Meet the Psychologist Using Psychedelics to Treat Racial Trauma

Monnica Williams was stuck. The veteran psychologist wanted to conduct a study using psychedelics, but her university told her they didn't have the expertise to evaluate it via an institutional review board, which is responsible for providing ethical and regulatory oversight for research that involves human participants. Instead, they directed her to a hospital, whose reviewers turned it down, citing research of a banned substance as unethical.
"I said, 'We're not using illegal psilocybin, we're going through Health Canada,'" Williams said. Psilocybin was banned in Canada in 1974, but can now be obtained with an exemption from Health Canada, the federal government's health policy department. After learning this, the hospital review board told Williams they couldn't review her proposal because she's not affiliated with the hospital, after all.
It's all part of balancing bureaucracy with research goals for Williams, a leading expert on racial trauma and psychedelic medicine, as well as obsessive compulsive disorder (OCD), at the University of Ottawa. She's exploring the use of hallucinogenic substances like MDMA and psilocybin — commonly known as ecstasy and magic mushrooms, respectively — to help people of color address the psychological impacts of systemic racism. A prolific researcher, Williams also works as an expert witness, offering clinical evaluations for racial trauma cases.
Scientists have long known that psychedelics produce an altered state of consciousness and openness to new perspectives. For people with mental health conditions who haven't benefited from traditional therapy, psychedelics may be able to help them discover what's causing their pain or trauma, including racial trauma—the mental and emotional injury spurred by racial bias.
"Using psychedelics can not only bring these pain points to the surface for healing, but can reduce the anxiety or response to these memories and allow them to speak openly about them without the pain they bring," Williams says. Her research harnesses the potential of psychedelics to increase neuroplasticity, which includes the brain's ability to build new pathways.
"People of color are dealing with racism all the time, in large and small ways, and even dealing with racism in healthcare, even dealing with racism in therapy."
But she says therapists of color aren't automatically equipped to treat racial trauma. First, she notes, people of color are "vastly underrepresented in the mental health workforce." This is doubly true in psychedelic-assisted psychotherapy, in which a person is guided through a psychedelic session by a therapist or team of therapists, then processes the experience in subsequent therapy sessions.
"On top of that, the therapists of color are getting the same training that the white therapists are getting, so it's not even really guaranteed that they're going to be any better at helping a person that may have racial trauma emerging as part of their experience," she says.
In her own training to become a clinical psychologist at the University of Virginia, Williams says she was taught "how to be a great psychologist for white people." Yet even people of color, she argues, need specialized training to work with marginalized groups, particularly when it comes to MDMA, psilocybin and other psychedelics. Because these drugs can lower natural psychological defense mechanisms, Williams says, it's important for providers to be specially trained.
"People of color are dealing with racism all the time, in large and small ways, and even dealing with racism in healthcare, even dealing with racism in therapy. So [they] generally develop a lot of defenses and coping strategies to ward off racism so that they can function." she says. This is particularly true with psychedelic-assisted psychotherapy: "One possibility is that you're going to be stripped of your defenses, you're going to be vulnerable. And so you have to work with a therapist who is going to understand that and not enact more racism in their work with you."
Williams has struggled to find funding and institutional approval for research involving psychedelics, or funding for investigations into racial trauma or the impacts of conditions like OCD and post-traumatic stress disorder (PTSD) in people of color. With the bulk of her work focusing on OCD, she hoped to focus on people of color, but found there was little funding for that type of research. In 2020, that started to change as structural racism garnered more media attention.
After the killing of George Floyd, a 46-year-old Black man, by a white police officer in May 2020, Williams was flooded with media requests. "Usually, when something like that happens, I get contacted a lot for a couple of weeks, and it dies off. But after George Floyd, it just never did."
