The Real Science Behind “Anti-Aging” Beauty Products

The beauty industry heavily hypes the nascent promise of stem cells for rejuvenation.
The beauty market abounds with high-end creams and serums that claim the use of stem cells to rejuvenate aging skin.
Selling on the internet and at department stores like Nordstrom, these products promise "breakthrough" applications to plump, smooth, and "reverse visible signs of aging," and at least one product offers to create a "regenerative firming serum, moisturizer, and eye cream" from customers' own stem cells – for a whopping $1200.
The beauty industry is heavily hyping glimmers of the nascent field of stem cell therapy.
Steeped in clinical-sounding terms like "proteins and peptides from pluripotent stem cells," the marketing of these products evokes a dramatic restoration of youthfulness based on cutting-edge science. But the beauty industry is heavily hyping glimmers of the nascent field of stem cell therapy. So what is real and what's not? And is there in fact a way to harness the potential of stem cells in the service of beauty?
Plant vs. Human Stem Cells
Stem cells do indeed have tremendous promise for treating a wide range of diseases and conditions. The cells come from early-stage embryos or, more commonly, from umbilical cord blood or our own bodies. Embryonic stem cells are considered the body's "master" cells because they can develop into any of our several hundred cell types. Adult stem cells, on the other hand, reside in mature tissues and organs like the brain, bone marrow, and skin, and their versatility is more limited. As an internal repair system for many tissue types, they replenish sick, injured, and worn-out cells.
Nowadays, with some sophisticated chemical coaxing, adult stem cells can be returned to an embryonic-like blank state, with the ability to become any cell type that the body might need.
Beauty product manufacturers convey in their advertising that the rejuvenating power of these cells could hold the key to the fountain of youth. But there's something the manufacturers don't always tell you: their products do not typically use human stem cells.
"The whole concept of stem cells is intriguing to the public," says Tamara Griffiths, a consultant dermatologist for the British Skin Foundation. "But what these products contain is plant stem cells and, more commonly, chemicals that have been derived from plant stem cells."
The plant stem cells are cultured in the lab with special media to get them to produce signaling proteins and peptides, like cytokines and chemokines. These have been shown to be good for reducing inflammation and promoting healthy cell functioning, even if derived from plants. However, according to Griffiths, there are so many active ingredients in these products that it's hard to say just what role each one of them plays. We do know that their ability to replenish human stem cells is extremely limited, and the effects of plant stem cells on human cells are unproven.
"...any cosmetic that is advertised to be anti-aging due to plant stem cells at this time is about as effective as all the skin creams without stem cells."
Whether products containing plant cell-derived ingredients work better than conventional skin products is unknown because these products are not regulated by the U.S. Food and Drug Administration and may rest on dubious, even more or less nonexistent, research. Cosmetics companies have conducted most of the research and the exact formulas they devise are considered proprietary information. They have no incentive to publish their research findings, and they don't have to meet standards imposed by the FDA unless they start using human cells in their products.
"There are biological limits to what you can do with plant cells in the first place," says Griffiths. "No plant stem cell is going to morph into a human skin cell no matter what magic medium you immerse it in. Nor is a plant cell likely to stimulate the production of human stem cells if applied to the skin."
According to Sarah Baucus, a cell biologist, for any type of stem cell to be of any use whatsoever, the cells must be alive. The processing needed to incorporate living cells into any type of cream or serum would inevitably kill them, rendering them useless. The splashy marketing of these products suggests that results may be drastic, but none of these creams is likely to produce the kind of rejuvenating effect that would be on par with a facelift or several other surgical or dermatological procedures.
"Plant stem cell therapy needs to move in the right direction to implement its inherent potential in skin care," researchers wrote in a 2017 paper in the journal Future Science OA. "This might happen in the next 20 years but any cosmetic that is advertised to be anti-aging due to plant stem cells at this time is about as effective as all the skin creams without stem cells."
From Beauty Counter to Doctor's Clinic
Where do you turn if you still want to harness the power of stem cells to reinvigorate the skin? Is there a legitimate treatment using human cells? The answer is possibly, but for that you have to switch from the Nordstrom cosmetics counter to a clinic with a lab, where plastic surgeons work with specialists who culture and manipulate living cells.
