Why you should (virtually) care

Virtual-first care, or V1C, could increase the quality of healthcare and make it more patient-centric by letting patients combine in-person visits with virtual options such as video for seeing their care providers.
As the pandemic turns endemic, healthcare providers have been eagerly urging patients to return to their offices to enjoy the benefits of in-person care.
But wait.
The last two years have forced all sorts of organizations to be nimble, adaptable and creative in how they work, and this includes healthcare providers’ efforts to maintain continuity of care under the most challenging of conditions. So before we go back to “business as usual,” don’t we owe it to those providers and ourselves to admit that business as usual did not work for most of the people the industry exists to help? If we’re going to embrace yet another period of change – periods that don’t happen often in our complex industry – shouldn’t we first stop and ask ourselves what we’re trying to achieve?
Certainly, COVID has shown that telehealth can be an invaluable tool, particularly for patients in rural and underserved communities that lack access to specialty care. It’s also become clear that many – though not all – healthcare encounters can be effectively conducted from afar. That said, the telehealth tactics that filled the gap during the pandemic were largely stitched together substitutes for existing visit-based workflows: with offices closed, patients scheduled video visits for help managing the side effects of their blood pressure medications or to see their endocrinologist for a quarterly check-in. Anyone whose children slogged through the last year or two of remote learning can tell you that simply virtualizing existing processes doesn’t necessarily improve the experience or the outcomes!
But what if our approach to post-pandemic healthcare came from a patient-driven perspective? We have a fleeting opportunity to advance a care model centered on convenient and equitable access that first prioritizes good outcomes, then selects approaches to care – and locations – tailored to each patient. Using the example of education, imagine how effective it would be if each student, regardless of their school district and aptitude, received such individualized attention.
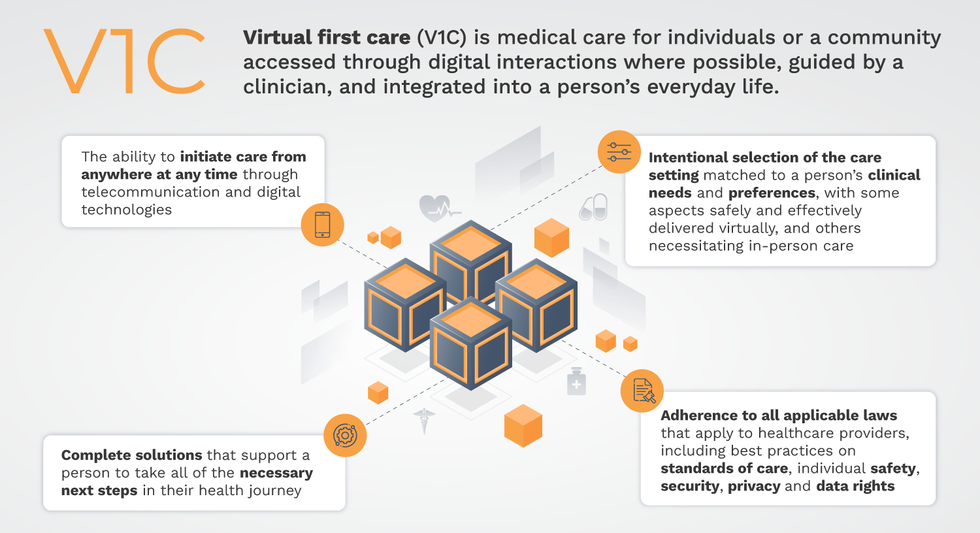
That’s the idea behind virtual-first care (V1C), a new care model centered on convenient, customized, high-quality care that integrates a full suite of services tailored directly to patients’ clinical needs and preferences. This package includes asynchronous communication such as texting; video and other live virtual modes; and in-person options.
V1C goes beyond what you might think of as standard “telehealth” by using evidence-based protocols and tools that include traditional and digital therapeutics and testing, personalized care plans, dynamic patient monitoring, and team-based approaches to care. This could include spit kits mailed for laboratory tests and complementing clinical care with health coaching. V1C also replaces some in-person exams with ongoing monitoring, using sensors for more ‘whole person’ care.
Amidst all this momentum, we have the opportunity to rethink the goals of healthcare innovation, but that means bringing together key stakeholders to demonstrate that sustainable V1C can redefine healthcare.