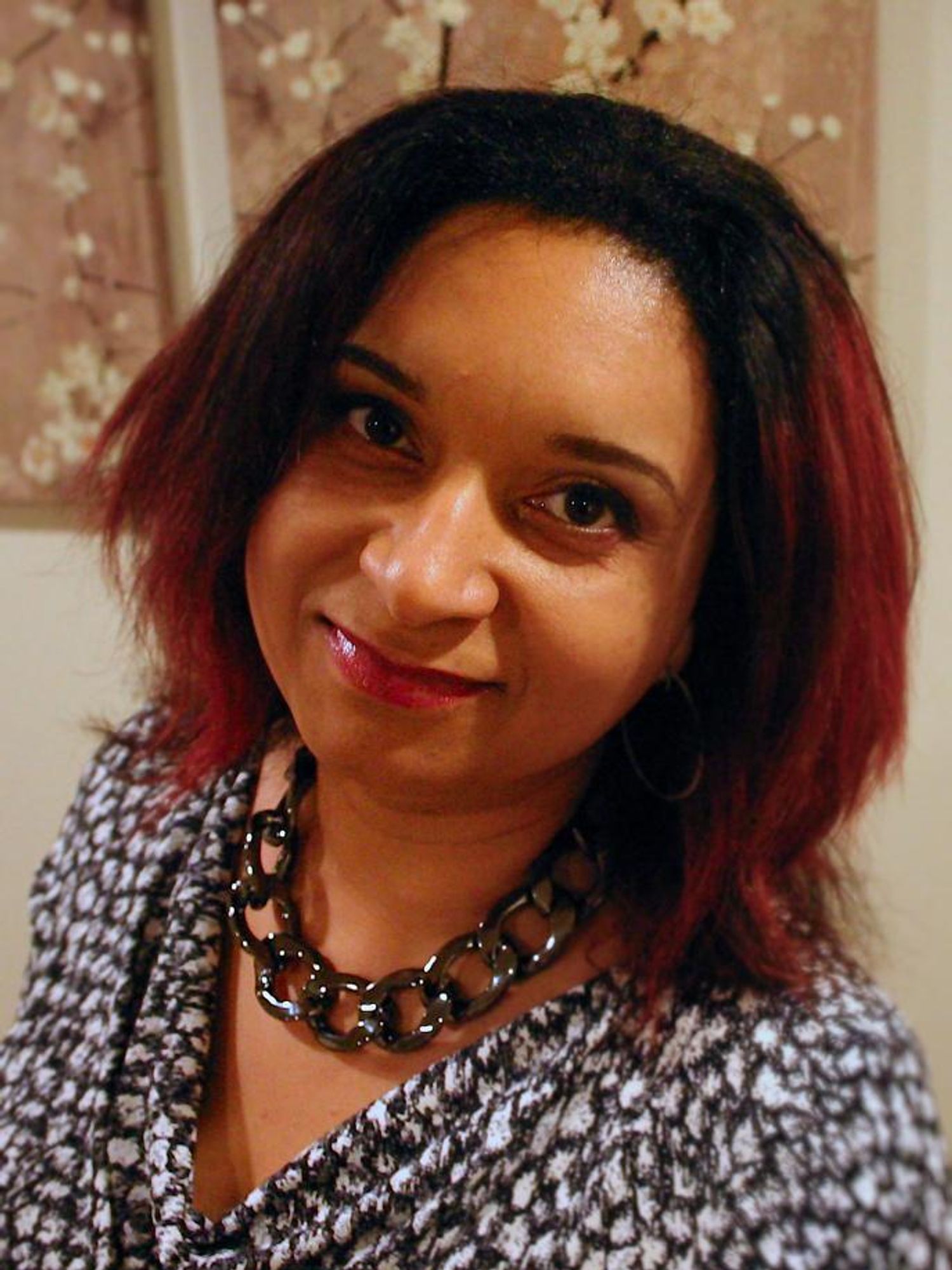
Monnica Williams, clinical psychologist at the University of Ottawa
Williams was no stranger to the questions that soon blazed across headlines: How can we mitigate microaggressions? How do race and ethnicity impact mental health? What terms should we use to discuss racial issues? What constitutes an ally, and why aren't there more of them? Why aren't there more people of color in academia, and so many other fields?
Now, she's hoping that the increased attention on racial justice will mean more acceptance for the kind of research she's doing.