Plastic surgeons are experts in wound healing, a process in which stem cells play a prominent role. Doctors have long used the technique of taking fat from the body and injecting it into hollowed-out or depressed areas of the face to fill in injuries, correct wrinkles, and improve the face's curvature. Lipotransfer, or the harvesting of body fat and injecting it into the face, has been around for many years in traditional plastic surgery clinics. In recent years, some plastic surgeons have started to cull stem cells from fat. One procedure that does just that is called cell-assisted lipotransfer, or CAL.
In CAL, adipose tissue, or fat, is harvested by liposuction, usually from the lower abdomen. Fat contains stem cells that can differentiate into several cell types, including skin, muscle, cartilage, and bone. Fat tissue has an especially stem cell-rich layer. These cells are then mixed with some regular fat, making in effect a very stem cell-rich fat solution, right in the doctor's office. The process of manipulating the fat cells takes about 90 to 110 minutes, and then the solution is ready to be injected into the skin, to fill in the lips, the cheeks, and the nasolabial folds, or the deep folds around the nose and mouth.
Unlike regular fat, which is often injected into the face, some experts claim that the cell-enriched fat has better, longer-lasting results. The tissue graft grows its own blood vessels, an advantage that may lead to a more long-lasting graft – though the research is mixed, with some studies showing they do and other studies showing the complete opposite.
For almost all stem cell products on the market today in the U.S., it is not yet known whether they are safe or effective, despite how they are marketed.
One of the pioneers in CAL, a plastic surgeon in Brazil named Dr. Aris Sterodimas, says that the stem cells secrete growth factors that rejuvenate the skin -- like the plant stem cells that are used in topical creams and serums. Except that these cells are human stem cells and hence have inherently more potential in the human body.
Note that CAL doesn't actually result in large numbers of fresh, new replacement cells, as might be imagined. It's simply fat tissue treated to make it richer in stem cells, to have more of the growth-inducing proteins and peptides delivered to the dermis layer of the skin.
Sterodimas works alongside a tissue engineer to provide CAL in his clinic. He uses it as a way to rebuild soft tissues in people disfigured by accidents or diseases, or who are suffering the after-effects of radiation treatments for cancer.
Plastic surgeons get plenty of these patients. But how widespread is CAL for beauty purposes? Sterodimas says that he regularly performs the procedure for Brazilians, and it's widely available in Europe and Japan. In the U.S., the procedure hasn't taken off because there is no FDA approval for the various methods used by different doctors and clinics. A few major academic centers in the U.S. offer the treatment on a clinical trials basis and there are several trials ongoing.
But there is a downside to all lipotransfers: the transplanted fat will eventually be absorbed by the body. Even the cell-enriched fat has a limited lifespan before reabsorption. That means if you like the cosmetic results of CAL, you'll have to repeat the treatment about every two years to maintain the plumping, firming, and smoothing effects on the skin. The results of CAL are "superior to the results of laser treatments and other plastic surgery interventions, though the effect is not as dramatic as a facelift," says Sterodimas.
Buyer Beware
For almost all stem cell products on the market today in the U.S., it is not yet known whether they are safe or effective, despite how they are marketed. There are around 700 clinics in the U.S. offering stem cell treatments and up to 20,000 people have received these therapies. However, the only FDA-approved stem cell treatments use cells from bone marrow or cord blood to treat cancers of the blood and bone marrow. Safety concerns have prompted the FDA to announce increased oversight of stem cell clinics.
As for CAL, most of the clinical trials so far have been focused on using it for breast reconstruction after mastectomy, and results are mixed. Experts warn that the procedure has yet to be proven safe as well as effective. It's important to remember that this newborn science is in the early stages of research.
One question that has also not been definitively settled is whether the transplanted stem cells may give rise to tumors — a risk that is ever-present any time stem cells are used. More research is required to assess the long-term safety and effectiveness of these treatments.
Given the lack of uniform industry standards, one can easily end up at a clinic that overpromises what it can deliver.
In the journal Plastic Reconstruction Surgery in 2014, Adrian McArdle and a team of Stanford University plastic surgeons examined the common claims of CAL's "stem cell facelifts" being offered by clinics across the world. McArdle and his team write: "…the marketplace is characterized by direct-to-consumer corporate medicine strategies that are characterized by unsubstantiated, and sometimes fraudulent claims, that put our patients at risk." Given the lack of uniform industry standards, one can easily end up at a clinic that overpromises what it can deliver.