Established V1C healthcare providers such as Omada, Headspace, and Heartbeat Health, as well as emerging market entrants like Oshi, Visana, and Wellinks, work with a variety of patients who have complicated long-term conditions such as diabetes, heart failure, gastrointestinal illness, endometriosis, and COPD. V1C is comprehensive in ways that are lacking in digital health and its other predecessors: it has the potential to integrate multiple data streams, incorporate more frequent touches and check-ins over time, and manage a much wider range of chronic health conditions, improving lives and reducing disease burden now and in the future.
Recognizing the pandemic-driven interest in virtual care, significant energy and resources are already flowing fast toward V1C. Some of the world’s largest innovators jumped into V1C early on: Verily, Alphabet’s Life Sciences Company, launched Onduo in 2016 to disrupt the diabetes healthcare market, and is now well positioned to scale its solutions. Major insurers like Aetna and United now offer virtual-first plans to members, responding as organizations expand virtual options for employees. Amidst all this momentum, we have the opportunity to rethink the goals of healthcare innovation, but that means bringing together key stakeholders to demonstrate that sustainable V1C can redefine healthcare.
That was the immediate impetus for IMPACT, a consortium of V1C companies, investors, payers and patients founded last year to ensure access to high-quality, evidence-based V1C. Developed by our team at the Digital Medicine Society (DiMe) in collaboration with the American Telemedicine Association (ATA), IMPACT has begun to explore key issues that include giving patients more integrated experiences when accessing both virtual and brick-and-mortar care.
Digital Medicine Society
V1C is not, nor should it be, virtual-only care. In this new era of hybrid healthcare, success will be defined by how well providers help patients navigate the transitions. How do we smoothly hand a patient off from an onsite primary care physician to, say, a virtual cardiologist? How do we get information from a brick-and-mortar to a digital portal? How do you manage dataflow while still staying HIPAA compliant? There are many complex regulatory implications for these new models, as well as an evolving landscape in terms of privacy, security and interoperability. It will be no small task for groups like IMPACT to determine the best path forward.
None of these factors matter unless the industry can recruit and retain clinicians. Our field is facing an unprecedented workforce crisis. Traditional healthcare is making clinicians miserable, and COVID has only accelerated the trend of overworked, disenchanted healthcare workers leaving in droves. Clinicians want more interactions with patients, and fewer with computer screens – call it “More face time, less FaceTime.” No new model will succeed unless the industry can more efficiently deploy its talent – arguably its most scarce and precious resource. V1C can help with alleviating the increasing burden and frustration borne by individual physicians in today’s status quo.
In healthcare, new technological approaches inevitably provoke no shortage of skepticism. Past lessons from Silicon Valley-driven fixes have led to understandable cynicism. But V1C is a different breed of animal. By building healthcare around the patient, not the clinic, V1C can make healthcare work better for patients, payers and providers. We’re at a fork in the road: we can revert back to a broken sick-care system, or dig in and do the hard work of figuring out how this future-forward healthcare system gets financed, organized and executed. As a field, we must find the courage and summon the energy to embrace this moment, and make it a moment of change.
Chris Mirabile sprints on a track in Sarasota, Florida, during his daily morning workout. He claims to be a superager already, at age 38, with test results to back it up.
Earlier this year, Harvard scientists reported that they used an anti-aging therapy to reverse blindness in elderly mice. Several other studies in the past decade have suggested that the aging process can be modified, at least in lab organisms. Considering mice and humans share virtually the same genetic makeup, what does the rodent-based study mean for the humans?
In truth, we don’t know. Maybe nothing.
What we do know, however, is that a growing number of people are dedicating themselves to defying the aging process, to turning back the clock – the biological clock, that is. Take Bryan Johnson, a man who is less mouse than human guinea pig. A very wealthy guinea pig.
The 45-year-old venture capitalist spends over $2 million per year reversing his biological clock. To do this, he employs a team of 30 medical doctors and other scientists. His goal is to eventually reset his biological clock to age 18, and “have all of his major organs — including his brain, liver, kidneys, teeth, skin, hair, penis and rectum — functioning as they were in his late teens,” according to a story earlier this year in the New York Post.
