The Sickest Babies Are Covered in Wires. New Tech Is Changing That.
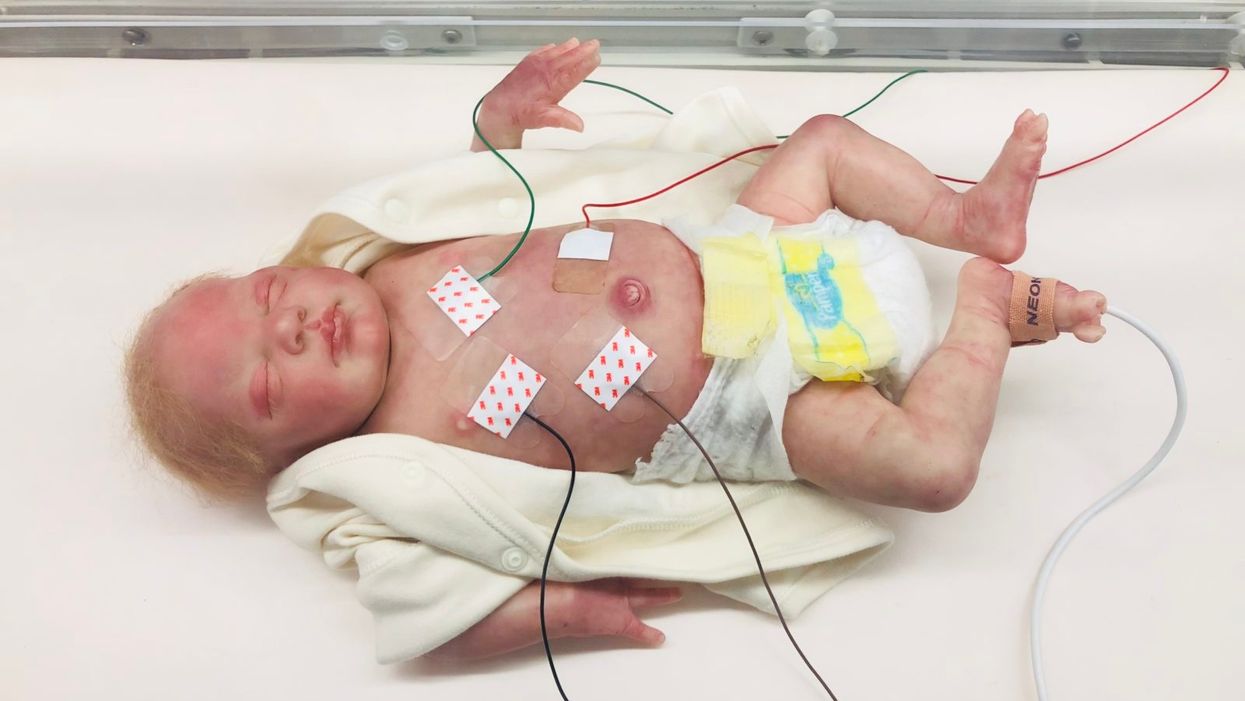
A wired baby in a neonatal intensive care unit.
I'll never forget the experience of having a child in the neonatal intensive care unit (NICU).
Now more than ever, we're working to remove the barriers between new parents and their infants.
It was another layer of uncertainty that filtered into my experience of being a first-time parent. There was so much I didn't know, and the wires attached to my son's small body for the first week of his life were a reminder of that.
I wanted to be the best mother possible. I deeply desired to bring my son home to start our lives. More than anything, I longed for a wireless baby whom I could hold and love freely without limitations.
The wires suggested my baby was fragile and it left me feeling severely unprepared, anxious, and depressed.
In recent years, research has documented the ways that NICU experiences take a toll on parents' mental health. But thankfully, medical technology is rapidly being developed to help reduce the emotional fallout of the NICU. Now more than ever, we're working to remove the barriers between new parents and their infants. The latest example is the first ever wireless monitoring system that was recently developed by a team at Northwestern University.
