Pregnant & Breastfeeding Women Who Get the COVID-19 Vaccine Are Protecting Their Infants, Research Suggests

Becky Cummings, who got vaccinated in December, snuggles her newborn, Clark, while he takes a nap.
Becky Cummings had multiple reasons to get vaccinated against COVID-19 while tending to her firstborn, Clark, who arrived in September 2020 at 27 weeks.
The 29-year-old intensive care unit nurse in Greensboro, North Carolina, had witnessed the devastation day in and day out as the virus took its toll on the young and old. But when she was offered the vaccine, she hesitated, skeptical of its rapid emergency use authorization.
Exclusion of pregnant and lactating mothers from clinical trials fueled her concerns. Ultimately, though, she concluded the benefits of vaccination outweighed the risks of contracting the potentially deadly virus.
"Long story short," Cummings says, in December "I got vaccinated to protect myself, my family, my patients, and the general public."
At the time, Cummings remained on the fence about breastfeeding, citing a lack of evidence to support its safety after vaccination, so she pumped and stashed breast milk in the freezer. Her son is adjusting to life as a preemie, requiring mother's milk to be thickened with formula, but she's becoming comfortable with the idea of breastfeeding as more research suggests it's safe.
"If I could pop him on the boob," she says, "I would do it in a heartbeat."
Now, a study recently published in the Journal of the American Medical Association found "robust secretion" of specific antibodies in the breast milk of mothers who received a COVID-19 vaccine, indicating a potentially protective effect against infection in their infants.
The presence of antibodies in the breast milk, detectable as early as two weeks after vaccination, lasted for six weeks after the second dose of the Pfizer-BioNTech vaccine.
"We believe antibody secretion into breast milk will persist for much longer than six weeks, but we first wanted to prove any secretion at all after vaccination," says Ilan Youngster, the study's corresponding author and head of pediatric infectious diseases at Shamir Medical Center in Zerifin, Israel.
That's why the research team performed a preliminary analysis at six weeks. "We are still collecting samples from participants and hope to soon be able to comment about the duration of secretion."
As with other respiratory illnesses, such as influenza and pertussis, secretion of antibodies in breast milk confers protection from infection in infants. The researchers expect a similar immune response from the COVID-19 vaccine and are expecting the findings to spur an increase in vaccine acceptance among pregnant and lactating women.
A COVID-19 outbreak struck three families the research team followed in the study, resulting in at least one non-breastfed sibling developing symptomatic infection; however, none of the breastfed babies became ill. "This is obviously not empirical proof," Youngster acknowledges, "but still a nice anecdote."
Leaps.org inquired whether infants who derive antibodies only through breast milk are likely to have a lower immunity than infants whose mothers were vaccinated while they were in utero. In other words, is maternal transmission of antibodies stronger during pregnancy than during breastfeeding, or about the same?
"This is a different kind of transmission," Youngster explains. "When a woman is infected or vaccinated during pregnancy, some antibodies will be transferred through the placenta to the baby's bloodstream and be present for several months." But in the nursing mother, that protection occurs through local action. "We always recommend breastfeeding whenever possible, and, in this case, it might have added benefits."
A study published online in March found COVID-19 vaccination provided pregnant and lactating women with robust immune responses comparable to those experienced by their nonpregnant counterparts. The study, appearing in the American Journal of Obstetrics and Gynecology, documented the presence of vaccine-generated antibodies in umbilical cord blood and breast milk after mothers had been vaccinated.
Natali Aziz, a maternal-fetal medicine specialist at Stanford University School of Medicine, notes that it's too early to draw firm conclusions about the reduction in COVID-19 infection rates among newborns of vaccinated mothers. Citing the two aforementioned research studies, she says it's biologically plausible that antibodies passed through the placenta and breast milk impart protective benefits. While thousands of pregnant and lactating women have been vaccinated against COVID-19, without incurring adverse outcomes, many are still wondering whether it's safe to breastfeed afterward.