But according to McArdle, further research on CAL, including clinical trials, is proceeding apace. It's possible that as more research on the potential of stem cells accrues, many of the technical hurdles will be crossed.
If you decide to try CAL in a research or clinical setting, be forewarned. You will be taking part in a young science, with many unknown questions. However, the next time someone offers to sell you stem cells in a jar, you'll know what you're paying for.
A vaccine for Lyme disease could be coming. But will patients accept it?
Hiking is one of Marci Flory's favorite activities, but her chronic Lyme disease sometimes renders her unable to walk. Biotech companies Pfizer and Valneva are testing a vaccine for Lyme disease in a Phase III clinical trial.
For more than two decades, Marci Flory, a 40-year-old emergency room nurse from Lawrence, Kan., has battled the recurring symptoms of chronic Lyme disease, an illness which she believes began after being bitten by a tick during her teenage years.
Over the years, Flory has been plagued by an array of mysterious ailments, ranging from fatigue to crippling pain in her eyes, joints and neck, and even postural tachycardia syndrome or PoTS, an abnormal increase in heart rate after sitting up or standing. Ten years ago, she began to experience the onset of neurological symptoms which ranged from brain fog to sudden headaches, and strange episodes of leg weakness which would leave her unable to walk.
“Initially doctors thought I had ALS, or less likely, multiple sclerosis,” she says. “But after repeated MRI scans for a year, they concluded I had a rare neurological condition called acute transverse myelitis.”
But Flory was not convinced. After ordering a variety of private blood tests, she discovered she was infected with a range of bacteria in the genus Borrelia that live in the guts of ticks, the infectious agents responsible for Lyme disease.
“It made sense,” she says. “Looking back, I was bitten in high school and misdiagnosed with mononucleosis. This was probably the start, and my immune system kept it under wraps for a while. The Lyme bacteria can burrow into every tissue in the body, go into cyst form and become dormant before reactivating.”
The reason why cases of Lyme disease are increasing is down to changing weather patterns, triggered by climate change, meaning that ticks are now found across a much wider geographic range than ever before.
When these species of bacteria are transmitted to humans, they can attack the nervous system, joints and even internal organs which can lead to serious health complications such as arthritis, meningitis and even heart failure. While Lyme disease can sometimes be successfully treated with antibiotics if spotted early on, not everyone responds to these drugs, and for patients who have developed chronic symptoms, there is no known cure. Flory says she knows of fellow Lyme disease patients who have spent hundreds of thousands of dollars seeking treatments.
Concerningly, statistics show that Lyme and other tick-borne diseases are on the rise. Recently released estimates based on health insurance records suggest that at least 476,000 Americans are diagnosed with Lyme disease every year, and many experts believe the true figure is far higher.
The reason why the numbers are growing is down to changing weather patterns, triggered by climate change, meaning that ticks are now found across a much wider geographic range than ever before. Health insurance data shows that cases of Lyme disease have increased fourfold in rural parts of the U.S. over the last 15 years, and 65 percent in urban regions.
As a result, many scientists who have studied Lyme disease feel that it is paramount to bring some form of protective vaccine to market which can be offered to people living in the most at-risk areas.
“Even the increased awareness for Lyme disease has not stopped the cases,” says Eva Sapi, professor of cellular and molecular biology at the University of New Haven. “Some of these patients are looking for answers for years, running from one doctor to another, so that is obviously a very big cost for our society at so many levels.”
Emerging vaccines – and backlash
But with the rising case numbers, interest has grown among the pharmaceutical industry and research communities. Vienna-based biotech Valneva have partnered with Pfizer to take their vaccine – a seasonal jab which offers protection against the six most common strains of Lyme disease in the northern hemisphere – into a Phase III clinical trial which began in August. Involving 6,000 participants in a number of U.S. states and northern Europe where Lyme disease is endemic, it could lead to a licensed vaccine by 2025, if it proves successful.
“For many years Lyme was considered a small market vaccine,” explains Monica E. Embers, assistant professor of parasitology at Tulane University in New Orleans. “Now we know that this is a much bigger problem, Pfizer has stepped up to invest in preventing this disease and other pharmaceutical companies may as well.”
Despite innovations, patient communities and their representatives remain ambivalent about the idea of a vaccine. Some of this skepticism dates back to the failed LYMErix vaccine which was developed in the late 1990s before being withdrawn from the market.