After listening to the needs of parents and medical staff, Debra Weese-Mayer, M.D., a professor of pediatric autonomic medicine at Feinberg School of Medicine, along with a team of materials scientists, engineers, dermatologists and pediatricians, set out to develop this potentially life-changing technology. Weese-Mayer believes wireless monitoring will have a significant impact for people on all sides of the NICU experience.
"With elimination of the cumbersome wires," she says, "the parents will find their infant more approachable/less intimidating and have improved access to their long-awaited but delivered-too-early infant, allowing them to begin skin-to-skin contact and holding with reduced concern for dislodging wires."
So how does the new system work?
Very thin "skin like" patches made of silicon rubber are placed on the surface of the skin to monitor vitals like heart rate, respiration rate, and body temperature. One patch is placed on the chest or back and the other is placed on the foot.
These patches are safer on the skin than previously used adhesives, reducing the cuts and infections associated with past methods. Finally, an antenna continuously delivers power, often from under the mattress.
The data collected from the patches stream from the body to a tablet or computer.

New wireless sensor technology is being studied to replace wired monitoring in NICUs in the coming years.
(Northwestern University)
Weese-Mayer hopes that wireless systems will be standard soon, but first they must undergo more thorough testing. "I would hope that in the next five years, wireless monitoring will be the standard in NICUs, but there are many essential validation steps before this technology will be embraced nationally," she says.
Until the new systems are ready, parents will be left struggling with the obstacles that wired monitoring presents.
Physical intimacy, for example, appears to have pain-reducing qualities -- something that is particularly important for babies who are battling serious illness. But wires make those cuddles more challenging.
There's also been minimal discussion about how wired monitoring can be particularly limiting for parents with disabilities and mobility aids, or even C-sections.
"When he was first born and I was recovering from my c-section, I couldn't deal with keeping the wires untangled while trying to sit down without hurting myself," says Rhiannon Giles, a writer from North Carolina, who delivered her son at just over 31 weeks after suffering from severe preeclampsia.
"The wires were awful," she remembers. "They fell off constantly when I shifted positions or he kicked a leg, which meant the monitors would alarm. It felt like an intrusion into the quiet little world I was trying to mentally create for us."
Over the last few years, researchers have begun to dive deeper into the literal and metaphorical challenges of wired monitoring.
For many parents, the wires prompt anxiety that worsens an already tense and vulnerable time.
I'll never forget the first time I got to hold my son without wires. It was the first time that motherhood felt manageable.
"Seeing my five-pound-babies covered in wires from head to toe rendered me completely overwhelmed," recalls Caila Smith, a mom of five from Indiana, whose NICU experience began when her twins were born pre-term. "The nurses seemed to handle them perfectly, but I was scared to touch them while they appeared so medically frail."
During the nine days it took for both twins to come home, the limited access she had to her babies started to impact her mental health. "If we would've had wireless sensors and monitors, it would've given us a much greater sense of freedom and confidence when snuggling our newborns," Smith says.
Besides enabling more natural interactions, wireless monitoring would make basic caregiving tasks much easier, like putting on a onesie.
"One thing I noticed is that many preemie outfits are made with zippers," points out Giles, "which just don't work well when your baby has wires coming off of them, head to toe."
Wired systems can pose issues for medical staff as well as parents.
"The main concern regarding wired systems is that they restrict access to the baby and often get tangled with other equipment, like IV lines," says Lamia Soghier, Medical Director of the Neonatal Intensive Care Unit at Children's National in Washington, D.C , who was also a NICU parent herself. "The nurses have to untangle the wires, which takes time, before handing the baby to the family."
I'll never forget the first time I got to hold my son without wires. It was the first time that motherhood felt manageable, and I couldn't stop myself from crying. Suddenly, anything felt possible and all the limitations from that first week of life seemed to fade away. The rise of wired-free monitoring will make some of the stressors that accompany NICU stays a thing of the past.
