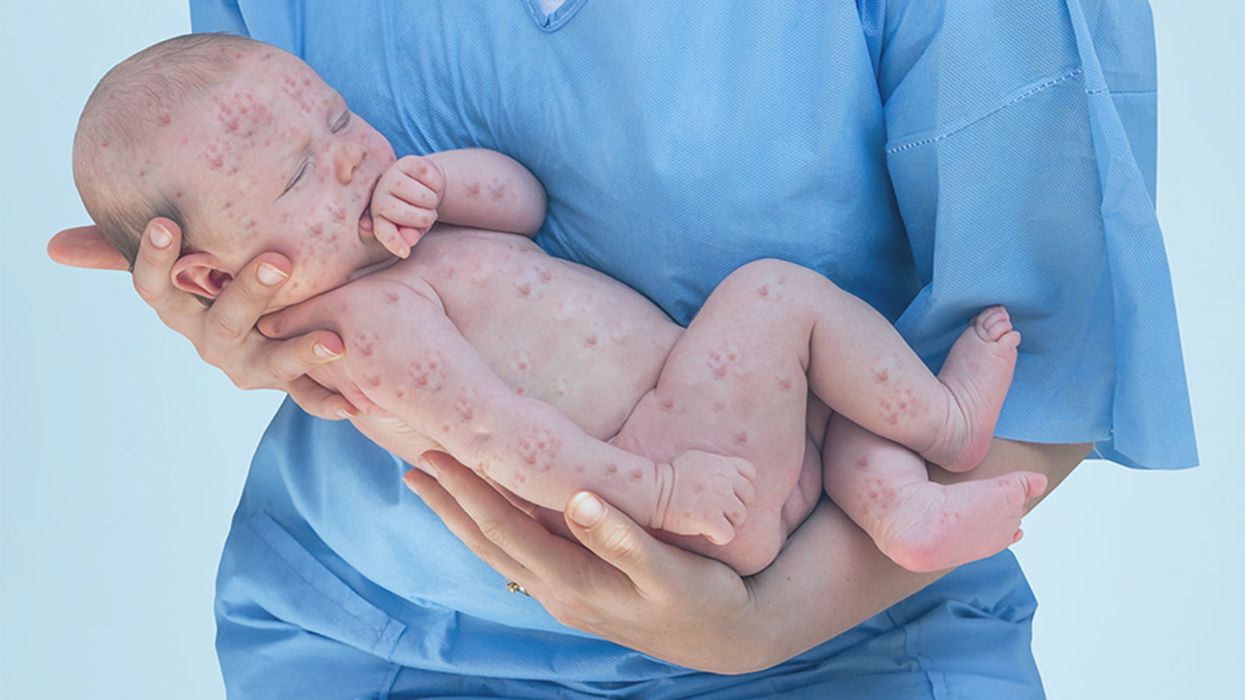
A doctor cradles a newborn who is sick with measles.
Ethan Lindenberger, the Ohio teenager who sought out vaccinations after he was denied them as a child, recently testified before Congress about why his parents became anti-vaxxers. The trouble, he believes, stems from the pervasiveness of misinformation online.
There is evidence that 'educating' people with facts about the benefits of vaccination may not be effective.
"For my mother, her love and affection and care as a parent was used to push an agenda to create a false distress," he told the Senate Committee. His mother read posts on social media saying vaccines are dangerous, and that was enough to persuade her against them.
His story is an example of how widespread and harmful the current discourse on vaccinations is—and more importantly—how traditional strategies to convince people about the merits of vaccination have largely failed.
As responsible members of society, all of us have implicitly signed on to what ethicists call the "Social Contract" -- we agree to abide by certain moral and political rules of behavior. This is what our societal values, norms, and often governments are based upon. However, with the unprecedented rise of social media, alternative facts, and fake news, it is evident that our understanding—and application—of the social contract must also evolve.
Nowhere is this breakdown of societal norms more visible than in the failure to contain the spread of vaccine-preventable diseases like measles. What started off as unexplained episodes in New York City last October, mostly in communities that are under-vaccinated, has exploded into a national epidemic: 880 cases of measles across 24 states in 2019, according to the CDC (as of May 17, 2019). In fact, the Unites States is only eight months away from losing its "measles free" status, joining Venezuela as the second country out of North and South America with that status.
The U.S. is not the only country facing this growing problem. Such constant and perilous reemergence of measles and other vaccine-preventable diseases in various parts of the world raises doubts about the efficacy of current vaccination policies. In addition to the loss of valuable life, these outbreaks lead to loss of millions of dollars in unnecessary expenditure of scarce healthcare resources. While we may be living through an age of information, we are also navigating an era whose hallmark is a massive onslaught on truth.
There is ample evidence on how these outbreaks start: low-vaccination rates. At the same time, there is evidence that 'educating' people with facts about the benefits of vaccination may not be effective. Indeed, human reasoning has a limit, and facts alone rarely change a person's opinion. In a fascinating report by researchers from the University of Pennsylvania, a small experiment revealed how "behavioral nudges" could inform policy decisions around vaccination.
