Can AI help create “smart borders” between countries?
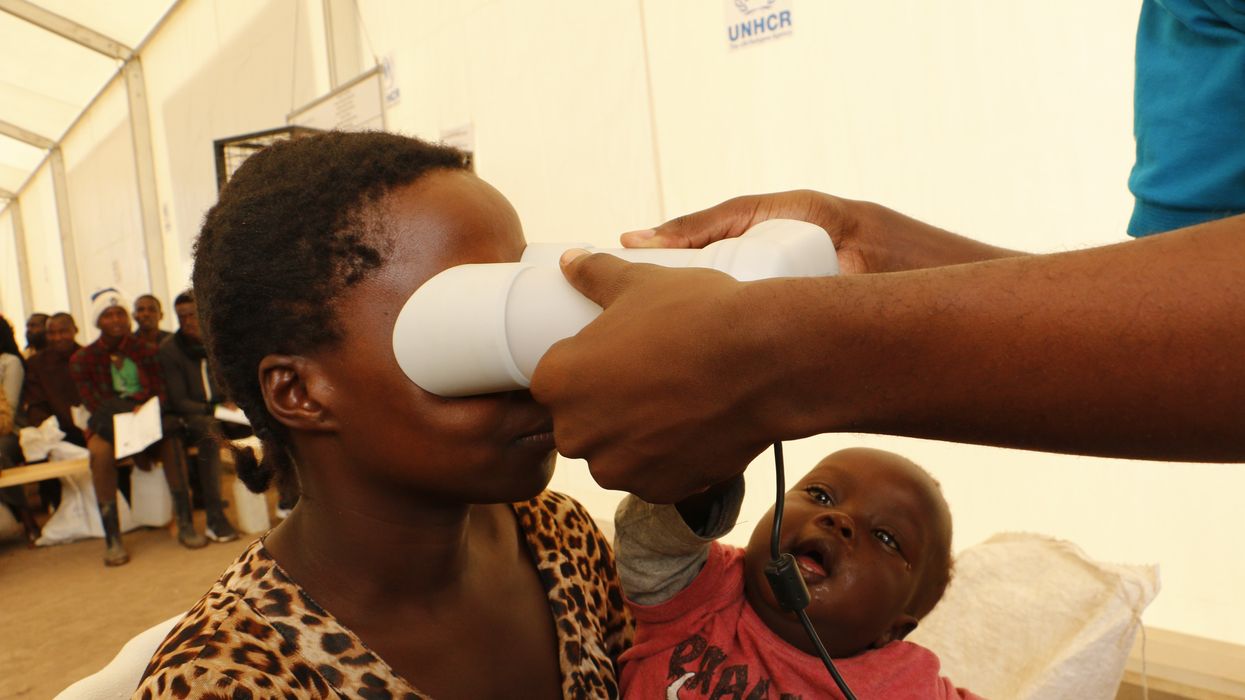
A refugee in Uganda’s Oruchinga settlement uses an iris scan to claim food assistance.
In 2016, border patrols in Greece, Latvia and Hungary received a prototype for an AI-powered lie detector to help screen asylum seekers. The detector, called iBorderCtrl, was funded by the European Commission in hopes to eventually mitigate refugee crises like the one sparked by the Syrian civil war a year prior.
iBorderCtrl, which analyzes micro expressions in the face, received but one slice of the Commission’s €34.9 billion border control and migration management budget. Still in development is the more ambitious EuMigraTool, a predictive AI system that will process internet news and social media posts to estimate not only the number of migrants heading for a particular country, but also the “risks of tensions between migrants and EU citizens.”
Both iBorderCtrl and EuMigraTool are part of a broader trend: the growing digitization of migration-related technologies. Outside of the EU, in refugee camps in Jordan, the United Nations introduced iris scanning software to distribute humanitarian aid, including food and medicine. And in the United States, Customs and Border Protection has attempted to automate its services through an app called CBP One, which both travelers and asylum seekers can use to apply for I-94 forms, the arrival-departure record cards for people who are not U.S. citizens or permanent residents.