It's important to bear in mind that pregnant women may develop more severe COVID-19 complications, which could lead to intubation or admittance to the intensive care unit. "We, in our practice, are supporting pregnant and breastfeeding patients to be vaccinated," says Aziz, who is also director of perinatal infectious diseases at Stanford Children's Health, which has been vaccinating new mothers and other hospitalized patients at discharge since late April.
Earlier in April, Huntington Hospital in Long Island, New York, began offering the COVID-19 vaccine to women after they gave birth. The hospital chose the one-shot Johnson & Johnson vaccine for postpartum patients, so they wouldn't need to return for a second shot while acclimating to life with a newborn, says Mitchell Kramer, chairman of obstetrics and gynecology.
The hospital suspended the program when the Food and Drug Administration and the Centers for Disease Control and Prevention paused use of the J&J vaccine starting April 13, while investigating several reports of dangerous blood clots and low platelet counts among more than 7 million people in the United States who had received that vaccine.
In lifting the pause April 23, the agencies announced the vaccine's fact sheets will bear a warning of the heightened risk for a rare but serious blood clot disorder among women under age 50. As a result, Kramer says, "we will likely not be using the J&J vaccine for our postpartum population."
So, would it make sense to vaccinate infants when one for them eventually becomes available, not just their mothers? "In general, most of the time, infants do not have as good of an immune response to vaccines," says Jonathan Temte, associate dean for public health and community engagement at the University of Wisconsin School of Medicine and Public Health in Madison.
"Many of our vaccines are held until children are six months of age. For example, the influenza vaccine starts at age six months, the measles vaccine typically starts one year of age, as do rubella and mumps. Immune response is typically not very good for viral illnesses in young infants under the age of six months."
So far, the FDA has granted emergency use authorization of the Pfizer-BioNTech vaccine for children as young as 16 years old. The agency is considering data from Pfizer to lower that age limit to 12. Studies are also underway in children under age 12. Meanwhile, data from Moderna on 12-to 17-year-olds and from Pfizer on 12- to 15-year-olds have not been made public. (Pfizer announced at the end of March that its vaccine is 100 percent effective in preventing COVID-19 in the latter age group, and FDA authorization for this population is expected soon.)
"There will be step-wise progression to younger children, with infants and toddlers being the last ones tested," says James Campbell, a pediatric infectious diseases physician and head of maternal and child clinical studies at the University of Maryland School of Medicine Center for Vaccine Development.
"Once the data are analyzed for safety, tolerability, optimal dose and regimen, and immune responses," he adds, "they could be authorized and recommended and made available to American children." The data on younger children are not expected until the end of this year, with regulatory authorization possible in early 2022.
For now, Vonnie Cesar, a family nurse practitioner in Smyrna, Georgia, is aiming to persuade expectant and new mothers to get vaccinated. She has observed that patients in metro Atlanta seem more inclined than their rural counterparts.
To quell some of their skepticism and fears, Cesar, who also teaches nursing students, conceived a visual way to demonstrate the novel mechanism behind the COVID-19 vaccine technology. Holding a palm-size physical therapy ball outfitted with clear-colored push pins, she simulates the spiked protein of the coronavirus. Slime slathered at the gaps permeates areas around the spikes—a process similar to how our antibodies build immunity to the virus.
These conversations often lead hesitant patients to discuss vaccination with their husbands or partners. "The majority of people I'm speaking with," she says, "are coming to the conclusion that this is the right thing for me, this is the common good, and they want to make sure that they're here for their children."
CORRECTION: An earlier version of this article mistakenly stated that the COVID-19 vaccines were granted emergency "approval." They have been granted emergency use authorization, not full FDA approval. We regret the error.
George Papanicolaou (1883-1962), Greek-born American physician developed a simple cytological test for cervical cancer in 1928.
For decades, women around the world have made the annual pilgrimage to their doctor for the dreaded but potentially life-saving Papanicolaou test, a gynecological exam to screen for cervical cancer named for Georgios Papanicolaou, the Greek immigrant who developed it.
The Pap smear, as it is commonly known, is credited for reducing cervical cancer mortality by 70% since the 1960s; the American Cancer Society (ACS) still ranks the Pap as the most successful screening test for preventing serious malignancies. Nonetheless, the agency, as well as other medical panels, including the US Preventive Services Task Force and the American College of Obstetrics and Gynecology are making a strong push to replace the Pap with the more sensitive high-risk HPV screening test for the human papillomavirus virus, which causes nearly all cases of cervical cancer.
