Tiny, tough “water bears” may help bring new vaccines and medicines to sub-Saharan Africa
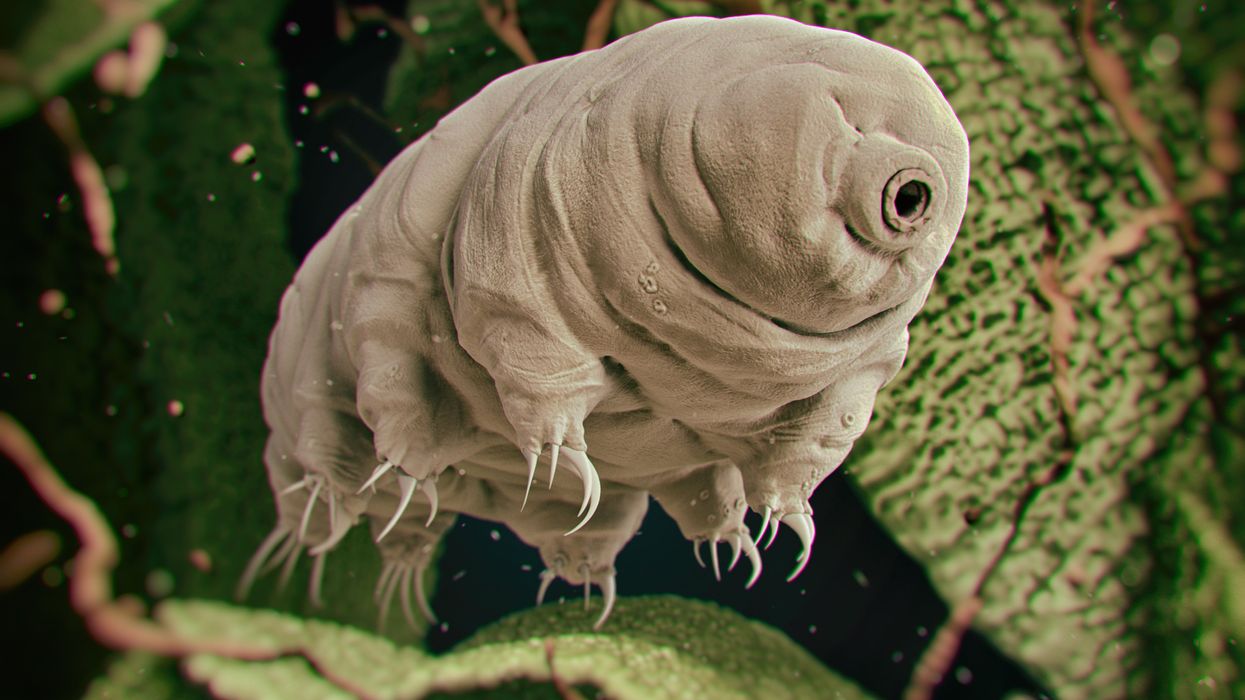
Tardigrades can completely dehydrate and later rehydrate themselves, a survival trick that scientists are harnessing to preserve medicines in hot temperatures.
Microscopic tardigrades, widely considered to be some of the toughest animals on earth, can survive for decades without oxygen or water and are thought to have lived through a crash-landing on the moon. Also known as water bears, they survive by fully dehydrating and later rehydrating themselves – a feat only a few animals can accomplish. Now scientists are harnessing tardigrades’ talents to make medicines that can be dried and stored at ambient temperatures and later rehydrated for use—instead of being kept refrigerated or frozen.
Many biologics—pharmaceutical products made by using living cells or synthesized from biological sources—require refrigeration, which isn’t always available in many remote locales or places with unreliable electricity. These products include mRNA and other vaccines, monoclonal antibodies and immuno-therapies for cancer, rheumatoid arthritis and other conditions. Cooling is also needed for medicines for blood clotting disorders like hemophilia and for trauma patients.
Formulating biologics to withstand drying and hot temperatures has been the holy grail for pharmaceutical researchers for decades. It’s a hard feat to manage. “Biologic pharmaceuticals are highly efficacious, but many are inherently unstable,” says Thomas Boothby, assistant professor of molecular biology at University of Wyoming. Therefore, during storage and shipping, they must be refrigerated at 2 to 8 degrees Celsius (35 to 46 degrees Fahrenheit). Some must be frozen, typically at -20 degrees Celsius, but sometimes as low -90 degrees Celsius as was the case with the Pfizer Covid vaccine.