In the reported experiment, the vaccination rate for employees of a company increased by 1.5 percent when they were prompted to name the date when they planned to get their flu shot. In the same experiment, when employees were prompted to name both a date and a time for their planned flu shot, vaccination rate increased by 4 percent.
A randomized trial revealed the subtle power of "announcements" – direct, brief, assertive statements by physicians that assumed parents were ready to vaccinate their children.
This experiment is a part of an emerging field of behavioral economics—a scientific undertaking that uses insights from psychology to understand human decision-making. The field was born from a humbling realization that humans probably do not possess an unlimited capacity for processing information. Work in this field could inform how we can formulate vaccination policy that is effective, conserves healthcare resources, and is applicable to current societal norms.
Take, for instance, the case of Human Papilloma Virus (HPV) that can cause several types of cancers in both men and women. Research into the quality of physician communication has repeatedly revealed how lukewarm recommendations for HPV vaccination by primary care physicians likely contributes to under-immunization of eligible adolescents and can cause confusion for parents.
A randomized trial revealed the subtle power of "announcements" – direct, brief, assertive statements by physicians that assumed parents were ready to vaccinate their children. These announcements increased vaccination rates by 5.4 percent. Lengthy, open-ended dialogues demonstrated no benefit in vaccination rates. It seems that uncertainty from the physician translates to unwillingness from a parent.
Choice architecture is another compelling concept. The premise is simple: We hardly make any of our decisions in vacuum; the environment in which these decisions are made has an influence. If health systems were designed with these insights in mind, people would be more likely to make better choices—without being forced.
This theory, proposed by Richard Thaler, who won the 2017 Nobel Prize in Economics, was put to the test by physicians at the University of Pennsylvania. In their study, flu vaccination rates at primary care practices increased by 9.5 percent all because the staff implemented "active choice intervention" in their electronic health records—a prompt that nudged doctors and nurses to ask patients if they'd gotten the vaccine yet. This study illustrated how an intervention as simple as a reminder can save lives.
To be sure, some bioethicists do worry about implementing these policies. Are behavioral nudges akin to increased scrutiny or a burden for the disadvantaged? For example, would incentives to quit smoking unfairly target the poor, who are more likely to receive criticism for bad choices?
The measles outbreak is a sober reminder of how devastating it can be when the social contract breaks down.
While this is a valid concern, behavioral economics offers one of the only ethical solutions to increasing vaccination rates by addressing the most critical—and often legal—challenge to universal vaccinations: mandates. Choice architecture and other interventions encourage and inform a choice, allowing an individual to retain his or her right to refuse unwanted treatment. This distinction is especially important, as evidence suggests that people who refuse vaccinations often do so as a result of cognitive biases – systematic errors in thinking resulting from emotional attachment or a lack of information.
For instance, people are prone to "confirmation bias," or a tendency to selectively believe in information that confirms their preexisting theories, rather than the available evidence. At the same time, people do not like mandates. In such situations, choice architecture provides a useful option: people are nudged to make the right choice via the design of health delivery systems, without needing policies that rely on force.
The measles outbreak is a sober reminder of how devastating it can be when the social contract breaks down and people fall prey to misinformation. But all is not lost. As we fight a larger societal battle against alternative facts, we now have another option in the trenches to subtly encourage people to make better choices.
Using insights from research in decision-making, we can all contribute meaningfully in controversial conversations with family, friends, neighbors, colleagues, and our representatives — and push for policies that protect those we care about. A little more than a hundred years ago, thousands of lives were routinely lost to preventive illnesses. We've come too far to let ignorance destroy us now.
Study of the DNA double helix will lead to transformative medical care and increasingly urgent questions about how to responsibly handle genetic engineering technology.
The news last November that a rogue Chinese scientist had genetically altered the embryos of a pair of Chinese twins shocked the world. But although this use of advanced technology to change the human gene pool was premature, it was a harbinger of how genetic science will alter our healthcare, the way we make babies, the nature of the babies we make, and, ultimately, our sense of who and what we are as a species.
The healthcare applications of the genetics revolution are merely stations along the way to the ultimate destination.
But while the genetics revolution has already begun, we aren't prepared to handle these Promethean technologies responsibly.
By identifying the structure of DNA in the 1950s, Watson, Crick, Wilkins, and Franklin showed that the book of life was written in the DNA double helix. When the human genome project was completed in 2003, we saw how this book of human life could be transcribed. Painstaking research paired with advanced computational algorithms then showed what increasing numbers of genes do and how the genetic book of life can be read.
Now, with the advent of precision gene editing tools like CRISPR, we are seeing that the book of life -- and all biology -- can be re-written. Biology is being recognized as another form of readable, writable, and hackable information technology with we humans as the coders.
The impact of this transformation is being first experienced in our healthcare. Gene therapies including those extracting, re-engineering, then reintroducing a person's own cells enhanced into cancer-fighting supercells are already performing miracles in clinical trials. Thousands of applications have already been submitted to regulators across the globe for trials using gene therapies to address a host of other diseases.
Recently, the first gene editing of cells inside a person's body was deployed to treat the genetically relatively simple metabolic disorder Hunter syndrome, with many more applications to come. These new approaches are only the very first steps in our shift from the current system of generalized medicine based on population averages to precision medicine based on each patient's individual biology to predictive medicine based on AI-generated estimations of a person's future health state.