At the same time, scientists at Yale University are developing a messenger RNA vaccine which aims to train the immune system to respond to tick bites by exposing it to 19 proteins found in tick saliva. Whereas the Valneva vaccine targets the bacteria within ticks, the Yale vaccine attempts to provoke an instant and aggressive immune response at the site of the bite. This causes the tick to fall off and limits the potential for transmitting dangerous infections.
But despite these innovations, patient communities and their representatives remain ambivalent about the idea of a vaccine. Some of this skepticism dates back to the failed LYMErix vaccine which was developed in the late 1990s before being withdrawn from the market in 2002 after concerns were raised that it might induce autoimmune reactions in humans.
While this theory was ultimately disproved, the lingering stigma attached to LYMErix meant that most vaccine manufacturers chose to stay away from the disease for many years, something which Gregory Poland, head of the Mayo Clinic’s Vaccine Research Group in Minnesota, describes as a tragedy.
“Since 2002, we have not had a human Lyme vaccine in the U.S. despite the increasing number of cases,” says Poland. “Pretty much everyone in the field thinks they’re ten times higher than the official numbers, so you’re probably talking at least 400,000 each year. It’s an incredible burden but because of concerns about anti-vax protestors, until very recently, no manufacturer has wanted to touch this.”
Such was the backlash surrounding the failed LYMErix program that scientists have even explored the most creative of workarounds for protecting people in tick-populated regions, without needing to actually vaccinate them. One research program at the University of Tennessee came up with the idea of leaving food pellets containing a vaccine in woodland areas with the idea that rodents would eat the pellets, and the vaccine would then kill Borrelia bacteria within any ticks which subsequently fed on the animals.
Even the Pfizer-Valneva vaccine has been cautiously designed to try and allay any lingering concerns, two decades after LYMErix. “The concept is the same as the original LYMErix vaccine, but it has been made safer by removing regions that had the potential to induce autoimmunity,” says Embers. “There will always be individuals who oppose vaccines, Lyme or otherwise, but it will be a tremendous boost to public health to have the option.”
Vaccine alternatives
Researchers are also considering alternative immunization approaches in case sufficiently large numbers of people choose to reject any Lyme vaccine which gets approved. Researchers at UMass Chan Medical School have developed an artificially generated antibody, administered via an annual injection, which is capable of killing Borrelia bacteria in the guts of ticks before they can get into the human host.
So far animal studies have shown it to be 100 percent effective, while the scientists have completed a Phase I trial in which they tested it for safety on 48 volunteers in Nebraska. Because this approach provides the antibody directly, rather than triggering the human immune system to produce the antibody like a vaccine would, Embers predicts that it could be a viable alternative for the vaccine hesitant as well as providing an option for immunocompromised individuals who cannot produce enough of their own antibodies.
At the same time, many patient groups still raise concerns over the fact that numerous diagnostic tests for Lyme disease have been reported to have a poor accuracy. Without this, they argue that it is difficult to prove whether vaccines or any other form of immunization actually work. “If the disease is not understood enough to create a more accurate test and a universally accepted treatment protocol, particularly for those who weren’t treated promptly, how can we be sure about the efficacy of a vaccine?” says Natasha Metcalf, co-founder of the organization Lyme Disease UK.
Flory points out that there are so many different types of Borrelia bacteria which cause Lyme disease, that the immunizations being developed may only stop a proportion of cases. In addition, she says that chronic Lyme patients often report a whole myriad of co-infections which remain poorly understood and are likely to also be involved in the disease process.