So, how was the Pap developed and how did it become the gold standard of cervical cancer detection for more than 60 years?
Born on May 13, 1883, on the island of Euboea, Greece, Georgios Papanicolaou attended the University of Athens where he majored in music and the humanities before earning his medical degree in 1904 and PhD from the University of Munich six years later. In Europe, Papanicolaou was an assistant military surgeon during the Balkan War, a psychologist for an expedition of the Oceanographic Institute of Monaco and a caregiver for leprosy patients.
When he and his wife, Andromache Mavroyenous (Mary), arrived at Ellis Island on October 19, 1913, the young couple had scarcely more than the $250 minimum required to immigrate, spoke no English and had no job prospects. They worked a series of menial jobs--department store sales clerk, rug salesman, newspaper clerk, restaurant violinist--before Papanicolaou landed a position as an anatomy assistant at Cornell University and Mary was hired as his lab assistant, an arrangement that would last for the next 50 years.
Papanikolaou would later say the discovery "was one of the greatest thrills I ever experienced during my scientific career."
In his early research, Papanikolaou used guinea pigs to prove that gender is determined by the X and Y chromosomes. Using a pediatric nasal speculum, he collected and microscopically examined vaginal secretions of guinea pigs, which revealed distinct cell changes connected to the menstrual cycle. He moved on to study reproductive patterns in humans, beginning with his faithful wife, Mary, who not only endured his almost-daily cervical exams for decades, but also recruited friends as early research participants.
Writing in the medical journal Growth in 1920, the scientist outlined his theory that a microscopic smear of vaginal fluid could detect the presence of cancer cells in the uterus. Papanikolaou would later say the discovery "was one of the greatest thrills I ever experienced during my scientific career."
At this time, cervical cancer was the number one cancer killer of American women but physicians were skeptical of these new findings. They continued to rely on biopsy and curettage to diagnose and treat the disease until Papanicolaou's discovery was published in American Journal of Obstetrics and Gynecology. An inexpensive, easy-to-perform test that could detect cervical cancer, precancerous dysplasia and other cytological diseases was a sea change. Between 1975 and 2001, the cervical cancer rate was cut in half.
Papanicolaou became Emeritus Professor at Cornell University Medical College and received numerous awards, including the Albert Lasker Award for Clinical Medical Research and the Medal of Honor from the American Cancer Society. His image was featured on the Greek currency and the US Post Office issued a commemorative stamp in his honor. But international acclaim didn't lead to a more relaxed schedule. The researcher continued to work seven days a week and refused to take vacations.
After nearly 50 years, Papanicolaou left Cornell to head and develop the Cancer Institute of Miami. He died of a heart attack on February 19, 1962, just three months after his arrival. Mary continued to work in the renamed Papanicolaou Cancer Research Institute until her death 20 years later.
The annual pap smear was originally tied to renewing a birth control prescription. Canada began recommending Pap exams every three years in 1978. The United States followed suit in 2012, noting that it takes many years for cervical cancer to develop. In September 2020, the American Cancer Society recommended delaying the first gynecological pelvic exam until age 25 and replacing the Pap test completely with the more accurate human papillomavirus (HPV) test every five years as the technology becomes more widely available.
Not everyone agrees that it's time to do away with this proven screening method, though. The incidence rate of cervical cancer among Hispanic women is 28% higher than for white women, and Black women are more likely to die of cervical cancer than any other racial or ethnicities.
Whether the Pap is administered every year, every three years or not at all, Papanicolaou will always be known as the medical hero who saved countless women who would otherwise have succumbed to cervical cancer.
Science Has Given Us the Power to Undermine Nature's Deadliest Creature: Should We Use It?
The Aedes aegypti mosquito, which can carry devastating diseases, was recently engineered by a biotech company to have a genetic "kill switch" intended to crash the local population in the Florida Keys.