“Young Blood” Transfusions Are Not Ready For Primetime – Yet
A young woman donates blood.
The world of dementia research erupted into cheers when news of the first real victory in a clinical trial against Alzheimer's Disease in over a decade was revealed last October.
By connecting the circulatory systems of a young and an old mouse, the regenerative potential of the young mouse decreased, and the old mouse became healthier.
Alzheimer's treatments have been famously difficult to develop; 99 percent of the 200-plus such clinical trials since 2000 have utterly failed. Even the few slight successes have failed to produce what is called 'disease modifying' agents that really help people with the disease. This makes the success, by the midsize Spanish pharma company Grifols, worthy of special attention.
However, the specifics of the Grifols treatment, a process called plasmapheresis, are atypical for another reason - they did not give patients a small molecule or an elaborate gene therapy, but rather simply the most common component of normal human blood plasma, a protein called albumin. A large portion of the patients' normal plasma was removed, and then a sterile solution of albumin was infused back into them to keep their overall blood volume relatively constant.
So why does replacing Alzheimer's patients' plasma with albumin seem to help their brains? One theory is that the action is direct. Alzheimer's patients have low levels of serum albumin, which is needed to clear out the plaques of amyloid that slowly build up in the brain. Supplementing those patients with extra albumin boosts their ability to clear the plaques and improves brain health. However, there is also evidence suggesting that the problem may be something present in the plasma of the sick person and pulling their plasma out and replacing it with a filler, like an albumin solution, may be what creates the purported benefit.
This scientific question is the tip of an iceberg that goes far beyond Alzheimer's Disease and albumin, to a debate that has been waged on the pages of scientific journals about the secrets of using young, healthy blood to extend youth and health.
This debate started long before the Grifols data was released, in 2014 when a group of researchers at Stanford found that by connecting the circulatory systems of a young and an old mouse, the regenerative potential of the young mouse decreased, and the old mouse became healthier. There was something either present in young blood that allowed tissues to regenerate, or something present in old blood that prevented regeneration. Whatever the biological reason, the effects in the experiment were extraordinary, providing a startling boost in health in the older mouse.
After the initial findings, multiple research groups got to work trying to identify the "active factor" of regeneration (or the inhibitor of that regeneration). They soon uncovered a variety of compounds such as insulin-like growth factor 1 (IGF1), CCL11, and GDF11, but none seemed to provide all the answers researchers were hoping for, with a number of high-profile retractions based on unsound experimental practices, or inconclusive data.
Years of research later, the simplest conclusion is that the story of plasma regeneration is not simple - there isn't a switch in our blood we can flip to turn back our biological clocks. That said, these hypotheses are far from dead, and many researchers continue to explore the possibility of using the rejuvenating ability of youthful plasma to treat a variety of diseases of aging.
But the bold claims of improved vigor thanks to young blood are so far unsupported by clinical evidence.
The data remain intriguing because of the astounding results from the conjoined circulatory system experiments. The current surge in interest in studying the biology of aging is likely to produce a new crop of interesting results in the next few years. Both CCL11 and GDF11 are being researched as potential drug targets by two startups, Alkahest and Elevian, respectively.
Without clarity on a single active factor driving rejuvenation, it's tempting to try a simpler approach: taking actual blood plasma provided by young people and infusing it into elderly subjects. This is what at least one startup company, Ambrosia, is now offering in five commercial clinics across the U.S. -- for $8,000 a liter.
By using whole plasma, the idea is to sidestep our ignorance, reaping the benefits of young plasma transfusion without knowing exactly what the active factors are that make the treatment work in mice. This space has attracted both established players in the plasmapheresis field – Alkahest and Grifols have teamed up to test fractions of whole plasma in Alzheimer's and Parkinson's – but also direct-to-consumer operations like Ambrosia that just want to offer patients access to treatments without regulatory oversight.