But his daily routine paints a picture that is far from appealing: for example, rigorously adhering to a sleep schedule of 8 p.m. to 5 a.m. and consuming more than 100 pills and precisely 1,977 calories daily. Considering all of Johnson’s sacrifices, one discovers a paradox:
To live forever, he must die a little every day until he reaches his goal - if he ever reaches his goal.
Less extreme examples seem more helpful for people interested in happy, healthy aging. Enter Chris Mirabile, a New Yorker who says on his website, SlowMyAge.com, that he successfully reversed his biological age by 13.6 years, from the chronological age of 37.2 to a biological age of 23.6. To put this achievement in perspective, Johnson, to date, has reversed his biological clock by 2.5 years.
Mirabile's habits and overall quest to turn back the clock trace back to a harrowing experience at age 16 during a school trip to Manhattan, when he woke up on the floor with his shirt soaked in blood.
Mirabile, who is now 38, supports his claim with blood tests that purport to measure biological age by assessing changes to a person’s epigenome, or the chemical marks that affect how genes are expressed. Mirabile’s tests have been run and verified independently by the same scientific lab that analyzes Johnson’s. (In an email to Leaps.org, the lab, TruDiagnostic, confirmed Mirabile’s claims about his test results.)
There is considerable uncertainty among scientists about the extent to which these tests can accurately measure biological age in individuals. Even so, Mirabile’s results are intriguing. They could reflect his smart lifestyle for healthy aging.
His habits and overall quest to turn back the clock trace back to a harrowing experience at age 16 during a school trip to Manhattan, when Mirabile woke up on the floor with his shirt soaked in blood. He’d severed his tongue after a seizure. He later learned it was caused by a tumor the size of a golf ball. As a result, “I found myself contemplating my life, what I had yet to experience, and mortality – a theme that stuck with me during my year of recovery and beyond,” Mirabile told me.
For the next 15 years, he researched health and biology, integrating his learnings into his lifestyle. Then, in his early 30s, he came across an article in the journal Cell, "The Hallmarks of Aging," that outlined nine mechanisms of the body that define the aging process. Although the paper says there are no known interventions to delay some of these mechanisms, others, such as inflammation, struck Mirabile as actionable. Reading the paper was his “moment of epiphany” when it came to the areas where he could assert control to maximize his longevity.
He also wanted “to create a resource that my family, friends, and community could benefit from in the short term,” he said. He turned this knowledge base into a company called NOVOS dedicated to extending lifespan.
His longevity advice is more accessible than Johnson’s multi-million dollar approach, as Mirabile spends a fraction of that amount. Mirabile takes one epigenetic test per year and has a gym membership at $45 per month. Unlike Johnson, who takes 100 pills per day, Mirabile takes 10, costing another $45 monthly, including a B-complex, fish oil, Vitamins D3 and K2, and two different multivitamin supplements.
Mirabile’s methods may be easier to apply in other ways as well, since they include activities that many people enjoy anyway. He’s passionate about outdoor activities, travels frequently, and has loving relationships with friends and family, including his girlfriend and collie.
Here are a few of daily routines that could, he thinks, contribute to his impressively young bio age:
After waking at 7:45 am, he immediately drinks 16 ounces of water, with 1/4 teaspoon of sodium and potassium to replenish electrolytes. He takes his morning vitamins, brushes and flosses his teeth, puts on a facial moisturizing sunblock and goes for a brisk, two-mile walk in the sun. At 8:30 am on Mondays, Wednesdays, and Fridays he lift weights, focusing on strength and power, especially in large muscle groups.
Tuesdays, Thursdays and Saturdays are intense cardio days. He runs 5-7 miles or bicycles for 60 minutes first thing in the morning at a brisk pace, listening to podcasts. Sunday morning cardio is more leisurely.
After working out each day, he’s back home at 9:20 am, where he makes black coffee, showers, then applies serum and moisturizing sunblock to his face. He works for about three hours on his laptop, then has a protein shake and fruit.
Mirabile is a dedicated intermittent faster, with a six hour eating window in between 18 hours fasts. At 3 pm, he has lunch. The Mediterranean lineup often features salmon, sardines, olive oil, pink Himalayan salt plus potassium salt for balance, and lots of dried herbs and spices. He almost always finishes with 1/3 to 1/2 bar of dark chocolate.
