Regenerative medicine has come a long way, baby

After a cloned baby sheep, what started as one of the most controversial areas in medicine is now promising to transform it.
The field of regenerative medicine had a shaky start. In 2002, when news spread about the first cloned animal, Dolly the sheep, a raucous debate ensued. Scary headlines and organized opposition groups put pressure on government leaders, who responded by tightening restrictions on this type of research.
Fast forward to today, and regenerative medicine, which focuses on making unhealthy tissues and organs healthy again, is rewriting the code to healing many disorders, though it’s still young enough to be considered nascent. What started as one of the most controversial areas in medicine is now promising to transform it.
Progress in the lab has addressed previous concerns. Back in the early 2000s, some of the most fervent controversy centered around somatic cell nuclear transfer (SCNT), the process used by scientists to produce Dolly. There was fear that this technique could be used in humans, with possibly adverse effects, considering the many medical problems of the animals who had been cloned.
But today, scientists have discovered better approaches with fewer risks. Pioneers in the field are embracing new possibilities for cellular reprogramming, 3D organ printing, AI collaboration, and even growing organs in space. It could bring a new era of personalized medicine for longer, healthier lives - while potentially sparking new controversies.
Engineering tissues from amniotic fluids
Work in regenerative medicine seeks to reverse damage to organs and tissues by culling, modifying and replacing cells in the human body. Scientists in this field reach deep into the mechanisms of diseases and the breakdowns of cells, the little workhorses that perform all life-giving processes. If cells can’t do their jobs, they take whole organs and systems down with them. Regenerative medicine seeks to harness the power of healthy cells derived from stem cells to do the work that can literally restore patients to a state of health—by giving them healthy, functioning tissues and organs.
Modern-day regenerative medicine takes its origin from the 1998 isolation of human embryonic stem cells, first achieved by John Gearhart at Johns Hopkins University. Gearhart isolated the pluripotent cells that can differentiate into virtually every kind of cell in the human body. There was a raging controversy about the use of these cells in research because at that time they came exclusively from early-stage embryos or fetal tissue.
Back then, the highly controversial SCNT cells were the only way to produce genetically matched stem cells to treat patients. Since then, the picture has changed radically because other sources of highly versatile stem cells have been developed. Today, scientists can derive stem cells from amniotic fluid or reprogram patients’ skin cells back to an immature state, so they can differentiate into whatever types of cells the patient needs.
In the context of medical history, the field of regenerative medicine is progressing at a dizzying speed. But for those living with aggressive or chronic illnesses, it can seem that the wheels of medical progress grind slowly.
The ethical debate has been dialed back and, in the last few decades, the field has produced important innovations, spurring the development of whole new FDA processes and categories, says Anthony Atala, a bioengineer and director of the Wake Forest Institute for Regenerative Medicine. Atala and a large team of researchers have pioneered many of the first applications of 3D printed tissues and organs using cells developed from patients or those obtained from amniotic fluid or placentas.
His lab, considered to be the largest devoted to translational regenerative medicine, is currently working with 40 different engineered human tissues. Sixteen of them have been transplanted into patients. That includes skin, bladders, urethras, muscles, kidneys and vaginal organs, to name just a few.
These achievements are made possible by converging disciplines and technologies, such as cell therapies, bioengineering, gene editing, nanotechnology and 3D printing, to create living tissues and organs for human transplants. Atala is currently overseeing clinical trials to test the safety of tissues and organs engineered in the Wake Forest lab, a significant step toward FDA approval.
In the context of medical history, the field of regenerative medicine is progressing at a dizzying speed. But for those living with aggressive or chronic illnesses, it can seem that the wheels of medical progress grind slowly.
“It’s never fast enough,” Atala says. “We want to get new treatments into the clinic faster, but the reality is that you have to dot all your i’s and cross all your t’s—and rightly so, for the sake of patient safety. People want predictions, but you can never predict how much work it will take to go from conceptualization to utilization.”
As a surgeon, he also treats patients and is able to follow transplant recipients. “At the end of the day, the goal is to get these technologies into patients, and working with the patients is a very rewarding experience,” he says. Will the 3D printed organs ever outrun the shortage of donated organs? “That’s the hope,” Atala says, “but this technology won’t eliminate the need for them in our lifetime.”