According to Koen Leurs, professor of gender, media and migration studies at Utrecht University in the Netherlands, we have arrived at a point where migration management has become so reliant on digital technology that the former can no longer be studied in isolation from the latter. Investigating this reliance for his new book, Digital Migration, Leurs came to the conclusion that applications like those mentioned above are more often than not a double-edged sword, presenting both benefits and drawbacks.
There has been “a huge acceleration” in the way digital technologies “dehumanize people,” says Koen Leurs, professor of gender, media and migration studies at Utrecht University in the Netherlands. Governments treat asylum seekers as test subjects for new inventions, all along the borders of the developed world.
On the one hand, digital technology can make migration management more efficient and less labor intensive, enabling countries to process larger numbers of people in a time when global movement is on the rise due to globalization and political instability. Leurs also discovered that informal knowledge networks such as Informed Immigrant, an online resource that connects migrants to social workers and community organizers, have positively impacted the lives of their users. The same, Leurs notes, is true of platforms like Twitter, Facebook, and WhatsApp, all of which migrants use to stay in touch with each other as well as their families back home. “The emotional support you receive through social media is something we all came to appreciate during the COVID pandemic,” Leurs says. “For refugees, this had already been common knowledge for years.”
On the flipside, automatization of migration management – particularly through the use of AI – has spawned extensive criticism from human rights activists. Sharing their sentiment, Leurs attests that many so-called innovations are making life harder for migrants, not easier. He also says there has been “a huge acceleration” in the way digital technologies “dehumanize people,” and that governments treat asylum seekers as test subjects for new inventions, all along the borders of the developed world.
In Jordan, for example, refugees had to scan their irises in order to collect aid, prompting the question of whether such measures are ethical. Speaking to Reuters, Petra Molnar, a fellow at Harvard University’s Berkman Klein Center for Internet and Society, said that she was troubled by the fact that this experiment was done on marginalized people. “The refugees are guinea pigs,” she said. “Imagine what would happen at your local grocery store if all of a sudden iris scanning became a thing,” she pointed out. “People would be up in arms. But somehow it is OK to do it in a refugee camp.”
Artificial intelligence programs have been scrutinized for their unreliability, their complex processing, thwarted by the race and gender biases picked up from training data. In 2019, a female reporter from The Intercept tested iBorderCtrl and, despite answering all questions truthfully, was accused by the machine of lying four out of 16 times. Had she been waiting at checkpoint on the Greek or Latvian border, she would have been flagged for additional screening – a measure that could jeopardize her chance of entry. Because of its biases, and the negative press that this attracted, iBorderCtrl did not move past its test phase.
While facial recognition caused problems on the European border, it was helpful in Ukraine, where programs like those developed by software company Clearview AI are used to spot Russian spies, identify dead soldiers, and check movement in and out of war zones.
In April 2021, not long after iBorderCtrl was shut down, the European Commission proposed the world’s first-ever legal framework for AI regulation: the Artificial Intelligence Act. The act, which is still being developed, promises to prevent potentially “harmful” AI practices from being used in migration management. In the most recent draft, approved by the European Parliament’s Liberties and Internal Market committees, the ban included emotion recognition systems (like iBorderCtrl), predictive policing systems (like EUMigraTool), and biometric categorization systems (like iris scanners). The act also stipulates that AI must be subject to strict oversight and accountability measures.
While some worry the AI Act is not comprehensive enough, others wonder if it is in fact going too far. Indeed, many proponents of machine learning argue that, by placing a categorical ban on certain systems, governments will thwart the development of potentially useful technology. While facial recognition caused problems on the European border, it was helpful in Ukraine, where programs like those developed by software company Clearview AI are used to spot Russian spies, identify dead soldiers, and check movement in and out of war zones.
Instead of flat-out banning AI, why not strive to make it more reliable? “One of the most compelling arguments against AI is that it is inherently biased,” says Vera Raposo, an assistant professor of law at NOVA University in Lisbon specializing in digital law. “In truth, AI itself is not biased; it becomes biased due to human influence. It seems that complete eradication of biases is unattainable, but mitigation is possible. We can strive to reduce biases by employing more comprehensive and unbiased data in AI training and encompassing a wider range of individuals. We can also work on developing less biased algorithms, although this is challenging given that coders, being human, inherently possess biases of their own.”