Lurking among the swaying palm trees, sugary sands and azure waters of the Florida Keys is the most dangerous animal on earth: the mosquito.
While there are thousands of varieties of mosquitoes, only a small percentage of them are responsible for causing disease. One of the leading culprits is Aedes aegypti, which thrives in the warm standing waters of South Florida, Central America and other tropical climes, and carries the viruses that cause yellow fever, dengue, chikungunya and Zika.
Dengue, a leading cause of death in many Asian and Latin American countries, causes bleeding and pain so severe that it's referred to as "breakbone fever." Chikungunya and yellow fever can both be fatal, and Zika, when contracted by a pregnant woman, can infect her fetus and cause devastating birth defects, including a condition called microcephaly. Babies born with this condition have abnormally small heads and lack proper brain development, which leads to profound, lifelong disabilities.
Decades of efforts to eradicate the disease-carrying Aedes aegypti mosquito from the Keys and other tropical locales have had limited impact. Since the advent of pesticides, homes and neighborhoods have been drenched with them, but after each spraying, the mosquito population quickly bounces back, and the pesticides have to be sprayed over and over. But thanks to genetic engineering, new approaches are underway that could possibly prove safer, cheaper and more effective than any pesticide.
One of those approaches involves, ironically, releasing more mosquitoes in the Florida Keys.
The kill-switch will ensure that the female offspring die before they reach maturity and thus, be unable to reproduce.
British biotech company Oxitec has engineered male mosquitoes to have a genetic "kill-switch" that could potentially crash the local population of Aedes aegypti, at least in the short-term. The modified males that are being released are intended to mate with wild females.
Males don't bite; it's the female that's deadly, always seeking out blood to gorge on to help mature her eggs. After settling her filament-thin legs on her prey, she sinks a needlelike proboscis into the skin and sucks the blood until her translucent belly is bloated and glowing red.
The kill-switch will ensure that the female offspring die before they reach maturity and thus, be unable to reproduce. In some experiments using genetically modified mosquitoes, the small number of females that survived were rendered unable to bite. The modification prevented the proboscis, the sickle-like needle that pierces the skin, from forming properly. But this isn't the case with Oxitec's mosquitoes; in the Oxitec release, the females simply die off before they can mate.
The modified mosquitoes are the second genetically engineered insect to be released in the U.S. by Oxitec. The first was a modified diamondback moth, an agricultural pest that doesn't bite humans. But with the mosquitoes, there are many questions about the long-term effects on wild ecosystems, other species in the food chain, and human health. With the Keys initiative, there has been vociferous opposition from environmental groups and some local residents, but some scientists and public health experts say that genetically modified insects pose less of a risk than the diseases they carry and the powerful, indiscriminant pesticides used to combat them.
Oxitec spent a decade developing the technology and engaging in a massive public education campaign before beginning the field test in April. Eventually, the company will release 750,000 of the insects from six locations on three islands of the Florida Keys. Although the release has been approved by the Environmental Protection Agency, the Florida Department of Agriculture and Consumer Services, and the Florida Keys Mosquito Control District, the company was never able to obtain unanimous approval among local residents, some of whom worry that the experiment could cause irreversible damage to the ecosystem.
The company has already begun distributing multiple blue and white boxes containing the eggs of thousands of the mosquitoes which, when water is added, will hatch legions of modified males.
There are a number of techniques available to genetically engineer animals and plants to minimize disease and maximize crop yields. According to Kevin Gorman, chief development officer for Oxitec, the company's mosquitoes were altered by injecting genetic material into the eggs, testing them, then re-injecting them if not enough of the new genes were incorporated into the developing embryos. "We insert genes, but take nothing away," he says.
Gorman points out that the Oxitec mosquitoes will only pass the kill-switch genes on to some of their offspring, and that they will die out fairly quickly. They should temporarily lessen diseases by reducing the local population of Aedes aegypti, but to have a long-term effect, repeated introductions of the altered mosquitoes would have to take place.
Critics say the Oxitec experiment is a precursor to a far more consequential, and more troubling development: the introduction of gene drives in modified species that aggressively tilt inheritance factors in a decided direction.