If you are what you eat, Mirabile is made of mostly plants and lean meats. He follows a Mediterranean diet full of vegetables, fruits, fatty fish and other meats full of protein and unsaturated fats. “These may cost more than a meal at an American fast-food joint, but then again, not by much,” he said. Each day, he spends $25 on all his meals combined.
At 6 pm, he takes the dog out for a two-mile walk, taking calls for work or from family members along the way. At 7 pm, he dines with his girlfriend. Like lunch, this meal is heavy on widely available ingredients, including fish, fresh garlic, and fermented food like kimchi. Mirabile finishes this meal with sweets, like coconut milk yogurt with cinnamon and clove, some stevia, a mix of fresh berries and cacao nibs.
If Mirabile's epigenetic tests are accurate, his young biological age could be thanks to his healthy lifestyle, or it could come from a stroke of luck if he inherited genes that protect against aging.
At 8 pm, he wraps up work duties and watches shows with his girlfriend, applies serum and moisturizer yet again, and then meditates with the lights off. This wind-down, he said, improves his sleep quality. Wearing a sleep mask and earplugs, he’s asleep by about 10:30.
“I’ve achieved stellar health outcomes, even after having had the physiological stressors of a brain tumor, without spending a fortune,” Mirabile said. “In fact, even during times when I wasn’t making much money as a startup founder with few savings, I still managed to live a very healthy, pro-longevity lifestyle on a modest budget.”
Mirabile said living a cleaner, healthier existence is a reality that many readers can achieve. It’s certainly true that many people live in food deserts and have limited time for exercise or no access to gyms, but James R. Doty, a clinical professor of neurosurgery at Stanford, thinks many can take more action to stack the odds that they’ll “be happy and live longer.” Many of his recommendations echo aspects of Mirabile’s lifestyle.
Each night, Doty said, it’s vital to get anywhere between 6-8 hours of good quality sleep. Those who sleep less than 6 hours per night are at an increased risk of developing a whole host of medical problems, including high blood pressure, type 2 diabetes, and stroke.
In addition, it’s critical to follow Mirabile’s prescription of exercise for about one hour each day, and intensity levels matter. Doty noted that, in 2017, researchers at Brigham Young University found that people who ran at a fast pace for 30-40 minutes five days per week were, on average, biologically younger by nine years, compared to those who subscribed to more moderate exercise programs, as well as those who rarely exercised.
When it comes to nutrition, one should consider fasting for 16 hours per day, Doty said. This is known as the 16/8 method, where one’s daily calories are consumed within an eight hour window, fasting for the remaining 16 hours, just like Mirabile. Intermittent fasting is associated with cellular repair and less inflammation, though it’s not for everyone, Doty added. Consult with a medical professional before trying a fasting regimen.
Finally, Doty advised to “avoid anger, avoid stress.” Easier said than done, but not impossible. “Between stimulus and response, there is a pause and within that pause lies your freedom,” Doty said. Mirabile’s daily meditation ritual could be key to lower stress for healthy aging. Research has linked regular, long-term meditation to having a lower epigenetic age, compared to control groups.
Many other factors could apply. Having a life purpose, as Mirabile does with his company, has also been associated with healthy aging and lower epigenetic age. Of course, Mirabile is just one person, so it’s hard to know how his experience will apply to others. If his tests are accurate, his young biological age could be thanks to his healthy lifestyle, or it could come from a stroke of luck if he inherited genes that protect against aging. Clearly, though, any such genes did not protect him from cancer at an early age.
The third and perhaps most likely explanation: Mirabile’s very young biological age results from a combination of these factors. Some research shows that genetics account for only 25 percent of longevity. That means environmental factors could be driving the other 75 percent, such as where you live, frequency of exercise, quality of nutrition and social support.
The middle-aged – even Brian Johnson – probably can’t ever be 18 again. But more modest goals are reasonable for many. Control what you can for a longer, healthier life.
FDA, researchers work to make clinical trials more diverse
The U.S. population is becoming more diverse, but clinical trials don't reflect that, experts say. Some are focusing on recruiting minorities to participate in research.
Nestled in a predominately Hispanic neighborhood, a new mural outside Guadalupe Centers Middle School in Kansas City, Missouri imparts a powerful message: “Clinical Research Needs Representation.” The colorful portraits painted above those words feature four cancer survivors of different racial and ethnic backgrounds. Two individuals identify as Hispanic, one as African American and another as Native American.