Jamie Metzl's groundbreaking new book, Hacking Darwin: Genetic Engineering and the Future of Humanity, explores how the genetic revolution is transforming our healthcare, the way we make babies, and the nature of and babies we make, what this means for each of us, and what we must all do now to prepare for what's coming.
This shift in our healthcare will ensure that millions and then billions of people will have their genomes sequenced as the foundation of their treatment. Big data analytics will then be used to compare at scale people's genotypes (what their genes say) to their phenotypes (how those genes are expressed over the course of their lives).
These massive datasets of genetic and life information will then make it possible to go far beyond the simple genetic analysis of today and to understand far more complex human diseases and traits influenced by hundreds or thousands of genes. Our understanding of this complex genetic system within the vaster ecosystem of our bodies and the environment around us will transform healthcare for the better and help us cure terrible diseases that have plagued our ancestors for millennia.
But as revolutionary as this challenge will be for medicine, the healthcare applications of the genetics revolution are merely stations along the way to the ultimate destination – a deep and fundamental transformation of our evolutionary trajectory as a species.
A first inkling of where we are heading can be seen in the direct-to-consumer genetic testing industry. Many people around the world have now sent their cheek swabs to companies like 23andMe for analysis. The information that comes back can tell people a lot about relatively simple genetic traits like carrier status for single gene mutation diseases, eye color, or whether they hate the taste of cilantro, but the information about complex traits like athletic predisposition, intelligence, or personality style today being shared by some of these companies is wildly misleading.
This will not always be the case. As the genetic and health data pools grow, analysis of large numbers of sequenced genomes will make it possible to apply big data analytics to predict some very complex genetic disease risks and the genetic components of traits like height, IQ, temperament, and personality style with increasing accuracy. This process, called "polygenic scoring," is already being offered in beta stage by a few companies and will become an ever bigger part of our lives going forward.
The most profound application of all this will be in our baby-making. Before making a decision about which of the fertilized eggs to implant, women undergoing in vitro fertilization can today elect to have a small number of cells extracted from their pre-implanted embryos and sequenced. With current technology, this can be used to screen for single-gene mutation diseases and other relatively simple disorders. Polygenic scoring, however, will soon make it possible to screen these early stage pre-implanted embryos to assess their risk of complex genetic diseases and even to make predictions about the heritable parts of complex human traits. The most intimate elements of being human will start feeling like high-pressure choices needing to be made by parents.
The limit of our imagination will become the most significant barrier to our recasting biology.
Adult stem cell technologies will then likely make it possible to generate hundreds or thousands of a woman's own eggs from her blood sample or skin graft. This would blow open the doors of reproductive possibility and allow parents to choose embryos with exceptional potential capabilities from a much larger set of options.
The complexity of human biology will place some limits to the extent of possible gene edits that might be made to these embryos, but all of biology, including our own, is extremely flexible. How else could all the diversity of life have emerged from a single cell nearly four billion years ago? The limit of our imagination will become the most significant barrier to our recasting biology.
But while we humans are gaining the powers of the gods, we aren't at all ready to use them.
The same tools that will help cure our worst afflictions, save our children, help us live longer, healthier, more robust lives will also open the door to potential abuses. Prospective parents with the best of intentions or governments with lax regulatory structures or aggressive ideas of how population-wide genetic engineering might be used to enhance national competitiveness or achieve some other goal could propel us into a genetic arms race that could undermine our essential diversity, dangerously divide societies, lead to dangerous, destabilizing, and potentially even deadly conflicts between us, and threaten our very humanity.
But while the advance of genetic technologies is inevitable, how it plays out is anything but. If we don't want the genetic revolution to undermine our species or lead to grave conflicts between genetic haves and have nots or between societies opting in and those opting out, now is the time when we need to make smart decisions based on our individual and collective best values. Although the technology driving the genetic revolution is new, the value systems we will need to optimize the benefits and minimize the harms of this massive transformation are ones we have been developing for thousands of years.
And while some very smart and well-intentioned scientists have been meeting to explore what comes next, it won't be enough for a few of even our wisest prophets to make decisions about the future of our species that will impact everyone. We'll also need smart regulations on both the national and international levels.
Every country will need to have its own regulatory guidelines for human genetic engineering based on both international best practices and the country's unique traditions and values. Because we are all one species, however, we will also ultimately need to develop guidelines that can apply to all of us.
As a first step toward making this possible, we must urgently launch a global, species-wide education effort and inclusive dialogue on the future of human genetic engineering that can eventually inform global norms that will need to underpin international regulations. This process will not be easy, but the alternative of an unregulated genetic arms race would be far worse.
The overlapping genomics and AI revolutions may seem like distant science fiction but are closer than you think. Far sooner than most people recognize, the inherent benefits of these technologies and competition between us will spark rapid adoption. Before that spark ignites, we have a brief moment to come together as a species like we never have before to articulate and translate into action the future we jointly envision. The north star of our best shared values can help us navigate the almost unimaginable opportunities and very real challenges that lie ahead.