Gene Drives
Gene drives coupled with the recent development of the gene-editing technique, CRISPR-Cas9, promise to be far more targeted and powerful than previous gene altering efforts. Gene drives override the normal laws of inheritance by harnessing natural processes involved in reproduction. The technique targets small sections of the animal's DNA and replaces it with an altered allele, or trait-determining snippet. Normally, when two members of a species mate, the offspring have a 50 percent chance of receiving an allele because they will receive one from each parent. But in a gene drive, each offspring ends up getting two copies of a desired allele from a single parent—the modified parent. The method "drives" the modified DNA into up to 100 percent of the animals' offspring.
In the case of gene drive mosquitoes, the modified males will mate with wild females. Upon fertilization of the egg, the offspring will start off with one copy of the targeted allele from each parent. But an enzyme, called Cas9, is introduced and acts as a kind of molecular scissors to cut, or damage, the "wild" allele. Then the developing embryo's genetic repair mechanisms kick in and, to repair the damage, copy the undamaged allele from the modified parent. In this way, the offspring ends up with two copies of the modified allele, and it will pass the modification on to virtually all of its progeny.
There is some debate among researchers and others about what constitutes a gene drive, but leaders in the nascent field, such as Andrea Crisanti, generally agree that the defining factor is the heritability of a change introduced into a species. A gene drive is not a particular gene or suite of genes, but a program that proliferates in a species because it is inherited by virtually all offspring.
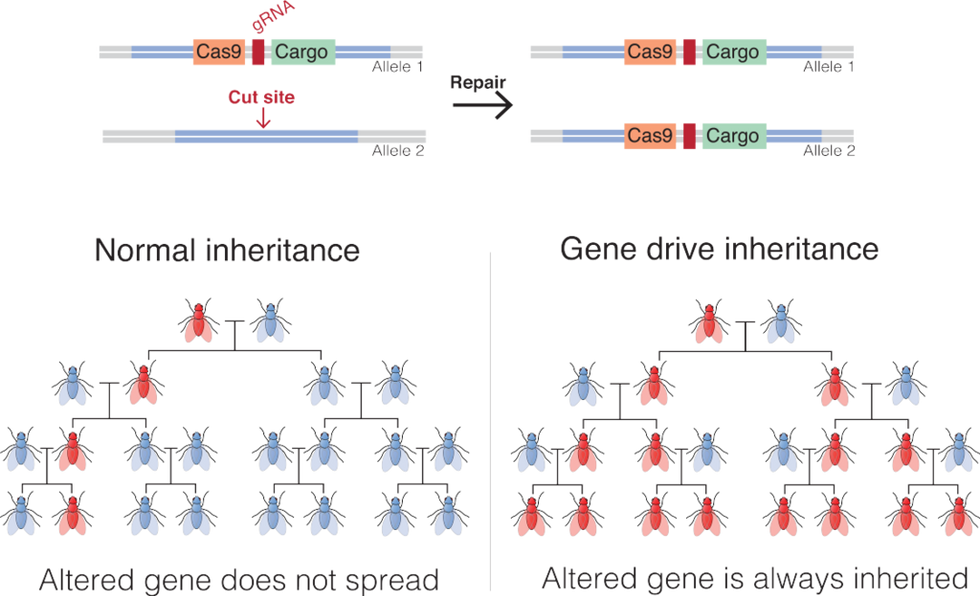
An illustration of how gene drives spread an altered gene through a population.
Mariuswalter, CC BY-SA 4.0 <https://creativecommons.org/licenses/by-sa/4.0>, via Wikimedia Commons
Of the experts who spoke with Leaps.org for this article, there was disagreement on whether the Oxitec mosquitoes carry a gene drive, but Gorman says they don't because they carry no inheritance advantage. The mosquitoes have baked-in limitations on their potential impact on the tropical ecosystem because the kill-switch should only temporarily affect the local population of Aedes aegypti. The modified mosquitoes will die pretty quickly. But modified organisms that do carry gene drives have the potential to spread widely and persist for an unknown period of time.