But the bold claims of improved vigor thanks to young blood are so far unsupported by clinical evidence. We simply haven't performed trials to test whether dosing a mostly healthy person with plasma can slow down aging, at least not yet. There is some evidence that plasma replacement works in mice, yes, but those experiments are all done in very different systems than what a human receiving young plasma might experience. To date, I have not seen any plasma transfusion clinic doing young blood plasmapheresis propose a clinical trial that is anything more than a shallow advertisement for their procedures.
The efforts I have seen to perform prophylactic plasmapheresis will fail to impact societal health. Without clearly defined endpoints and proper clinical trials, we won't know whether the procedure really lowers the risk of disease or helps with conditions of aging. So even if their hypothesis is correct, the lack of strong evidence to fall back on means that the procedure will never spread beyond the fringe groups willing to take the risk. If their hypothesis is wrong, then people are paying a huge amount of money for false hope, just as they do, sadly, at the phony stem cell clinics that started popping up all through the 2000s when stem cell hype was at its peak.
Until then, prophylactic plasma transfusions will be the domain of the optimistic and the gullible.
The real progress in the field will be made slowly, using carefully defined products either directly isolated from blood or targeting a bloodborne factor, just as the serious pharma and biotech players are doing already.
The field will progress in stages, first creating and carefully testing treatments for well-defined diseases, and only then will it progress to large-scale clinical trials in relatively healthy people to look for the prevention of disease. Most of us will choose to wait for this second stage of trials before undergoing any new treatments. Until then, prophylactic plasma transfusions will be the domain of the optimistic and the gullible.
Who’s Responsible for Curbing the Teen Vaping Epidemic?
A teenage girl vaping in a park.
E-cigarettes are big business. In 2017, American consumers bought more than $250 million in vapes and juice-filled pods, and spent $1 billion in 2018. By 2023, the global market could be worth $44 billion a year.
"My nine-year-old actually knows what Juuling is. In many cases the [school] bathroom is now referred to as 'the Juuling room.'"
Investors are trying to capitalize on the phenomenal growth. In July 2018, Juul Labs, the company that owns 70 percent of the U.S. e-cigarette market share, raised $1.25 billion at a $16 billion valuation, then sold a 35 percent stake to Phillip Morris USA owner Altria Group in December. The second transaction valued the company at $38 billion. While the traditional tobacco market remains much larger, it's projected to grow at less than two percent a year, making the attractiveness of the rapidly expanding e-cigarette market obvious.
While Juul and other e-cigarette manufacturers argue that their products help adults quit smoking – and there's some research to back this narrative up – much of the growth has been driven by children and teenagers. One CDC study showed a 48 percent rise in e-cigarette use by middle schoolers and a 78 percent increase by high schoolers between 2017 and 2018, a jump from 1.5 million kids to 3.6 million. In response to the study, F.D.A. Commissioner Scott Gottlieb said, "We see clear signs that youth use of electronic cigarettes has reached an epidemic proportion."
Another study found that teenagers between 15 and 17 were 16 times more likely to use Juul than people aged 25-34. In December, Surgeon General Jerome Adams said, "My nine-year-old actually knows what Juuling is. In many cases the [school] bathroom is now referred to as 'the Juuling room.'"
And the product is seriously addictive. A single Juul pod contains as much nicotine as a pack of 20 regular cigarettes. Considering that 90 percent of smokers are addicted by 18 years old, it's clear that steps need to be taken to combat the growing epidemic.
But who should take the lead? Juul and other e-cigarette companies? The F.D.A. and other government regulators? Schools? Parents?
The Surgeon General's website has a list of earnest possible texts that parents can send to their teens to dissuade them from Juuling, like: "Hope none of your friends use e-cigarettes around you. Even breathing the cloud they exhale can expose you to nicotine and chemicals that can be dangerous to your health." While parents can attempt to police their teens, many experts believe that the primary push should come at a federal level.