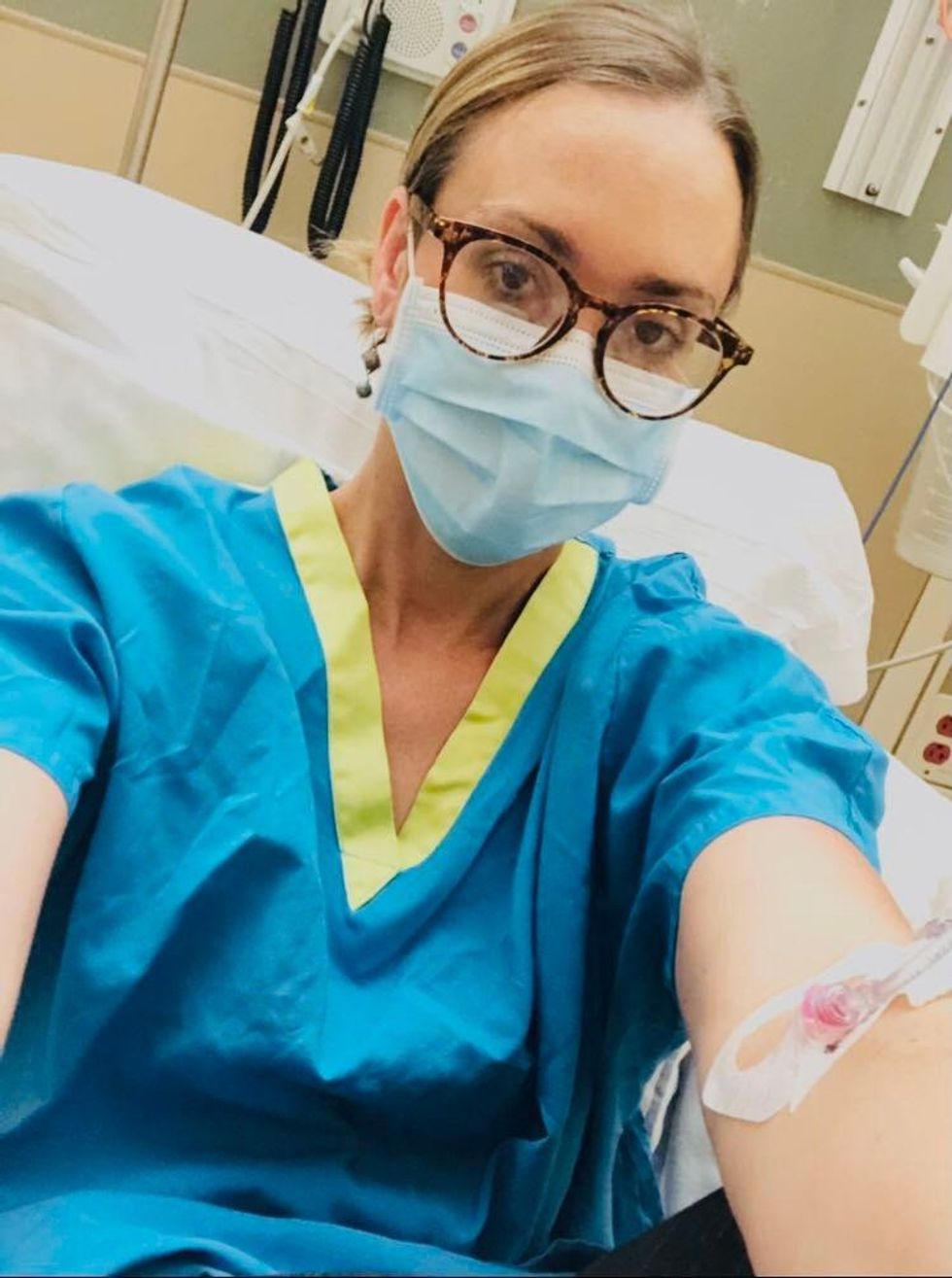
Marci Flory undergoes an infusion in an attempt to treat her Lyme disease symptoms.
Marci Flory
“I would love to see an effective Lyme vaccine but I have my reservations,” she says. “I am infected with four types of Borrelia bacteria, plus many co-infections – Babesia, Bartonella, Erlichiosis, Rickettsia, and Mycoplasma – all from a single Douglas County Kansas tick bite. Lyme never travels alone and the vaccine won’t protect against all the many strains of Borrelia and co-infections.”
Valneva CEO Thomas Lingelbach admits that the Pfizer-Valneva vaccine is not perfect, but predicts that it will still have significant impact if approved.
“We expect the vaccine to have 75 percent plus efficacy,” he says. “There is this legacy around the old Lyme vaccines, but the world is very, very different today. The number of clinical manifestations known to be caused by infection with Lyme Borreliosis has significantly increased, and the understanding around severity has certainly increased.”
Embers agrees that while it will still be important for doctors to monitor for other tick-borne infections which are not necessarily covered by the vaccine, having any clinically approved jab would still represent a major step forward in the fight against the disease.
“I think that any vaccine must be properly vetted, and these companies are performing extensive clinical trials to do just that,” she says. “Lyme is the most common tick-borne disease in the U.S. so the public health impact could be significant. However, clinicians and the general public must remain aware of all of the other tick-borne diseases such as Babesia and Anaplasma, and continue to screen for those when a tick bite is suspected.”