One of the patients depicted in the mural is Kim Jones, a 51-year-old African American breast cancer survivor since 2012. She advocated for an African American friend who participated in several clinical trials for ovarian cancer. Her friend was diagnosed in an advanced stage at age 26 but lived nine more years, thanks to the trials testing new therapeutics. “They are definitely giving people a longer, extended life and a better quality of life,” said Jones, who owns a nail salon. And that’s the message the mural aims to send to the community: Clinical trials need diverse participants.
While racial and ethnic minority groups represent almost half of the U.S. population, the lack of diversity in clinical trials poses serious challenges. Limited awareness and access impede equitable representation, which is necessary to prove the safety and effectiveness of medical interventions across different groups.
A Yale University study on clinical trial diversity published last year in BMJ Medicine found that while 81 percent of trials testing the new cancer drugs approved by the U.S. Food and Drug Administration between 2012 and 2017 included women, only 23 percent included older adults and 5 percent fairly included racial and ethnic minorities. “It’s both a public health and social justice issue,” said Jennifer E. Miller, an associate professor of medicine at Yale School of Medicine. “We need to know how medicines and vaccines work for all clinically distinct groups, not just healthy young White males.” A recent JAMA Oncology editorial stresses out the need for legislation that would require diversity action plans for certain types of trials.
Ensuring meaningful representation of racial and ethnic minorities in clinical trials for regulated medical products is fundamental to public health.--FDA Commissioner Robert M. Califf.
But change is on the horizon. Last April, the FDA issued a new draft guidance encouraging industry to find ways to revamp recruitment into clinical trials. The announcement, which expanded on previous efforts, called for including more participants from underrepresented racial and ethnic segments of the population.
“The U.S. population has become increasingly diverse, and ensuring meaningful representation of racial and ethnic minorities in clinical trials for regulated medical products is fundamental to public health,” FDA commissioner Robert M. Califf, a physician, said in a statement. “Going forward, achieving greater diversity will be a key focus throughout the FDA to facilitate the development of better treatments and better ways to fight diseases that often disproportionately impact diverse communities. This guidance also further demonstrates how we support the Administration’s Cancer Moonshot goal of addressing inequities in cancer care, helping to ensure that every community in America has access to cutting-edge cancer diagnostics, therapeutics and clinical trials.”
Lola Fashoyin-Aje, associate director for Science and Policy to Address Disparities in the Oncology Center of Excellence at the FDA, said that the agency “has long held the view that clinical trial participants should reflect the clinical and demographic characteristics of the patients who will ultimately receive the drug once approved.” However, “numerous studies over many decades” have measured the extent of underrepresentation. One FDA analysis found that the proportion of White patients enrolled in U.S. clinical trials (88 percent) is much higher than their numbers in country's population. Meanwhile, the enrollment of African American and Native Hawaiian/American Indian and Alaskan Native patients is below their national numbers.
The FDA’s guidance is accelerating researchers’ efforts to be more inclusive of diverse groups in clinical trials, said Joyce Sackey, a clinical professor of medicine and associate dean at Stanford School of Medicine. Underrepresentation is “a huge issue,” she noted. Sackey is focusing on this in her role as the inaugural chief equity, diversity and inclusion officer at Stanford Medicine, which encompasses the medical school and two hospitals.
Until the early 1990s, Sackey pointed out, clinical trials were based on research that mainly included men, as investigators were concerned that women could become pregnant, which would affect the results. This has led to some unfortunate consequences, such as indications and dosages for drugs that cause more side effects in women due to biological differences. “We’ve made some progress in including women, but we have a long way to go in including people of different ethnic and racial groups,” she said.

A new mural outside Guadalupe Centers Middle School in Kansas City, Missouri, advocates for increasing diversity in clinical trials. Kim Jones, 51-year-old African American breast cancer survivor, is second on the left.
Artwork by Vania Soto. Photo by Megan Peters.
Among racial and ethnic minorities, distrust of clinical trials is deeply rooted in a history of medical racism. A prime example is the Tuskegee Study, a syphilis research experiment that started in 1932 and spanned 40 years, involving hundreds of Black men with low incomes without their informed consent. They were lured with inducements of free meals, health care and burial stipends to participate in the study undertaken by the U.S. Public Health Service and the Tuskegee Institute in Alabama.