New methods are out of this world
Jeanne Loring, another pioneer in the field and director of the Center for Regenerative Medicine at Scripps Research Institute in San Diego, says that investment in regenerative medicine is not only paying off, but is leading to truly personalized medicine, one of the holy grails of modern science.
This is because a patient’s own skin cells can be reprogrammed to become replacements for various malfunctioning cells causing incurable diseases, such as diabetes, heart disease, macular degeneration and Parkinson’s. If the cells are obtained from a source other than the patient, they can be rejected by the immune system. This means that patients need lifelong immunosuppression, which isn’t ideal. “With Covid,” says Loring, “I became acutely aware of the dangers of immunosuppression.” Using the patient’s own cells eliminates that problem.
Microgravity conditions make it easier for the cells to form three-dimensional structures, which could more easily lead to the growing of whole organs. In fact, Loring's own cells have been sent to the ISS for study.
Loring has a special interest in neurons, or brain cells that can be developed by manipulating cells found in the skin. She is looking to eventually treat Parkinson’s disease using them. The manipulated cells produce dopamine, the critical hormone or neurotransmitter lacking in the brains of patients. A company she founded plans to start a Phase I clinical trial using cell therapies for Parkinson’s soon, she says.
This is the culmination of many years of basic research on her part, some of it on her own cells. In 2007, Loring had her own cells reprogrammed, so there’s a cell line that carries her DNA. “They’re just like embryonic stem cells, but personal,” she said.
Loring has another special interest—sending immature cells into space to be studied at the International Space Station. There, microgravity conditions make it easier for the cells to form three-dimensional structures, which could more easily lead to the growing of whole organs. In fact, her own cells have been sent to the ISS for study. “My colleagues and I have completed four missions at the space station,” she says. “The last cells came down last August. They were my own cells reprogrammed into pluripotent cells in 2009. No one else can say that,” she adds.
Future controversies and tipping points
Although the original SCNT debate has calmed down, more controversies may arise, Loring thinks.
One of them could concern growing synthetic embryos. The embryos are ultimately derived from embryonic stem cells, and it’s not clear to what stage these embryos can or will be grown in an artificial uterus—another recent invention. The science, so far done only in animals, is still new and has not been widely publicized but, eventually, “People will notice the production of synthetic embryos and growing them in an artificial uterus,” Loring says. It’s likely to incite many of the same reactions as the use of embryonic stem cells.
Bernard Siegel, the founder and director of the Regenerative Medicine Foundation and executive director of the newly formed Healthspan Action Coalition (HSAC), believes that stem cell science is rapidly approaching tipping point and changing all of medical science. (For disclosure, I do consulting work for HSAC). Siegel says that regenerative medicine has become a new pillar of medicine that has recently been fast-tracked by new technology.
Artificial intelligence is speeding up discoveries and the convergence of key disciplines, as demonstrated in Atala’s lab, which is creating complex new medical products that replace the body’s natural parts. Just as importantly, those parts are genetically matched and pose no risk of rejection.
These new technologies must be regulated, which can be a challenge, Siegel notes. “Cell therapies represent a challenge to the existing regulatory structure, including payment, reimbursement and infrastructure issues that 20 years ago, didn’t exist.” Now the FDA and other agencies are faced with this revolution, and they’re just beginning to adapt.
Siegel cited the 2021 FDA Modernization Act as a major step. The Act allows drug developers to use alternatives to animal testing in investigating the safety and efficacy of new compounds, loosening the agency’s requirement for extensive animal testing before a new drug can move into clinical trials. The Act is a recognition of the profound effect that cultured human cells are having on research. Being able to test drugs using actual human cells promises to be far safer and more accurate in predicting how they will act in the human body, and could accelerate drug development.
Siegel, a longtime veteran and founding father of several health advocacy organizations, believes this work helped bring cell therapies to people sooner rather than later. His new focus, through the HSAC, is to leverage regenerative medicine into extending not just the lifespan but the worldwide human healthspan, the period of life lived with health and vigor. “When you look at the HSAC as a tree,” asks Siegel, “what are the roots of that tree? Stem cell science and the huge ecosystem it has created.” The study of human aging is another root to the tree that has potential to lengthen healthspans.