In fact, Williams herself has used psychedelics in order to gain a better understanding of how to use them to treat racial trauma. In a study published in January, she and two other Black female psychotherapists took MDMA in a supervised setting, guided by a team of mental health practitioners who helped them process issues that came up as the session progressed. Williams, who was also the study's lead author, found that participants' experiences centered around processing and finding release from racial identities, and, in one case, of simply feeling wholly human without the burden of racial identity for the first time.
The purpose of the study was twofold: to understand how Black women react to psychedelics and to provide safe, firsthand, psychedelic experiences to Black mental health practitioners. One of the other study participants has since gone on to offer psychedelic-assisted psychotherapy to her own patients.
Psychedelic research, and psilocybin in particular, has become a hot topic of late, particularly after Oregon became the first state to legalize it for therapeutic use last November. A survey-based, observational study with 313 participants, published in 2020, paved the way for Williams' more recent MDMA experiments by describing improvements in depression, anxiety and racial trauma among people of color who had used LSD, psilocybin or MDMA in a non-research setting.
Williams and her team included only respondents who reported a moderate to strong psychoactive effect of past psychedelic consumption and believed these experiences provided "relief from the challenging effects of ethnic discrimination." Participants reported a memorable psychedelic experience as well as its acute and lasting effects, completing assessments of psychological insight, mystical experience and emotional challenges experienced during psychedelic experience, then describing their mental health — including depression, anxiety and trauma symptoms — before and after that experience.
Still, Williams says addressing racism is much more complex than treating racial trauma. "One of the questions I get asked a lot is, 'How can Black people cope with racism?' And I don't really like that question," she says. "I think it's important and I don't mind answering it, but I think the more important question is, how can we end racism? What can Black people do to stop racism that's happening to them and what can we do as a society to stop racism? And people aren't really asking this question."
A new injection is helping stave off RSV this season
The FDA approved a single-dose, long-acting injection to protect babies and toddlers from RSV over the fall and winter.
In November 2021, Mickayla Wininger’s then one-month-old son, Malcolm, endured a terrifying bout with RSV, the respiratory syncytial (sin-SISH-uhl) virus—a common ailment that affects all age groups. Most people recover from mild, cold-like symptoms in a week or two, but RSV can be life-threatening in others, particularly infants.
Wininger, who lives in southern Illinois, was dressing Malcolm for bed when she noticed what seemed to be a minor irregularity with this breathing. She and her fiancé, Gavin McCullough, planned to take him to the hospital the next day. The matter became urgent when, in the morning, the boy’s breathing appeared to have stopped.
After they dialed 911, Malcolm started breathing again, but he ended up being hospitalized three times for RSV and defects in his heart. Eventually, he recovered fully from RSV, but “it was our worst nightmare coming to life,” Wininger recalled.
It’s a scenario that the federal government is taking steps to prevent. In July, the Food and Drug Administration approved a single-dose, long-acting injection to protect babies and toddlers. The injection, called Beyfortus, or nirsevimab, became available this October. It reduces the incidence of RSV in pre-term babies and other infants for their first RSV season. Children at highest risk for severe RSV are those who were born prematurely and have either chronic lung disease of prematurity or congenital heart disease. In those cases, RSV can progress to lower respiratory tract diseases such as pneumonia and bronchiolitis, or swelling of the lung’s small airway passages.
Each year, RSV is responsible for 2.1 million outpatient visits among children younger than five-years-old, 58,000 to 80,000 hospitalizations in this age group, and between 100 and 300 deaths, according to the Centers for Disease Control and Prevention. Transmitted through close contact with an infected person, the virus circulates on a seasonal basis in most regions of the country, typically emerging in the fall and peaking in the winter.
In August, however, the CDC issued a health advisory on a late-summer surge in severe cases of RSV among young children in Florida and Georgia. The agency predicts "increased RSV activity spreading north and west over the following two to three months.”
Infants are generally more susceptible to RSV than older people because their airways are very small, and their mechanisms to clear these passages are underdeveloped. RSV also causes mucus production and inflammation, which is more of a problem when the airway is smaller, said Jennifer Duchon, an associate professor of newborn medicine and pediatrics in the Icahn School of Medicine at Mount Sinai in New York.