A mosquito under the microscope.
Two years, six million deaths and still counting, scientists are searching for answers to prevent another COVID-19-like tragedy from ever occurring again. And it’s a gargantuan task.
Our disturbed ecosystems are creating more favorable conditions for the spread of infectious disease. Global warming, deforestation, rising sea levels and flooding have contributed to a rise in mosquito-borne infections and longer tick seasons. Disease-carrying animals are in closer range to other species and humans as they migrate to escape the heat. Bats are thought to have carried the SARS-CoV-2 virus to Wuhan, either directly or through another host animal, but thousands of novel viruses are lurking within other wild creatures.
Understanding how climate change contributes to the spread of disease is critical in predicting and thwarting future calamities. But the problem is that predictive models aren’t yet where they need to be for forecasting with certainty beyond the next year, as we could for weather, for instance.
The association between climate and infectious disease is poorly understood, says Irina Tezaur, a computational scientist at Sandia National Laboratories. “Correlations have been observed but it’s not known if these correlations translate to causal relationships.”
To make accurate longer-term predictions, scientists need more empirical data, multiple datasets specific to locations and diseases, and the ability to calculate risks that depend on unpredictable nature and human behavior. Another obstacle is that climate scientists and epidemiologists are not collaborating effectively, so some researchers are calling for a multidisciplinary approach, a new field called Outbreak Science.
Climate scientists are far ahead of epidemiologists in gathering essential data.
Earth System Models—combining the interactions of atmosphere, ocean, land, ice and biosphere—have been in place for two decades to monitor the effects of global climate change. These models must be combined with epidemiological and human model research, areas that are easily skewed by unpredictable elements, from extreme weather events to public environmental policy shifts.
“There is never just one driver in tracking the impact of climate on infectious disease,” says Joacim Rocklöv, a professor at the Heidelberg Institute of Global Health & Heidelberg Interdisciplinary Centre for Scientific Computing in Germany. Rocklöv has studied how climate affects vector-borne diseases—those transmitted to humans by mosquitoes, ticks or fleas. “You need to disentangle the variables to find out how much difference climate makes to the outcome and how much is other factors.” Determinants from deforestation to population density to lack of healthcare access influence the spread of disease.
Even though climate change is not the primary driver of infectious disease today, it poses a major threat to public health in the future, says Rocklöv.
The promise of predictive modeling
“Models are simplifications of a system we’re trying to understand,” says Jeremy Hess, who directs the Center for Health and the Global Environment at University of Washington in Seattle. “They’re tools for learning that improve over time with new observations.”
Accurate predictions depend on high-quality, long-term observational data but models must start with assumptions. “It’s not possible to apply an evidence-based approach for the next 40 years,” says Rocklöv. “Using models to experiment and learn is the only way to figure out what climate means for infectious disease. We collect data and analyze what already happened. What we do today will not make a difference for several decades.”
To improve accuracy, scientists develop and draw on thousands of models to cover as many scenarios as possible. One model may capture the dynamics of disease transmission while another focuses on immunity data or ocean influences or seasonal components of a virus. Further, each model needs to be disease-specific and often location-specific to be useful.
“All models have biases so it’s important to use a suite of models,” Tezaur stresses.
The modeling scientist chooses the drivers of change and parameters based on the question explored. The drivers could be increased precipitation, poverty or mosquito prevalence, for instance. Later, the scientist may need to isolate the effect of one driver so that will require another model.
There have been some related successes, such as the latest models for mosquito-borne diseases like Dengue, Zika and malaria as well as those for flu and tick-borne diseases, says Hess.
Rocklöv was part of a research team that used test data from 2018 and 2019 to identify regions at risk for West Nile virus outbreaks. Using AI, scientists were able to forecast outbreaks of the virus for the entire transmission season in Europe. “In the end, we want data-driven models; that’s what AI can accomplish,” says Rocklöv. Other researchers are making an important headway in creating a framework to predict novel host–parasite interactions.
Modeling studies can run months, years or decades. “The scientist is working with layers of data. The challenge is how to transform and couple different models together on a planetary scale,” says Jeanne Fair, a scientist at Los Alamos National Laboratory, Biosecurity and Public Health, in New Mexico.