For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
The costly cold chain
The logistics network that ensures those temperature requirements are met from production to administration is called the cold chain. This cold chain network is often unreliable or entirely lacking in remote, rural areas in developing nations that have malfunctioning electrical grids. “Almost all routine vaccines require a cold chain,” says Christopher Fox, senior vice president of formulations at the Access to Advanced Health Institute. But when the power goes out, so does refrigeration, putting refrigerated or frozen medical products at risk. Consequently, the mRNA vaccines developed for Covid-19 and other conditions, as well as more traditional vaccines for cholera, tetanus and other diseases, often can’t be delivered to the most remote parts of the world.
To understand the scope of the challenge, consider this: In the U.S., more than 984 million doses of Covid-19 vaccine have been distributed so far. Each one needed refrigeration that, even in the U.S., proved challenging. Now extrapolate to all vaccines and the entire world. For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
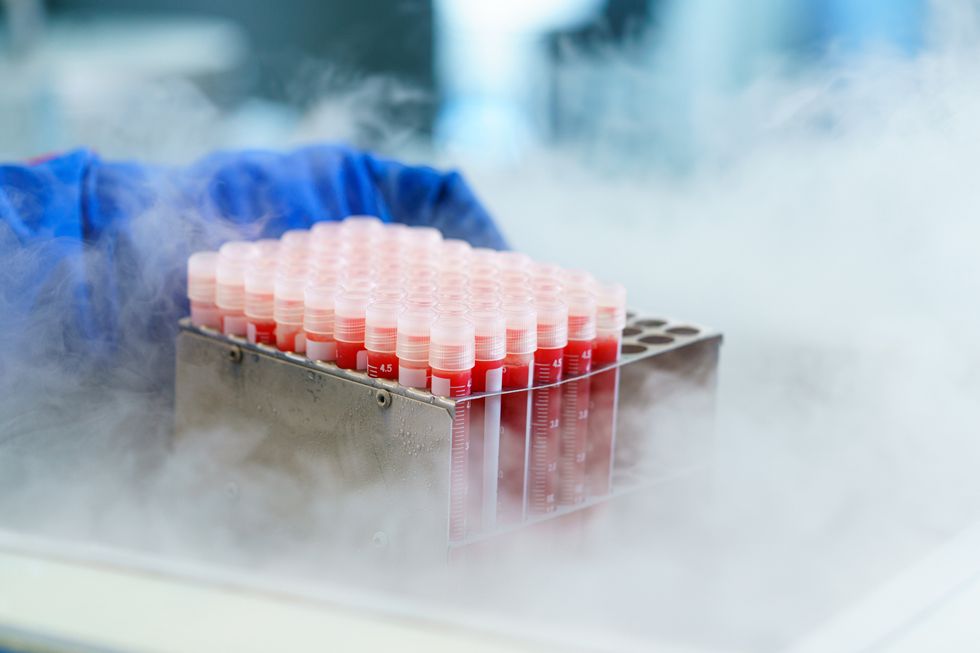
Globally, the cold chain packaging market is valued at over $15 billion and is expected to exceed $60 billion by 2033.
Adobe Stock
Freeze-drying, also called lyophilization, which is common for many vaccines, isn’t always an option. Many freeze-dried vaccines still need refrigeration, and even medicines approved for storage at ambient temperatures break down in the heat of sub-Saharan Africa. “Even in a freeze-dried state, biologics often will undergo partial rehydration and dehydration, which can be extremely damaging,” Boothby explains.
The cold chain is also very expensive to maintain. The global pharmaceutical cold chain packaging market is valued at more than $15 billion, and is expected to exceed $60 billion by 2033, according to a report by Future Market Insights. This cost is only expected to grow. According to the consulting company Accenture, the number of medicines that require the cold chain are expected to grow by 48 percent, compared to only 21 percent for non-cold-chain therapies.
Tardigrades to the rescue
Tardigrades are only about a millimeter long – with four legs and claws, and they lumber around like bears, thus their nickname – but could provide a big solution. “Tardigrades are unique in the animal kingdom, in that they’re able to survive a vast array of environmental insults,” says Boothby, the Wyoming professor. “They can be dried out, frozen, heated past the boiling point of water and irradiated at levels that are thousands of times more than you or I could survive.” So, his team is gradually unlocking tardigrades’ survival secrets and applying them to biologic pharmaceuticals to make them withstand both extreme heat and desiccation without losing efficacy.
Boothby’s team is focusing on blood clotting factor VIII, which, as the name implies, causes blood to clot. Currently, Boothby is concentrating on the so-called cytoplasmic abundant heat soluble (CAHS) protein family, which is found only in tardigrades, protecting them when they dry out. “We showed we can desiccate a biologic (blood clotting factor VIII, a key clotting component) in the presence of tardigrade proteins,” he says—without losing any of its effectiveness.