Since it has such a reproductive advantage, animals modified by CRISPR and carrying gene drives can quickly replace wild species that compete with them. On the other hand, if the gene drive carries a kill-switch, it can theoretically cause a whole species to collapse.
This makes many people uneasy in an age of mass extinctions, when animals and ecosystems are already under extreme stress due to climate change and the ceaseless destruction of their habitats. Ecosystems are intricate, delicately balanced mosaics where one animal's competitor is another animal's food. The interconnectedness of nature is only partially understood and still contains many mysteries as to what effects human intervention could eventually cause.
But there's a compelling case to be made for the use of gene drives in general. Economies throughout the world are often based on the ecosystem and its animals, which rely on a natural food chain that was evolved over billions of years. But diseases carried by mosquitoes and other animals cause massive damage, both economically and in terms of human suffering.
Malaria alone is a case in point. In 2019, the World Health Organization reported 229 million cases of malaria, which led to 449,000 deaths worldwide. Over 70 percent of those deaths were in children under the age of 12. Efforts to combat malaria-carrying mosquitoes rely on fogging the home with chemical pesticides and sleeping under pesticide-soaked nets, and while this has reduced the occurrence of malaria in recent years, the result is nowhere near as effective as eradicating the Anopheles gambiae mosquito that carries the disease.
Pesticides, a known carcinogen for animals and humans, are a blunt instrument, says Anthony Shelton, a biologist and entomologist at Cornell University. "There are no pesticides so specific that they just get the animal you want to target. They get pollinators. They get predators and parasites. They negatively affect the ecosystem, and they get into our bodies." And it's not uncommon for insects to develop resistance to pesticides, necessitating the continuous development of new, more powerful chemicals to control them.
"The harm of insecticides is not debatable," says Shelton. With gene drives, the potential harm is less clear.
Shelton also points out that although genetic modification sounds radical, people have been altering the genes of animals since before recorded history, through the selective breeding of farm and domesticated animals. While critics of genetic modification decry the possibility of changing the trajectory of evolution in animals, "We've been doing it for centuries," says Shelton. "Gene drives are just a much faster way to do what we've been doing all along."
Still, one might argue that farms are closed experiments, because animals enclosed within farms don't mate with wild animals. This limits the impact of human changes on the larger ecosystem. And getting new genes to work their way through multiple generations in longer-lived animals through breeding can take centuries, which imposes the element of time to ascertain the relative benefits of any introduced change. Gene drives fast-forward change in ways that have never been harnessed before.
The unique thing about gene drives, Shelton says, is that they only affect the targeted species, because those animals will only breed with their own species. Although the Oxitec mosquitoes are modified but not imbued with a gene drive, they illustrate the point. Aedes aegypti will only mate with its own species, and not with any of the other 3,000 varieties of mosquito. According to Shelton, "If they were to disappear, it would have no effect on the fish, bats and birds that feed on them." But should gene drives become widely used, this won't always be true of animals that play a larger part in the food chain. This will be especially true if gene drives are used in mammals.
One factor, cited by both proponents of gene drives and those who want a complete moratorium on them, is that once a gene drive is released into the wild, animals tend to evolve strategies to resist them. In a 2017 article in Nature, Philip Messer, a population geneticist at Cornell, says that gene drives create "the ideal conditions for resistant organisms to flourish."
Sometimes, when CRISPR is used and the Cas9 enzyme cuts an allele soon after egg fertilization, the animal's repair mechanism, rather than creating a straight copy of the desired allele, inserts random DNA letters. The gene drive won't recognize the new sequence, and the change will slip through. In this way, nature has a way of overriding gene drives.
In caged experiments using CRISPR-modified mosquitoes, while the gene drive initially worked, resistance has developed fairly rapidly. Scientists working for Target Malaria, the massive anti-malaria enterprise funded by the Bill and Melinda Gates Foundation, are now working on developing a new version of a gene drive that is not so vulnerable to genetic resistance. But cage conditions are not representative of complex natural ecosystems, and to figure out how a modified species is going to affect the big picture, ultimately they will have to be tested in the wild.