AI is most effective when it enhances human performance rather than replacing it.
Accessibility is another obstacle that needs to be overcome. Leurs points out that, in migration management, AI often functions as a “black box” because the migration officers operating it are unable to comprehend its complex decision-making process and thus unable to scrutinize its results. One solution to this problem is to have law enforcement work closely with AI experts. Alternatively, machine learning could be limited to gathering and summarizing information, leaving evaluation of that information to actual people.
Raposo agrees AI is most effective when it enhances human performance rather than replacing it. On the topic of transparency, she does note that making an AI that is both sophisticated and easy to understand is a little bit like having your cake and eating it too. “In numerous domains,” she explains, “we might need to accept a reduced level of explainability in exchange for a high degree of accuracy (assuming we cannot have both).” Using healthcare as an analogy, she adds that “some medications work in ways not fully understood by either doctors or pharma companies, yet persist due to demonstrated efficacy in clinical trials.”
Leurs believes digital technologies used in migration management can be improved through a push for more conscientious research. “Technology is a poison and a medicine for that poison,” he argues, which is why new tech should be developed with its potential applications in mind. “Ethics has become a major concern in recent years. Increasingly, and particularly in the study of forced migration, researchers are posing critical questions like ‘what happens with the data that is gathered?’ and ‘who will this harm?’” In some cases, Leurs thinks, that last question may need to be reversed: we should be thinking about how we can actively disarm oppressive structures. “After all, our work should align with the interests of the communities it is going to affect.”
Meet Dr. Renee Wegrzyn, the first Director of President Biden's new health agency, ARPA-H
Today's podcast guest, Dr. Renee Wegrzyn, directs ARPA-H, a new agency formed last year to spearhead health innovations. Time will tell if ARPA-H will produce advances on the level of its fellow agency, DARPA.
In today’s podcast episode, I talk with Renee Wegrzyn, appointed by President Biden as the first director of a health agency created last year, the Advanced Research Projects Agency for Health, or ARPA-H. It’s inspired by DARPA, the agency that develops innovations for the Defense department and has been credited with hatching world-changing technologies such as ARPANET, which became the internet.
Time will tell if ARPA-H will lead to similar achievements in the realm of health. That’s what President Biden and Congress expect in return for funding ARPA-H at 2.5 billion dollars over three years.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
How will the agency figure out which projects to take on, especially with so many patient advocates for different diseases demanding moonshot funding for rapid progress?
I talked with Dr. Wegrzyn about the opportunities and challenges, what lessons ARPA-H is borrowing from Operation Warp Speed, how she decided on the first ARPA-H project that was announced recently, why a separate agency was needed instead of reforming HHS and the National Institutes of Health to be better at innovation, and how ARPA-H will make progress on disease prevention in addition to treatments for cancer, Alzheimer’s and diabetes, among many other health priorities.
Dr. Wegrzyn’s resume leaves no doubt of her suitability for this role. She was a program manager at DARPA where she focused on applying gene editing and synthetic biology to the goal of improving biosecurity. For her work there, she received the Superior Public Service Medal and, in case that wasn’t enough ARPA experience, she also worked at another ARPA that leads advanced projects in intelligence, called I-ARPA. Before that, she ran technical teams in the private sector working on gene therapies and disease diagnostics, among other areas. She has been a vice president of business development at Gingko Bioworks and headed innovation at Concentric by Gingko. Her training and education includes a PhD and undergraduate degree in applied biology from the Georgia Institute of Technology and she did her postdoc as an Alexander von Humboldt Fellow in Heidelberg, Germany.
Dr. Wegrzyn told me that she’s “in the hot seat.” The pressure is on for ARPA-H especially after the need and potential for health innovation was spot lit by the pandemic and the unprecedented speed of vaccine development. We'll soon find out if ARPA-H can produce gamechangers in health that are equivalent to DARPA’s creation of the internet.