Disease forecasting will require a significant investment into the infrastructure needed to collect data about the environment, vectors, and hosts a tall spatial and temporal resolutions.
And it’s a constantly changing picture. A modeling study in an April 2022 issue of Nature predicted that thousands of animals will migrate to cooler locales as temperatures rise. This means that various species will come into closer contact with people and other mammals for the first time. This is likely to increase the risk of emerging infectious disease transmitted from animals to humans, especially in Africa and Asia.
Other things can happen too. Global warming could precipitate viral mutations or new infectious diseases that don’t respond to antimicrobial treatments. Insecticide-resistant mosquitoes could evolve. Weather-related food insecurity could increase malnutrition and weaken people’s immune systems. And the impact of an epidemic will be worse if it co-occurs during a heatwave, flood, or drought, says Hess.
The devil is in the climate variables
Solid predictions about the future of climate and disease are not possible with so many uncertainties. Difficult-to-measure drivers must be added to the empirical model mix, such as land and water use, ecosystem changes or the public’s willingness to accept a vaccine or practice social distancing. Nor is there any precedent for calculating the effect of climate changes that are accelerating at a faster speed than ever before.
The most critical climate variables thought to influence disease spread are temperature, precipitation, humidity, sunshine and wind, according to Tezaur’s research. And then there are variables within variables. Influenza scientists, for example, found that warm winters were predictors of the most severe flu seasons in the following year.
The human factor may be the most challenging determinant. To what degree will people curtail greenhouse gas emissions, if at all? The swift development of effective COVID-19 vaccines was a game-changer, but will scientists be able to repeat it during the next pandemic? Plus, no model could predict the amount of internet-fueled COVID-19 misinformation, Fair noted. To tackle this issue, infectious disease teams are looking to include more sociologists and political scientists in their modeling.
Addressing the gaps
Currently, researchers are focusing on the near future, predicting for next year, says Fair. “When it comes to long-term, that’s where we have the most work to do.” While scientists cannot foresee how political influences and misinformation spread will affect models, they are positioned to make headway in collecting and assessing new data streams that have never been merged.
Disease forecasting will require a significant investment into the infrastructure needed to collect data about the environment, vectors, and hosts at all spatial and temporal resolutions, Fair and her co-authors stated in their recent study. For example real-time data on mosquito prevalence and diversity in various settings and times is limited or non-existent. Fair also would like to see standards set in mosquito data collection in every country. “Standardizing across the US would be a huge accomplishment,” she says.

Understanding how climate change contributes to the spread of disease is critical for thwarting future calamities.
Jeanne Fair
Hess points to a dearth of data in local and regional datasets about how extreme weather events play out in different geographic locations. His research indicates that Africa and the Middle East experienced substantial climate shifts, for example, but are unrepresented in the evidentiary database, which limits conclusions. “A model for dengue may be good in Singapore but not necessarily in Port-au-Prince,” Hess explains. And, he adds, scientists need a way of evaluating models for how effective they are.
The hope, Rocklöv says, is that in the future we will have data-driven models rather than theoretical ones. In turn, sharper statistical analyses can inform resource allocation and intervention strategies to prevent outbreaks.
Most of all, experts emphasize that epidemiologists and climate scientists must stop working in silos. If scientists can successfully merge epidemiological data with climatic, biological, environmental, ecological and demographic data, they will make better predictions about complex disease patterns. Modeling “cross talk” and among disciplines and, in some cases, refusal to release data between countries is hindering discovery and advances.
It’s time for bold transdisciplinary action, says Hess. He points to initiatives that need funding in disease surveillance and control; developing and testing interventions; community education and social mobilization; decision-support analytics to predict when and where infections will emerge; advanced methodologies to improve modeling; training scientists in data management and integrated surveillance.
Establishing a new field of Outbreak Science to coordinate collaboration would accelerate progress. Investment in decision-support modeling tools for public health teams, policy makers, and other long-term planning stakeholders is imperative, too. We need to invest in programs that encourage people from climate modeling and epidemiology to work together in a cohesive fashion, says Tezaur. Joining forces is the only way to solve the formidable challenges ahead.
This article originally appeared in One Health/One Planet, a single-issue magazine that explores how climate change and other environmental shifts are increasing vulnerabilities to infectious diseases by land and by sea. The magazine probes how scientists are making progress with leaders in other fields toward solutions that embrace diverse perspectives and the interconnectedness of all lifeforms and the planet.