The regulation battle has already begun. In September, the F.D.A. announced that Juul had 60 days to show a plan that would prevent youth from getting their hands on the product. The result was for the company to announce that it wouldn't sell flavored pods in retail stores except for tobacco, menthol, and mint; Juul also shuttered its Instagram and Facebook accounts. These regulations mirrored an F.D.A. mandate two days later that required flavored e-cigarettes to be sold in closed-off areas. "This policy will make sure the fruity flavors are no longer accessible to kids in retail sites, plan and simple," Commissioner Gottlieb said when announcing the moves. "That's where they're getting access to the e-cigs and we intend to end those sales."
"There isn't a great history of the tobacco industry acting responsibly and being able to in any way police itself."
While so far, Gottlieb – who drew concerns about conflict of interest due to his past position as a board member at e-cigarette company, Kure – has pleased anti-smoking advocates with his efforts, some observers also argue that it needs to go further. "Overall, we didn't know what to expect when a new commissioner came in, but it's been quite refreshing how much attention has been paid to the tobacco industry by the F.D.A.," Robin Koval, CEO and president of Truth Initiative, said a day after the F.D.A. announced the proposed regulations. "It's important to have a start. I certainly want to give credit for that. But we were really hoping and feel that what was announced...doesn't go far enough."
The issue is the industry's inability or unwillingness to police itself in the past. Juul, however, claims that it's now proactively working to prevent young people from taking up its product. "Juul Labs and F.D.A. share a common goal – preventing youth from initiating on nicotine," a company representative said in an email. "To paraphrase Commissioner Gottlieb, we want to be the off-ramp for adult smokers to switch from cigarettes, not an on-ramp for America's youth to initiate on nicotine. We won't be successful in our mission to serve adult smokers if we don't narrow the on-ramp... Our intent was never to have youth use Juul products. But intent is not enough, the numbers are what matter, and the numbers tell us underage use of e-cigarette products is a problem. We must solve it."
Juul argues that its products help adults quit – even offering a calculator on the website showing how much people will save – and that it didn't target youth. But studies show otherwise. Furthermore, the youth smoking prevention curriculum the company released was poorly received. "It's what Philip Morris did years ago," said Bonnie Halpern-Felsher, a professor of pediatrics at Stanford who helped author a study on the program's faults. "They aren't talking about their named product. They are talking about vapes or e-cigarettes. Youth don't consider Juuls to be vapes or e-cigarettes. [Teens] don't talk about flavors. They don't talk about marketing. They did it to look good. But if you look at what [Juul] put together, it's a pretty awful curriculum that was put together pretty quickly."
The American Lung Association gave the FDA an "F" for failing to take mint and menthol e-cigs off the market, since those flavors remain popular with teens.
Add this all up, and in the end, it's hard to see the industry being able to police itself, critics say. Neither the past examples of other tobacco companies nor the present self-imposed regulations indicate that this will succeed.
"There isn't a great history of the tobacco industry acting responsibly and being able to in any way police itself," Koval said. "That job is best left to the F.D.A., and to the states and localities in what they can regulate and legislate to protect young people."
Halpern-Felsher agreed. "I think we need independent bodies. I really don't think that a voluntary ban or a regulation on the part of the industry is a good idea, nor do I think it will work," she said. "It's pretty much the same story, of repeating itself."
Just last week, the American Association of Pediatrics issued a new policy statement calling for the F.D.A. to immediately ban the sale of e-cigarettes to anyone under age 21 and to prohibit the online sale of vaping products and solutions, among other measures. And in its annual report, the American Lung Association gave the F.D.A. an "F" for failing to take mint and menthol e-cigs off the market, since those flavors remain popular with teens.
Few, if any people involved, want more regulation from the federal government. In an ideal world, this wouldn't be necessary. But many experts agree that it is. Anything else is just blowing smoke.