Show links:
ARPA-H - https://arpa-h.gov/
Dr. Wegrzyn profile - https://arpa-h.gov/people/renee-wegrzyn/
Dr. Wegrzyn Twitter - https://twitter.com/rwegrzyn?lang=en
President Biden Announces Dr. Wegrzyn's appointment - https://www.whitehouse.gov/briefing-room/statement...
Leaps.org coverage of ARPA-H - https://leaps.org/arpa/
ARPA-H program for joints to heal themselves - https://arpa-h.gov/news/nitro/ -
ARPA-H virtual talent search - https://arpa-h.gov/news/aco-talent-search/

Dr. Renee Wegrzyn was appointed director of ARPA-H last October.
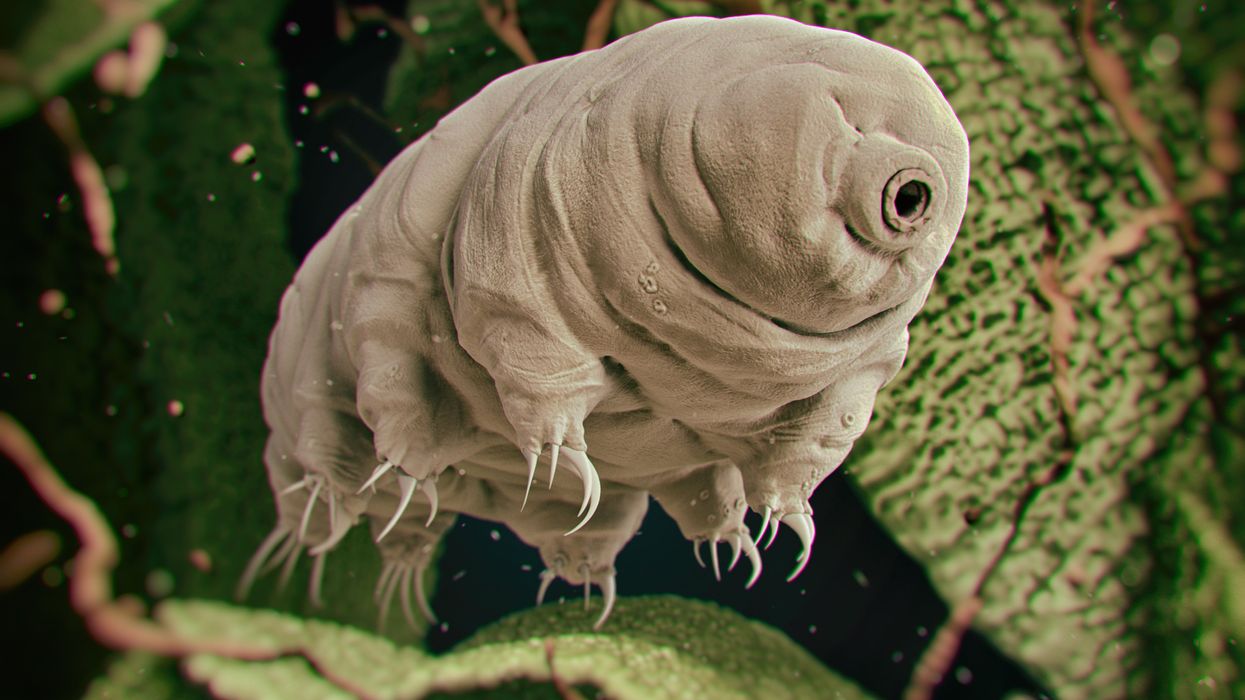
Tiny, tough “water bears” may help bring new vaccines and medicines to sub-Saharan Africa
Tardigrades can completely dehydrate and later rehydrate themselves, a survival trick that scientists are harnessing to preserve medicines in hot temperatures.
Microscopic tardigrades, widely considered to be some of the toughest animals on earth, can survive for decades without oxygen or water and are thought to have lived through a crash-landing on the moon. Also known as water bears, they survive by fully dehydrating and later rehydrating themselves – a feat only a few animals can accomplish. Now scientists are harnessing tardigrades’ talents to make medicines that can be dried and stored at ambient temperatures and later rehydrated for use—instead of being kept refrigerated or frozen.