The revolutionary science underlying the extension of the healthspan needs to be available to the whole world, Siegel says. “We need to take all these roots and come up with a way to improve the life of all mankind,” he says. “Everyone should be able to take advantage of this promising new world.”
Stefania Sterling with her son Charlie, who was diagnosed at age 3 with autism.
Stefania Sterling was just 21 when she had her son, Charlie. She was young and healthy, with no genetic issues apparent in either her or her husband's family, so she expected Charlie to be typical.
"It is surprising that the prevalence of a significant disorder like autism has risen so consistently over a relatively brief period."
It wasn't until she went to a Mommy and Me music class when he was one, and she saw all the other one-year-olds walking, that she realized how different her son was. He could barely crawl, didn't speak, and made no eye contact. By the time he was three, he was diagnosed as being on the lower functioning end of the autism spectrum.
She isn't sure why it happened – and researchers, too, are still trying to understand the basis of the complex condition. Studies suggest that genes can act together with influences from the environment to affect development in ways that lead to Autism Spectrum Disorder (ASD). But rates of ASD are rising dramatically, making the need to figure out why it's happening all the more urgent.
The Latest News
Indeed, the CDC's latest autism report, released last week, which uses 2016 data, found that the prevalence of ASD in four-year-old children was one in 64 children, or 15.6 affected children per 1,000. That's more than the 14.1 rate they found in 2014, for the 11 states included in the study. New Jersey, as in years past, was the highest, with 25.3 per 1,000, compared to Missouri, which had just 8.8 per 1,000.
The rate for eight-year-olds had risen as well. Researchers found the ASD prevalence nationwide was 18.5 per 1,000, or one in 54, about 10 percent higher than the 16.8 rate found in 2014. New Jersey, again, was the highest, at one in 32 kids, compared to Colorado, which had the lowest rate, at one in 76 kids. For New Jersey, that's a 175 percent rise from the baseline number taken in 2000, when the state had just one in 101 kids.
"It is surprising that the prevalence of a significant disorder like autism has risen so consistently over a relatively brief period," said Walter Zahorodny, an associate professor of pediatrics at Rutgers New Jersey Medical School, who was involved in collecting the data.
The study echoed the findings of a surprising 2011 study in South Korea that found 1 in every 38 students had ASD. That was the the first comprehensive study of autism prevalence using a total population sample: A team of investigators from the U.S., South Korea, and Canada looked at 55,000 children ages 7 to 12 living in a community in South Korea and found that 2.64 percent of them had some level of autism.
Searching for Answers
Scientists can't put their finger on why rates are rising. Some say it's better diagnosis. That is, it's not that more people have autism. It's that we're better at detecting it. Others attribute it to changes in the diagnostic criteria. Specifically, the May 2013 update of the Diagnostic and Statistical Manual of Mental Disorders-5 -- the standard classification of mental disorders -- removed the communication deficit from the autism definition, which made more children fall under that category. Cynical observers believe physicians and therapists are handing out the diagnosis more freely to allow access to services available only to children with autism, but that are also effective for other children.
Alycia Halladay, chief science officer for the Autism Science Foundation in New York, said she wishes there were just one answer, but there's not. While she believes the rising ASD numbers are due in part to factors like better diagnosis and a change in the definition, she does not believe that accounts for the entire rise in prevalence. As for the high numbers in New Jersey, she said the state has always had a higher prevalence of autism compared to other states. It is also one of the few states that does a good job at recording cases of autism in its educational records, meaning that children in New Jersey are more likely to be counted compared to kids in other states.
"Not every state is as good as New Jersey," she said. "That accounts for some of the difference compared to elsewhere, but we don't know if it's all of the difference in prevalence, or most of it, or what."
"What we do know is that vaccinations do not cause autism."
There is simply no defined proven reason for these increases, said Scott Badesch, outgoing president and CEO of the Autism Society of America.