The researchers mixed the tardigrade protein with the blood clotting factor and then dried and rehydrated that substance six times without damaging the latter. This suggests that biologics protected with tardigrade proteins can withstand real-world fluctuations in humidity.
Furthermore, Boothby’s team found that when the blood clotting factor was dried and stabilized with tardigrade proteins, it retained its efficacy at temperatures as high as 95 degrees Celsius. That’s over 200 degrees Fahrenheit, much hotter than the 58 degrees Celsius that the World Meteorological Organization lists as the hottest recorded air temperature on earth. In contrast, without the protein, the blood clotting factor degraded significantly. The team published their findings in the journal Nature in March.
Although tardigrades rarely live more than 2.5 years, they have survived in a desiccated state for up to two decades, according to Animal Diversity Web. This suggests that tardigrades’ CAHS protein can protect biologic pharmaceuticals nearly indefinitely without refrigeration or freezing, which makes it significantly easier to deliver them in locations where refrigeration is unreliable or doesn’t exist.
The tricks of the tardigrades
Besides the CAHS proteins, tardigrades rely on a type of sugar called trehalose and some other protectants. So, rather than drying up, their cells solidify into rigid, glass-like structures. As that happens, viscosity between cells increases, thereby slowing their biological functions so much that they all but stop.
Now Boothby is combining CAHS D, one of the proteins in the CAHS family, with trehalose. He found that CAHS D and trehalose each protected proteins through repeated drying and rehydrating cycles. They also work synergistically, which means that together they might stabilize biologics under a variety of dry storage conditions.
“We’re finding the protective effect is not just additive but actually is synergistic,” he says. “We’re keen to see if something like that also holds true with different protein combinations.” If so, combinations could possibly protect against a variety of conditions.
Commercialization outlook
Before any stabilization technology for biologics can be commercialized, it first must be approved by the appropriate regulators. In the U.S., that’s the U.S. Food and Drug Administration. Developing a new formulation would require clinical testing and vast numbers of participants. So existing vaccines and biologics likely won’t be re-formulated for dry storage. “Many were developed decades ago,” says Fox. “They‘re not going to be reformulated into thermo-stable vaccines overnight,” if ever, he predicts.
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits.
Instead, this technology is most likely to be used for the new products and formulations that are just being created. New and improved vaccines will be the first to benefit. Good candidates include the plethora of mRNA vaccines, as well as biologic pharmaceuticals for neglected diseases that affect parts of the world where reliable cold chain is difficult to maintain, Boothby says. Some examples include new, more effective vaccines for malaria and for pathogenic Escherichia coli, which causes diarrhea.
Tallying up the benefits
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits. For instance, MenAfriVac, a meningitis vaccine (without tardigrade proteins) developed for sub-Saharan Africa, can be stored at up to 40 degrees Celsius for four days before administration. “If you have a few days where you don’t need to maintain the cold chain, it’s easier to transport vaccines to remote areas,” Fox says, where refrigeration does not exist or is not reliable.
Better health is an obvious benefit. MenAfriVac reduced suspected meningitis cases by 57 percent in the overall population and more than 99 percent among vaccinated individuals.
Lower healthcare costs are another benefit. One study done in Togo found that the cold chain-related costs increased the per dose vaccine price up to 11-fold. The ability to ship the vaccines using the usual cold chain, but transporting them at ambient temperatures for the final few days cut the cost in half.
There are environmental benefits, too, such as reducing fuel consumption and greenhouse gas emissions. Cold chain transports consume 20 percent more fuel than non-cold chain shipping, due to refrigeration equipment, according to the International Trade Administration.
A study by researchers at Johns Hopkins University compared the greenhouse gas emissions of the new, oral Vaxart COVID-19 vaccine (which doesn’t require refrigeration) with four intramuscular vaccines (which require refrigeration or freezing). While the Vaxart vaccine is still in clinical trials, the study found that “up to 82.25 million kilograms of CO2 could be averted by using oral vaccines in the U.S. alone.” That is akin to taking 17,700 vehicles out of service for one year.
Although tardigrades’ protective proteins won’t be a component of biologic pharmaceutics for several years, scientists are proving that this approach is viable. They are hopeful that a day will come when vaccines and biologics can be delivered anywhere in the world without needing refrigerators or freezers en route.
On May 13th, scientific and medical experts will discuss and answer questions about the vaccine for those under 16.
This virtual event convened leading scientific and medical experts to address the public's questions and concerns about Covid-19 vaccines in kids and teens. Highlight video below.