Many biologics—pharmaceutical products made by using living cells or synthesized from biological sources—require refrigeration, which isn’t always available in many remote locales or places with unreliable electricity. These products include mRNA and other vaccines, monoclonal antibodies and immuno-therapies for cancer, rheumatoid arthritis and other conditions. Cooling is also needed for medicines for blood clotting disorders like hemophilia and for trauma patients.
Formulating biologics to withstand drying and hot temperatures has been the holy grail for pharmaceutical researchers for decades. It’s a hard feat to manage. “Biologic pharmaceuticals are highly efficacious, but many are inherently unstable,” says Thomas Boothby, assistant professor of molecular biology at University of Wyoming. Therefore, during storage and shipping, they must be refrigerated at 2 to 8 degrees Celsius (35 to 46 degrees Fahrenheit). Some must be frozen, typically at -20 degrees Celsius, but sometimes as low -90 degrees Celsius as was the case with the Pfizer Covid vaccine.
For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
The costly cold chain
The logistics network that ensures those temperature requirements are met from production to administration is called the cold chain. This cold chain network is often unreliable or entirely lacking in remote, rural areas in developing nations that have malfunctioning electrical grids. “Almost all routine vaccines require a cold chain,” says Christopher Fox, senior vice president of formulations at the Access to Advanced Health Institute. But when the power goes out, so does refrigeration, putting refrigerated or frozen medical products at risk. Consequently, the mRNA vaccines developed for Covid-19 and other conditions, as well as more traditional vaccines for cholera, tetanus and other diseases, often can’t be delivered to the most remote parts of the world.
To understand the scope of the challenge, consider this: In the U.S., more than 984 million doses of Covid-19 vaccine have been distributed so far. Each one needed refrigeration that, even in the U.S., proved challenging. Now extrapolate to all vaccines and the entire world. For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
Globally, the cold chain packaging market is valued at over $15 billion and is expected to exceed $60 billion by 2033.
Adobe Stock
Freeze-drying, also called lyophilization, which is common for many vaccines, isn’t always an option. Many freeze-dried vaccines still need refrigeration, and even medicines approved for storage at ambient temperatures break down in the heat of sub-Saharan Africa. “Even in a freeze-dried state, biologics often will undergo partial rehydration and dehydration, which can be extremely damaging,” Boothby explains.
The cold chain is also very expensive to maintain. The global pharmaceutical cold chain packaging market is valued at more than $15 billion, and is expected to exceed $60 billion by 2033, according to a report by Future Market Insights. This cost is only expected to grow. According to the consulting company Accenture, the number of medicines that require the cold chain are expected to grow by 48 percent, compared to only 21 percent for non-cold-chain therapies.
Tardigrades to the rescue
Tardigrades are only about a millimeter long – with four legs and claws, and they lumber around like bears, thus their nickname – but could provide a big solution. “Tardigrades are unique in the animal kingdom, in that they’re able to survive a vast array of environmental insults,” says Boothby, the Wyoming professor. “They can be dried out, frozen, heated past the boiling point of water and irradiated at levels that are thousands of times more than you or I could survive.” So, his team is gradually unlocking tardigrades’ survival secrets and applying them to biologic pharmaceuticals to make them withstand both extreme heat and desiccation without losing efficacy.
Boothby’s team is focusing on blood clotting factor VIII, which, as the name implies, causes blood to clot. Currently, Boothby is concentrating on the so-called cytoplasmic abundant heat soluble (CAHS) protein family, which is found only in tardigrades, protecting them when they dry out. “We showed we can desiccate a biologic (blood clotting factor VIII, a key clotting component) in the presence of tardigrade proteins,” he says—without losing any of its effectiveness.
The researchers mixed the tardigrade protein with the blood clotting factor and then dried and rehydrated that substance six times without damaging the latter. This suggests that biologics protected with tardigrade proteins can withstand real-world fluctuations in humidity.