"There are suggestions that it is based on better diagnosis, but there are also suggestions that the incidence of autism is in fact increasing due to reasons that have yet been determined," he said, adding, "What we do know is that vaccinations do not cause autism."
Zahorodny, the pediatrics professor, believes something is going on beyond better detection or evolving definitions.
"Changes in awareness and shifts in how children are identified or diagnosed are relevant, but they only take you so far in accounting for an increase of this magnitude," he said. "We don't know what is driving the surge in autism recorded by the ADDM Network and others."
He suggested that the increase in prevalence could be due to non-genetic environmental triggers or risk factors we do not yet know about, citing possibilities including parental age, prematurity, low birth rate, multiplicity, breech presentation, or C-section delivery. It may not be one, but rather several factors combined, he said.
"Increases in ASD prevalence have affected the whole population, so the triggers or risks must be very widely dispersed across all strata," he added.
There are studies that find new risk factors for ASD almost on a daily basis, said Idan Menashe, assistant professor in the Department of Health at Ben-Gurion University of the Negev, the fastest growing research university in Israel.
"There are plenty of studies that find new genetic variants (and new genes)," he said. In addition, various prenatal and perinatal risk factors are associated with a risk of ASD. He cited a study his university conducted last year on the relationship between C-section births and ASD, which found that exposure to general anesthesia may explain the association.
Whatever the cause, health practitioners are seeing the consequences in real time.
"People say rates are higher because of the changes in the diagnostic criteria," said Dr. Roseann Capanna-Hodge, a psychologist in Ridgefield, CT. "And they say it's easier for children to get identified. I say that's not the truth and that I've been doing this for 30 years, and that even 10 years ago, I did not see the level of autism that I do see today."
Sure, we're better at detecting autism, she added, but the detection improvements have largely occurred at the low- to mid- level part of the spectrum. The higher rates of autism are occurring at the more severe end, in her experience.
A Polarizing Theory
Among the more controversial risk factors scientists are exploring is the role environmental toxins may play in the development of autism. Some scientists, doctors and mental health experts suspect that toxins like heavy metals, pesticides, chemicals, or pollution may interrupt the way genes are expressed or the way endocrine systems function, manifesting in symptoms of autism. But others firmly resist such claims, at least until more evidence comes forth. To date, studies have been mixed and many have been more associative than causative.
"Today, scientists are still trying to figure out whether there are other environmental changes that can explain this rise, but studies of this question didn't provide any conclusive answer," said Menashe, who also serves as the scientific director of the National Autism Research Center at BGU.
"It's not everything that makes Charlie. He's just like any other kid."
That inconclusiveness has not dissuaded some doctors from taking the perspective that toxins do play a role. "Autism rates are rising because there is a mismatch between our genes and our environment," said Julia Getzelman, a pediatrician in San Francisco. "The majority of our evolution didn't include the kinds of toxic hits we are experiencing. The planet has changed drastically in just the last 75 years –- it has become more and more polluted with tens of thousands of unregulated chemicals being used by industry that are having effects on our most vulnerable."
She cites BPA, an industrial chemical that has been used since the 1960s to make certain plastics and resins. A large body of research, she says, has shown its impact on human health and the endocrine system. BPA binds to our own hormone receptors, so it may negatively impact the thyroid and brain. A study in 2015 was the first to identify a link between BPA and some children with autism, but the relationship was associative, not causative. Meanwhile, the Food and Drug Administration maintains that BPA is safe at the current levels occurring in food, based on its ongoing review of the available scientific evidence.
Michael Mooney, President of St. Louis-based Delta Genesis, a non-profit organization that treats children struggling with neurodevelopmental delays like autism, suspects a strong role for epigenetics, which refers to changes in how genes are expressed as a result of environmental influences, lifestyle behaviors, age, or disease states.
He believes some children are genetically predisposed to the disorder, and some unknown influence or combination of influences pushes them over the edge, triggering epigenetic changes that result in symptoms of autism.
For Stefania Sterling, it doesn't really matter how or why she had an autistic child. That's only one part of Charlie.
"It's not everything that makes Charlie," she said. "He's just like any other kid. He comes with happy moments. He comes with sad moments. Just like my other three kids."