DATE:
Thursday, May 13th, 2021
12:30 p.m. - 1:45 p.m. EDT
Dr. H. Dele Davies, M.D., MHCM

Senior Vice Chancellor for Academic Affairs and Dean for Graduate Studies at the University of Nebraska Medical (UNMC). He is an internationally recognized expert in pediatric infectious diseases and a leader in community health.
Dr. Emily Oster, Ph.D.

Professor of Economics at Brown University. She is a best-selling author and parenting guru who has pioneered a method of assessing school safety.
Dr. Tina Q. Tan, M.D.

Professor of Pediatrics at the Feinberg School of Medicine, Northwestern University. She has been involved in several vaccine survey studies that examine the awareness, acceptance, barriers and utilization of recommended preventative vaccines.
Dr. Inci Yildirim, M.D., Ph.D., M.Sc.

Associate Professor of Pediatrics (Infectious Disease); Medical Director, Transplant Infectious Diseases at Yale School of Medicine; Associate Professor of Global Health, Yale Institute for Global Health. She is an investigator for the multi-institutional COVID-19 Prevention Network's (CoVPN) Moderna mRNA-1273 clinical trial for children 6 months to 12 years of age.
About the Event Series
This event is the second of a four-part series co-hosted by Leaps.org, the Aspen Institute Science & Society Program, and the Sabin–Aspen Vaccine Science & Policy Group, with generous support from the Gordon and Betty Moore Foundation and the Howard Hughes Medical Institute.
:
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
Pregnant & Breastfeeding Women Who Get the COVID-19 Vaccine Are Protecting Their Infants, Research Suggests
Becky Cummings, who got vaccinated in December, snuggles her newborn, Clark, while he takes a nap.
Becky Cummings had multiple reasons to get vaccinated against COVID-19 while tending to her firstborn, Clark, who arrived in September 2020 at 27 weeks.
The 29-year-old intensive care unit nurse in Greensboro, North Carolina, had witnessed the devastation day in and day out as the virus took its toll on the young and old. But when she was offered the vaccine, she hesitated, skeptical of its rapid emergency use authorization.
Exclusion of pregnant and lactating mothers from clinical trials fueled her concerns. Ultimately, though, she concluded the benefits of vaccination outweighed the risks of contracting the potentially deadly virus.
"Long story short," Cummings says, in December "I got vaccinated to protect myself, my family, my patients, and the general public."
At the time, Cummings remained on the fence about breastfeeding, citing a lack of evidence to support its safety after vaccination, so she pumped and stashed breast milk in the freezer. Her son is adjusting to life as a preemie, requiring mother's milk to be thickened with formula, but she's becoming comfortable with the idea of breastfeeding as more research suggests it's safe.
"If I could pop him on the boob," she says, "I would do it in a heartbeat."
Now, a study recently published in the Journal of the American Medical Association found "robust secretion" of specific antibodies in the breast milk of mothers who received a COVID-19 vaccine, indicating a potentially protective effect against infection in their infants.
The presence of antibodies in the breast milk, detectable as early as two weeks after vaccination, lasted for six weeks after the second dose of the Pfizer-BioNTech vaccine.
"We believe antibody secretion into breast milk will persist for much longer than six weeks, but we first wanted to prove any secretion at all after vaccination," says Ilan Youngster, the study's corresponding author and head of pediatric infectious diseases at Shamir Medical Center in Zerifin, Israel.
That's why the research team performed a preliminary analysis at six weeks. "We are still collecting samples from participants and hope to soon be able to comment about the duration of secretion."
As with other respiratory illnesses, such as influenza and pertussis, secretion of antibodies in breast milk confers protection from infection in infants. The researchers expect a similar immune response from the COVID-19 vaccine and are expecting the findings to spur an increase in vaccine acceptance among pregnant and lactating women.
A COVID-19 outbreak struck three families the research team followed in the study, resulting in at least one non-breastfed sibling developing symptomatic infection; however, none of the breastfed babies became ill. "This is obviously not empirical proof," Youngster acknowledges, "but still a nice anecdote."
Leaps.org inquired whether infants who derive antibodies only through breast milk are likely to have a lower immunity than infants whose mothers were vaccinated while they were in utero. In other words, is maternal transmission of antibodies stronger during pregnancy than during breastfeeding, or about the same?
"This is a different kind of transmission," Youngster explains. "When a woman is infected or vaccinated during pregnancy, some antibodies will be transferred through the placenta to the baby's bloodstream and be present for several months." But in the nursing mother, that protection occurs through local action. "We always recommend breastfeeding whenever possible, and, in this case, it might have added benefits."