In 2021 and 2022, RSV cases spiked, sending many to emergency departments. “RSV can cause serious disease in infants and some children and results in a large number of emergency department and physician office visits each year,” John Farley, director of the Office of Infectious Diseases in the FDA’s Center for Drug Evaluation and Research, said in a news release announcing the approval of the RSV drug. The decision “addresses the great need for products to help reduce the impact of RSV disease on children, families and the health care system.”
Sean O’Leary, chair of the committee on infectious diseases for the American Academy of Pediatrics, says that “we’ve never had a product like this for routine use in children, so this is very exciting news.” It is recommended for all kids under eight months old for their first RSV season. “I would encourage nirsevimab for all eligible children when it becomes available,” O’Leary said.
For those children at elevated risk of severe RSV and between the ages of 8 and 19 months, the CDC recommends one dose in their second RSV season.
The drug will be “really helpful to keep babies healthy and out of the hospital,” said O’Leary, a professor of pediatrics at the University of Colorado Anschutz Medical Campus/Children’s Hospital Colorado in Denver.
An antiviral drug called Synagis (palivizumab) has been an option to prevent serious RSV illness in high-risk infants since it was approved by the FDA in 1998. The injection must be given monthly during RSV season. However, its use is limited to “certain children considered at high risk for complications, does not help cure or treat children already suffering from serious RSV disease, and cannot prevent RSV infection,” according to the National Foundation for Infectious Diseases.
Until the approval this summer of the new monoclonal antibody, nirsevimab, there wasn’t a reliable method to prevent infection in most healthy infants.
Both nirsevimab and palivizumab are monoclonal antibodies that act against RSV. Monoclonal antibodies are lab-made proteins that mimic the immune system’s ability to fight off harmful pathogens such as viruses. A single intramuscular injection of nirsevimab preceding or during RSV season may provide protection.
The strategy with the new monoclonal antibody is “to extend protection to healthy infants who nonetheless are at risk because of their age, as well as infants with additional medical risk factors,” said Philippa Gordon, a pediatrician and infectious disease specialist in Brooklyn, New York, and medical adviser to Park Slope Parents, an online community support group.
No specific preventive measure is needed for older and healthier kids because they will develop active immunity, which is more durable. Meanwhile, older adults, who are also vulnerable to RSV, can receive one of two new vaccines. So can pregnant women, who pass on immunity to the fetus, Gordon said.
Until the approval this summer of the new monoclonal antibody, nirsevimab, there wasn’t a reliable method to prevent infection in most healthy infants, “nor is there any treatment other than giving oxygen or supportive care,” said Stanley Spinner, chief medical officer and vice president of Texas Children’s Pediatrics and Texas Children’s Urgent Care.
As with any virus, washing hands frequently and keeping infants and children away from sick people are the best defenses, Duchon said. This approach isn’t foolproof because viruses can run rampant in daycare centers, schools and parents’ workplaces, she added.
Mickayla Wininger, Malcolm’s mother, insists that family and friends wear masks, wash their hands and use hand sanitizer when they’re around her daughter and two sons. She doesn’t allow them to kiss or touch the children. Some people take it personally, but she would rather be safe than sorry.
Wininger recalls the severe anxiety caused by Malcolm's ordeal with RSV. After returning with her infant from his hospital stays, she was terrified to go to sleep. “My fiancé and I would trade shifts, so that someone was watching over our son 24 hours a day,” she said. “I was doing a night shift, so I would take caffeine pills to try and keep myself awake and would end up crashing early hours in the morning and wake up frantically thinking something happened to my son.”
Two years later, her anxiety has become more manageable, and Malcolm is doing well. “He is thriving now,” Wininger said. He recently had his second birthday and "is just the spunkiest boy you will ever meet. He looked death straight in the eyes and fought to be here today.”
AI was integral to creating Moderna's mRNA vaccine against COVID.
Story by Big Think
For most of history, artificial intelligence (AI) has been relegated almost entirely to the realm of science fiction. Then, in late 2022, it burst into reality — seemingly out of nowhere — with the popular launch of ChatGPT, the generative AI chatbot that solves tricky problems, designs rockets, has deep conversations with users, and even aces the Bar exam.