Furthermore, Boothby’s team found that when the blood clotting factor was dried and stabilized with tardigrade proteins, it retained its efficacy at temperatures as high as 95 degrees Celsius. That’s over 200 degrees Fahrenheit, much hotter than the 58 degrees Celsius that the World Meteorological Organization lists as the hottest recorded air temperature on earth. In contrast, without the protein, the blood clotting factor degraded significantly. The team published their findings in the journal Nature in March.
Although tardigrades rarely live more than 2.5 years, they have survived in a desiccated state for up to two decades, according to Animal Diversity Web. This suggests that tardigrades’ CAHS protein can protect biologic pharmaceuticals nearly indefinitely without refrigeration or freezing, which makes it significantly easier to deliver them in locations where refrigeration is unreliable or doesn’t exist.
The tricks of the tardigrades
Besides the CAHS proteins, tardigrades rely on a type of sugar called trehalose and some other protectants. So, rather than drying up, their cells solidify into rigid, glass-like structures. As that happens, viscosity between cells increases, thereby slowing their biological functions so much that they all but stop.
Now Boothby is combining CAHS D, one of the proteins in the CAHS family, with trehalose. He found that CAHS D and trehalose each protected proteins through repeated drying and rehydrating cycles. They also work synergistically, which means that together they might stabilize biologics under a variety of dry storage conditions.
“We’re finding the protective effect is not just additive but actually is synergistic,” he says. “We’re keen to see if something like that also holds true with different protein combinations.” If so, combinations could possibly protect against a variety of conditions.
Commercialization outlook
Before any stabilization technology for biologics can be commercialized, it first must be approved by the appropriate regulators. In the U.S., that’s the U.S. Food and Drug Administration. Developing a new formulation would require clinical testing and vast numbers of participants. So existing vaccines and biologics likely won’t be re-formulated for dry storage. “Many were developed decades ago,” says Fox. “They‘re not going to be reformulated into thermo-stable vaccines overnight,” if ever, he predicts.
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits.
Instead, this technology is most likely to be used for the new products and formulations that are just being created. New and improved vaccines will be the first to benefit. Good candidates include the plethora of mRNA vaccines, as well as biologic pharmaceuticals for neglected diseases that affect parts of the world where reliable cold chain is difficult to maintain, Boothby says. Some examples include new, more effective vaccines for malaria and for pathogenic Escherichia coli, which causes diarrhea.
Tallying up the benefits
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits. For instance, MenAfriVac, a meningitis vaccine (without tardigrade proteins) developed for sub-Saharan Africa, can be stored at up to 40 degrees Celsius for four days before administration. “If you have a few days where you don’t need to maintain the cold chain, it’s easier to transport vaccines to remote areas,” Fox says, where refrigeration does not exist or is not reliable.
Better health is an obvious benefit. MenAfriVac reduced suspected meningitis cases by 57 percent in the overall population and more than 99 percent among vaccinated individuals.
Lower healthcare costs are another benefit. One study done in Togo found that the cold chain-related costs increased the per dose vaccine price up to 11-fold. The ability to ship the vaccines using the usual cold chain, but transporting them at ambient temperatures for the final few days cut the cost in half.
There are environmental benefits, too, such as reducing fuel consumption and greenhouse gas emissions. Cold chain transports consume 20 percent more fuel than non-cold chain shipping, due to refrigeration equipment, according to the International Trade Administration.
A study by researchers at Johns Hopkins University compared the greenhouse gas emissions of the new, oral Vaxart COVID-19 vaccine (which doesn’t require refrigeration) with four intramuscular vaccines (which require refrigeration or freezing). While the Vaxart vaccine is still in clinical trials, the study found that “up to 82.25 million kilograms of CO2 could be averted by using oral vaccines in the U.S. alone.” That is akin to taking 17,700 vehicles out of service for one year.
Although tardigrades’ protective proteins won’t be a component of biologic pharmaceutics for several years, scientists are proving that this approach is viable. They are hopeful that a day will come when vaccines and biologics can be delivered anywhere in the world without needing refrigerators or freezers en route.