A study published online in March found COVID-19 vaccination provided pregnant and lactating women with robust immune responses comparable to those experienced by their nonpregnant counterparts. The study, appearing in the American Journal of Obstetrics and Gynecology, documented the presence of vaccine-generated antibodies in umbilical cord blood and breast milk after mothers had been vaccinated.
Natali Aziz, a maternal-fetal medicine specialist at Stanford University School of Medicine, notes that it's too early to draw firm conclusions about the reduction in COVID-19 infection rates among newborns of vaccinated mothers. Citing the two aforementioned research studies, she says it's biologically plausible that antibodies passed through the placenta and breast milk impart protective benefits. While thousands of pregnant and lactating women have been vaccinated against COVID-19, without incurring adverse outcomes, many are still wondering whether it's safe to breastfeed afterward.
It's important to bear in mind that pregnant women may develop more severe COVID-19 complications, which could lead to intubation or admittance to the intensive care unit. "We, in our practice, are supporting pregnant and breastfeeding patients to be vaccinated," says Aziz, who is also director of perinatal infectious diseases at Stanford Children's Health, which has been vaccinating new mothers and other hospitalized patients at discharge since late April.
Earlier in April, Huntington Hospital in Long Island, New York, began offering the COVID-19 vaccine to women after they gave birth. The hospital chose the one-shot Johnson & Johnson vaccine for postpartum patients, so they wouldn't need to return for a second shot while acclimating to life with a newborn, says Mitchell Kramer, chairman of obstetrics and gynecology.
The hospital suspended the program when the Food and Drug Administration and the Centers for Disease Control and Prevention paused use of the J&J vaccine starting April 13, while investigating several reports of dangerous blood clots and low platelet counts among more than 7 million people in the United States who had received that vaccine.
In lifting the pause April 23, the agencies announced the vaccine's fact sheets will bear a warning of the heightened risk for a rare but serious blood clot disorder among women under age 50. As a result, Kramer says, "we will likely not be using the J&J vaccine for our postpartum population."
So, would it make sense to vaccinate infants when one for them eventually becomes available, not just their mothers? "In general, most of the time, infants do not have as good of an immune response to vaccines," says Jonathan Temte, associate dean for public health and community engagement at the University of Wisconsin School of Medicine and Public Health in Madison.
"Many of our vaccines are held until children are six months of age. For example, the influenza vaccine starts at age six months, the measles vaccine typically starts one year of age, as do rubella and mumps. Immune response is typically not very good for viral illnesses in young infants under the age of six months."
So far, the FDA has granted emergency use authorization of the Pfizer-BioNTech vaccine for children as young as 16 years old. The agency is considering data from Pfizer to lower that age limit to 12. Studies are also underway in children under age 12. Meanwhile, data from Moderna on 12-to 17-year-olds and from Pfizer on 12- to 15-year-olds have not been made public. (Pfizer announced at the end of March that its vaccine is 100 percent effective in preventing COVID-19 in the latter age group, and FDA authorization for this population is expected soon.)
"There will be step-wise progression to younger children, with infants and toddlers being the last ones tested," says James Campbell, a pediatric infectious diseases physician and head of maternal and child clinical studies at the University of Maryland School of Medicine Center for Vaccine Development.
"Once the data are analyzed for safety, tolerability, optimal dose and regimen, and immune responses," he adds, "they could be authorized and recommended and made available to American children." The data on younger children are not expected until the end of this year, with regulatory authorization possible in early 2022.
For now, Vonnie Cesar, a family nurse practitioner in Smyrna, Georgia, is aiming to persuade expectant and new mothers to get vaccinated. She has observed that patients in metro Atlanta seem more inclined than their rural counterparts.
To quell some of their skepticism and fears, Cesar, who also teaches nursing students, conceived a visual way to demonstrate the novel mechanism behind the COVID-19 vaccine technology. Holding a palm-size physical therapy ball outfitted with clear-colored push pins, she simulates the spiked protein of the coronavirus. Slime slathered at the gaps permeates areas around the spikes—a process similar to how our antibodies build immunity to the virus.
These conversations often lead hesitant patients to discuss vaccination with their husbands or partners. "The majority of people I'm speaking with," she says, "are coming to the conclusion that this is the right thing for me, this is the common good, and they want to make sure that they're here for their children."
CORRECTION: An earlier version of this article mistakenly stated that the COVID-19 vaccines were granted emergency "approval." They have been granted emergency use authorization, not full FDA approval. We regret the error.