But the truth is that before ChatGPT nabbed the public’s attention, AI was already here, and it was doing more important things than writing essays for lazy college students. Case in point: It was key to saving the lives of tens of millions of people.
AI-designed mRNA vaccines
As Dave Johnson, chief data and AI officer at Moderna, told MIT Technology Review‘s In Machines We Trust podcast in 2022, AI was integral to creating the company’s highly effective mRNA vaccine against COVID. Moderna and Pfizer/BioNTech’s mRNA vaccines collectively saved between 15 and 20 million lives, according to one estimate from 2022.
Johnson described how AI was hard at work at Moderna, well before COVID arose to infect billions. The pharmaceutical company focuses on finding mRNA therapies to fight off infectious disease, treat cancer, or thwart genetic illness, among other medical applications. Messenger RNA molecules are essentially molecular instructions for cells that tell them how to create specific proteins, which do everything from fighting infection, to catalyzing reactions, to relaying cellular messages.
Johnson and his team put AI and automated robots to work making lots of different mRNAs for scientists to experiment with. Moderna quickly went from making about 30 per month to more than one thousand. They then created AI algorithms to optimize mRNA to maximize protein production in the body — more bang for the biological buck.
For Johnson and his team’s next trick, they used AI to automate science, itself. Once Moderna’s scientists have an mRNA to experiment with, they do pre-clinical tests in the lab. They then pore over reams of data to see which mRNAs could progress to the next stage: animal trials. This process is long, repetitive, and soul-sucking — ill-suited to a creative scientist but great for a mindless AI algorithm. With scientists’ input, models were made to automate this tedious process.
“We don’t think about AI in the context of replacing humans,” says Dave Johnson, chief data and AI officer at Moderna. “We always think about it in terms of this human-machine collaboration, because they’re good at different things. Humans are really good at creativity and flexibility and insight, whereas machines are really good at precision and giving the exact same result every single time and doing it at scale and speed.”
All these AI systems were in put in place over the past decade. Then COVID showed up. So when the genome sequence of the coronavirus was made public in January 2020, Moderna was off to the races pumping out and testing mRNAs that would tell cells how to manufacture the coronavirus’s spike protein so that the body’s immune system would recognize and destroy it. Within 42 days, the company had an mRNA vaccine ready to be tested in humans. It eventually went into hundreds of millions of arms.
Biotech harnesses the power of AI
Moderna is now turning its attention to other ailments that could be solved with mRNA, and the company is continuing to lean on AI. Scientists are still coming to Johnson with automation requests, which he happily obliges.
“We don’t think about AI in the context of replacing humans,” he told the Me, Myself, and AI podcast. “We always think about it in terms of this human-machine collaboration, because they’re good at different things. Humans are really good at creativity and flexibility and insight, whereas machines are really good at precision and giving the exact same result every single time and doing it at scale and speed.”
Moderna, which was founded as a “digital biotech,” is undoubtedly the poster child of AI use in mRNA vaccines. Moderna recently signed a deal with IBM to use the company’s quantum computers as well as its proprietary generative AI, MoLFormer.
Moderna’s success is encouraging other companies to follow its example. In January, BioNTech, which partnered with Pfizer to make the other highly effective mRNA vaccine against COVID, acquired the company InstaDeep for $440 million to implement its machine learning AI across its mRNA medicine platform. And in May, Chinese technology giant Baidu announced an AI tool that designs super-optimized mRNA sequences in minutes. A nearly countless number of mRNA molecules can code for the same protein, but some are more stable and result in the production of more proteins. Baidu’s AI, called “LinearDesign,” finds these mRNAs. The company licensed the tool to French pharmaceutical company Sanofi.
Writing in the journal Accounts of Chemical Research in late 2021, Sebastian M. Castillo-Hair and Georg Seelig, computer engineers who focus on synthetic biology at the University of Washington, forecast that AI machine learning models will further accelerate the biotechnology research process, putting mRNA medicine into overdrive to the benefit of all.
This article originally appeared on Big Think, home of the brightest minds and biggest ideas of all time.