Because there are so many unknowns, such testing is just too dangerous to undertake, according to environmentalists such as Dana Perls of the Friends of the Earth, an international consortium of environmental organizations headquartered in Amsterdam. "There's no safe way to experiment in the wild," she says. "Extinction is permanent, and to drive any species to extinction could have major environmental problems. At a time when we're seeing species disappearing at a high rate, we need to focus on safe processes and a slow approach rather than assume there's a silver bullet."
She cites a number of possible harmful outcomes from genetic modification, including the possible creation of dangerous hybrids that could be more effective at spreading disease and more resistant to pesticides. She points to a 2019 paper in Scientific Reports in which Yale researchers suggested there's evidence that genetically modified species can interbreed with organisms outside their own species. The researchers claimed that when Oxitec tested its modified Aedes aegypti mosquitoes in Brazil, the release resulted in a dangerous hybrid due to the altered animals breeding with two other varieties of mosquito. They suggested that the hybrid mosquito was more robust than the original gene drive mosquitoes.
The paper contributed to breathless headlines in the media and made a big splash with the anti-GMO community. However, it turned out that when other scientists reviewed the data, they found it didn't support the authors' claims. In a short time, the editors of Nature ran an Editorial Expression of Concern for the article, noting that of the insects examined by the researchers, none of them contained the transgenes of the released mosquitoes. Among multiple concerns, Nature found that the researchers didn't follow the released population for more than a short time, and that previous work from the same authors had shown that after a short time, transgenes would have faded from the population.
Of course, unintended consequences are always a concern any time we interfere with nature, says Michael Montague, a senior scholar at Johns Hopkins University's Center for Health Security. "Unpredictability is part of living in the world," he says. Still, he's relatively comfortable with the limited Florida Keys release.
"Even if one type of mosquito was eliminated in the Keys, the ecosystem wouldn't notice," he says. This is because of the thousands of other species of mosquito. He says that while the Keys initiative is ultimately a test, "Oxitec has done their due diligence."
Montague addressed another concern voiced by Perls. The Oxitec mosquitoes were developed so that the female larvae will only hatch in water containing the antibiotic tetracycline. Perls and others caution that, because of the widespread use of antibiotics, the drug inevitably makes its way into the water system, and could be present in the standing pools of water that mosquitoes mate and lay their eggs in.
It's highly unlikely that tetracycline would exist in concentrations high enough to make any difference, says Montague. "But even if it did happen, and the modified females hatched out and mated with wild males, many of their offspring would inherit the modification and only be able to hatch in tetracycline-laced water. The worst-case scenario would be that the pest control didn't work. Net effect: Zero," he says.
As for comparing GMO mosquitoes with insecticides, Montague says, "We 100 percent know insecticides have a harmful effect on human health, whereas modified [male] mosquitoes don't bite humans. They're essentially a chemical-free insecticide, and if there were to be some harmful effect on human health, it would have to be some complicated, convoluted effect" that no one has predicted.
It's not clear, though, given the transitory nature of self-limiting genetically modified insects, whether any effects on the ecosystem would be long-lasting. Certainly in the case of the Oxitec mosquitoes, any effect on the environment would likely be subtle. However, there are other species that are far more important to the food chain, and humans have been greatly impacting them for centuries, sometimes with disastrous effects.
The world's oceans are particularly vulnerable to the effects of human actions. "Codfish used to dominate the North Atlantic ecosystem," says Montague, but due to overfishing, there were huge changes to that ecosystem, including the expansion of their prey—lobsters, crabs and shrimp. The whole system got out of balance." The fish illustrate the international nature of the issues related to gene drives, because wild species have few boundaries and a change in one region can easily spread far and wide.
On the other hand, gene drives can be used for beneficial purposes beyond eliminating disease-carrying species. They could also be used to combat invasive species, fight crop-destroying insects, promote biodiversity, and give a leg up to endangered species that would otherwise die out.
Today nearly 90 percent of the world's islands have been invaded by disease-carrying rodents that have over-multiplied and are driving other island species to extinction. Common rodents such as rats and mice normally encounter a large number of predators in mainland territories, and this controls their numbers. Once they are introduced into island ecosystems, however, they have few predators and often become invasive. Because of this, they are a prevalent cause of the extinction of both animals and plants globally. The primary way to combat them has been to spread powerful toxicants that, when ingested, cause death. Not only has this inhumane practice had limited impact, the toxicants can be eaten by untargeted species and are toxic to humans.