How Excessive Regulation Helped Ignite COVID-19's Rampant Spread
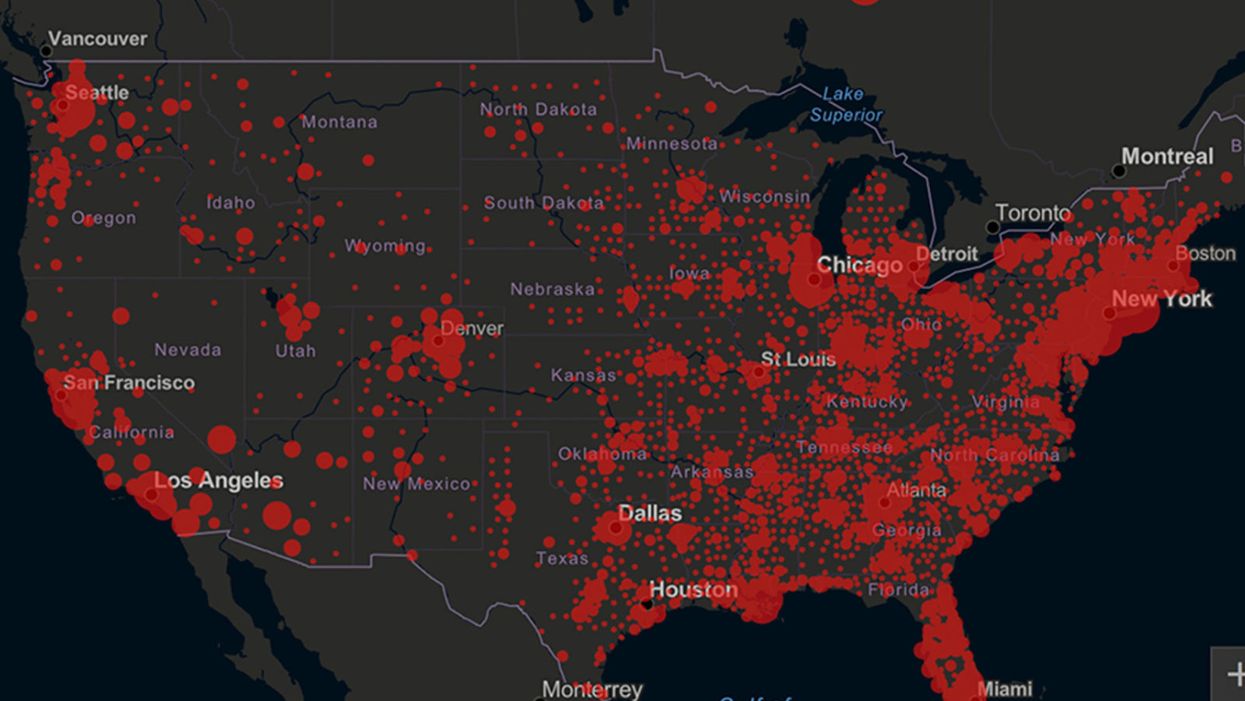
Screenshot of an interactive map of coronavirus cases across the United States, current as of 1:45 p.m. Pacific time on Tuesday, March 31st. Full map accessible at https://coronavirus.jhu.edu/map.html
When historians of the future look back at the 2020 pandemic, the heroic work of Helen Y. Chu, a flu researcher at the University of Washington, will be worthy of recognition.
Chu's team bravely defied the order and conducted the testing anyway.
In late January, Chu was testing nasal swabs for the Seattle Flu Study to monitor influenza spread when she learned of the first case of COVID-19 in Washington state. She deemed it a pressing public health matter to document if and how the illness was spreading locally, so that early containment efforts could succeed. So she sought regulatory approval to adapt the Flu Study to test for the coronavirus, but the federal government denied the request because the original project was funded to study only influenza.
Aware of the urgency, Chu's team bravely defied the order and conducted the testing anyway. Soon they identified a local case in a teenager without any travel history, followed by others. Still, the government tried to shutter their efforts until the outbreak grew dangerous enough to command attention.