By 1947, scientists had figured out that they could provide penicillin to help patients with syphilis, but leaders of the Tuskegee research failed to offer penicillin to their participants throughout the rest of the study, which lasted until 1972.
Opeyemi Olabisi, an assistant professor of medicine at Duke University Medical Center, aims to increase the participation of African Americans in clinical research. As a nephrologist and researcher, he is the principal investigator of a clinical trial focusing on the high rate of kidney disease fueled by two genetic variants of the apolipoprotein L1 (APOL1) gene in people of recent African ancestry. Individuals of this background are four times more likely to develop kidney failure than European Americans, with these two variants accounting for much of the excess risk, Olabisi noted.
The trial is part of an initiative, CARE and JUSTICE for APOL1-Mediated Kidney Disease, through which Olabisi hopes to diversify study participants. “We seek ways to engage African Americans by meeting folks in the community, providing accessible information and addressing structural hindrances that prevent them from participating in clinical trials,” Olabisi said. The researchers go to churches and community organizations to enroll people who do not visit academic medical centers, which typically lead clinical trials. Since last fall, the initiative has screened more than 250 African Americans in North Carolina for the genetic variants, he said.
Other key efforts are underway. “Breaking down barriers, including addressing access, awareness, discrimination and racism, and workforce diversity, are pivotal to increasing clinical trial participation in racial and ethnic minority groups,” said Joshua J. Joseph, assistant professor of medicine at the Ohio State University Wexner Medical Center. Along with the university’s colleges of medicine and nursing, researchers at the medical center partnered with the African American Male Wellness Agency, Genentech and Pfizer to host webinars soliciting solutions from almost 450 community members, civic representatives, health care providers, government organizations and biotechnology professionals in 25 states and five countries.
Their findings, published in February in the journal PLOS One, suggested that including incentives or compensation as part of the research budget at the institutional level may help resolve some issues that hinder racial and ethnic minorities from participating in clinical trials. Compared to other groups, more Blacks and Hispanics have jobs in service, production and transportation, the authors note. It can be difficult to get paid leave in these sectors, so employees often can’t join clinical trials during regular business hours. If more leaders of trials offer money for participating, that could make a difference.
Obstacles include geographic access, language and other communications issues, limited awareness of research options, cost and lack of trust.
Christopher Corsico, senior vice president of development at GSK, formerly GlaxoSmithKline, said the pharmaceutical company conducted a 17-year retrospective study on U.S. clinical trial diversity. “We are using epidemiology and patients most impacted by a particular disease as the foundation for all our enrollment guidance, including study diversity plans,” Corsico said. “We are also sharing our results and ideas across the pharmaceutical industry.”
Judy Sewards, vice president and head of clinical trial experience at Pfizer’s headquarters in New York, said the company has committed to achieving racially and ethnically diverse participation at or above U.S. census or disease prevalence levels (as appropriate) in all trials. “Today, barriers to clinical trial participation persist,” Sewards said. She noted that these obstacles include geographic access, language and other communications issues, limited awareness of research options, cost and lack of trust. “Addressing these challenges takes a village. All stakeholders must come together and work collaboratively to increase diversity in clinical trials.”
It takes a village indeed. Hope Krebill, executive director of the Masonic Cancer Alliance, the outreach network of the University of Kansas Cancer Center in Kansas City, which commissioned the mural, understood that well. So her team actively worked with their metaphorical “village.” “We partnered with the community to understand their concerns, knowledge and attitudes toward clinical trials and research,” said Krebill. “With that information, we created a clinical trials video and a social media campaign, and finally, the mural to encourage people to consider clinical trials as an option for care.”
Besides its encouraging imagery, the mural will also be informational. It will include a QR code that viewers can scan to find relevant clinical trials in their location, said Vania Soto, a Mexican artist who completed the rendition in late February. “I’m so honored to paint people that are survivors and are living proof that clinical trials worked for them,” she said.
Jones, the cancer survivor depicted in the mural, hopes the image will prompt people to feel more open to partaking in clinical trials. “Hopefully, it will encourage people to inquire about what they can do — how they can participate,” she said.