The Genetic Biocontrol of Invasive Rodents program (GBIRd), an international consortium of scientists, ethicists, regulatory experts, sociologists, conservationists and others, is exploring the possible development of a genetically modified mouse that could be introduced to islands where rodents are invasive. Similar to the Oxitec mosquitoes, the mice would carry a modification that results in the appearance of only one sex, and they would also carry a gene drive. Theoretically, once they mate with the wild mice, all of the surviving offspring would be either male or female, and the species would disappear from the islands, giving other, threatened species an opportunity to revive.
GBIRd is moving slowly by design and is currently focused on asking if a genetically engineered mouse should be developed. The program is a potential model for how gene drives can be ethically developed with maximum foresight and the least impact on complex ecosystems. By first releasing a genetically engineered mouse on an island — likely years from now — the impact would naturally be contained within a limited locale.
Regulating GM Insects
While multiple agencies in the U.S. were involved in approving the release of the Oxitec mosquitoes, most experts agree that there is not a straightforward path to regulating genetically modified organisms released into the environment. Clearly, international regulation is needed as genetically modified organisms are released into open environments like the air and the ocean.
The United Nations' Convention on Biological Diversity, which oversees environmental issues at an international level, recently met to continue a process of hammering out voluntary protocols concerning gene drives. Multiple nations have already signed on to already-established protocols, but the United States has not and, according to Montague, is not expected to. "The U.S. will never be signatory to CBD agreements because agricultural companies are huge businesses" that may not see them as in their best interests, he says. Bans or limitations on the release of genetically modified organisms could limit crop yields, for example, thereby limiting profits.
Even if every nation signed on to international regulations of gene drives, cooperation is voluntary. The regulations wouldn't prevent bad actors from using the technology in nefarious ways, such as developing gene drives that can be used as weapons, according to Perls. An example would be unleashing a genetically modified invasive insect to destroy the crops of enemy nations. Or the releasing of a swarm of disease-carrying insects. But in this scenario, it would be very hard to limit the genetically modified species to a specific environment, and the bad actors could be unleashing disaster on themselves.
Because of the risks of misuse, scientists disagree on whether to openly share their gene drive research with others. But Montague believes that there should be a universal registry of gene drives, because "one gene drive can mess up another one. Two groups using the same species should know about each other," he says.
Ultimately, the decision of whether and when to release gene drives into nature rests with not one group, but with society as a whole. This includes not only diverse experts and regulatory bodies, but the general public, a group Oxitec spent considerable time and resources interacting with for their Florida Keys project. In the end, they gained approval for the initiative by a majority of Keys residents, but never gained a total consensus.
There's no escaping the fact that the use of gene drives is a nascent field, and even geneticists and regulators are still grapping with the best ways to develop, oversee, regulate, and control them. Much more data is needed to fully ascertain its risks and benefits.
Experts agree that the Oxitec venture isn't likely to have a noticeable effect on the larger ecosystem unless something truly catastrophic goes wrong. But following the GMO mosquitoes over time will give scientists more real-world data about the long-term effects of genetically altered species. If the release doesn't work, nothing about the ecosystem will change and Aedes aegypti will continue to be a menace to human health. But if something goes horribly wrong, it could hinder the field for years, if not forever.
On the other hand, if the Oxitec mosquitoes and other early initiatives achieve their goals of reducing disease, increasing crop yields, and protecting biodiversity, in the words of Anthony Shelton, "Maybe, 25 to 50 years from now, people will wonder what all the fuss was about."
Correction: The original version of this article mistakenly stated that the modified Oxitec mosquitoes would not be able to form a proper proboscis to bite humans. That is true for some modified mosquitoes but not the Oxitec ones, whose female offspring die off before they reach maturity. Additionally, the Oxitec release was not approved by the FDA and CDC, as originally stated. The FDA and CDC withdrew their role and passed the oversight to other regulatory entities.