Needless testing delays, prompted by excessive regulatory interference, eliminated any chances of curbing the pandemic at its initial stages. Even after Chu went out on a limb to sound alarms, a heavy-handed bureaucracy crushed the nation's ability to roll out early and widespread testing across the country. The Centers for Disease Control and Prevention infamously blundered its own test, while also impeding state and private labs from coming on board, fueling a massive shortage.
The long holdup created "a backlog of testing that needed to be done," says Amesh Adalja, an infectious disease specialist who is a senior scholar at the Johns Hopkins University Center for Health Security.
In a public health crisis, "the ideal situation" would allow the government's test to be "supplanted by private laboratories" without such "a lag in that transition," Adalja says. Only after the eventual release of CDC's test could private industry "begin in earnest" to develop its own versions under the Food and Drug Administration's emergency use authorization.
In a statement, CDC acknowledged that "this process has not gone as smoothly as we would have liked, but there is currently no backlog for testing at CDC."
Now, universities and corporations are in a race against time, playing catch up as the virus continues its relentless spread, also afflicting many health care workers on the front lines.
"Home-testing accessibility is key to preventing further spread of the COVID-19 pandemic."
Hospitals are attempting to add the novel coronavirus to the testing panel of their existent diagnostic machines, which would reduce the results processing time from 48 hours to as little as four hours. Meanwhile, at least four companies announced plans to deliver at-home collection tests to help meet the demand – before a startling injunction by the FDA halted their plans.
Everlywell, an Austin, Texas-based digital health company, had been set to launch online sales of at-home collection kits directly to consumers last week. Scaling up in a matter of days to an initial supply of 30,000 tests, Everlywell collaborated with multiple laboratories where consumers could ship their nasal swab samples overnight, projecting capacity to screen a quarter-million individuals on a weekly basis, says Frank Ong, chief medical and scientific officer.
Secure digital results would have been available online within 48 hours of a sample's arrival at the lab, as well as a telehealth consultation with an independent, board-certified doctor if someone tested positive, for an inclusive $135 cost. The test has a less than 3 percent false-negative rate, Ong says, and in the event of an inadequate self-swab, the lab would not report a conclusive finding. "Home-testing accessibility," he says, "is key to preventing further spread of the COVID-19 pandemic."
But on March 20, the FDA announced restrictions on home collection tests due to concerns about accuracy. The agency did note "the public health value in expanding the availability of COVID-19 testing through safe and accurate tests that may include home collection," while adding that "we are actively working with test developers in this space."
After the restrictions were announced, Everlywell decided to allocate its initial supply of COVID-19 collection kits to hospitals, clinics, nursing homes, and other qualifying health care companies that can commit to no-cost screening of frontline workers and high-risk symptomatic patients. For now, no consumers can order a home-collection test.
"Losing two months is close to disastrous, and that's what we did."
Currently, the U.S. has ramped up to testing an estimated 100,000 people a day, according to Stat News. But 150,000 or more Americans should be tested every day, says Ashish Jha, professor and director of the Harvard Global Health Institute. Due to the dearth of tests, many sick people who suspect they are infected still cannot get confirmation unless they need to be hospitalized.
To give a concrete sense of how far behind we are in testing, consider Palm Beach County, Fla. The state's only drive-thru test center just opened there, requiring an appointment. The center aims to test 750 people per day, but more than 330,000 people have already called to try to book a slot.
"This is such a rapidly moving infection that losing a few days is bad, and losing a couple of weeks is terrible," says Jha, a practicing general internist. "Losing two months is close to disastrous, and that's what we did."
At this point, it will take a long time to fully ramp up. "We are blindfolded," he adds, "and I'd like to take the blindfolds off so we can fight this battle with our eyes wide open."
Better late than never: Yesterday, FDA Commissioner Stephen Hahn said in a statement that the agency has worked with more than 230 test developers and has approved 20 tests since January. An especially notable one was authorized last Friday – 67 days since the country's first known case in Washington state. It's a rapid point-of-care test from medical-device firm Abbott that provides positive results in five minutes and negative results in 13 minutes. Abbott will send 50,000 tests a day to urgent care settings. The first tests are expected to ship tomorrow.