Would You Want to Know a Decade Early If You Were Getting Alzheimer's?

The author pictured with her husband Dallas, who has Alzheimer's.
Editor's Note: A team of researchers in Italy recently used artificial intelligence and machine learning to diagnose Alzheimer's disease on a brain scan an entire decade before symptoms show up in the patient. While some people argue that early detection is critical, others believe the knowledge would do more harm than good. LeapsMag invited contributors with opposite opinions to share their perspectives.
I first realized something was wrong with my dad when I came home for Thanksgiving 20 years ago.
I hadn't seen my family for more than a year after moving from New York to California. My father was meticulous, a multi-shower a day man, a regular Beau Brummell. He was never officially diagnosed with dementia, but it was easy to figure out after he stopped leaving the house, stopped reading, stopped being himself. My mother knew, but she never sought help. After his illness showed itself, I asked her if she considered a nursing home. "Never," she told me. "I can take care of him." And she did.
She gave herself a break once to visit me, and it was the first time she traveled separately from him since they eloped at seventeen. My brother watched my father, and it was not smooth. Dad was angry, hallucinating, and demanding his gun, which had been disposed of long ago. While Mom was visiting me in California, we played some board games. One demanded honest answers. The card read, What are you most afraid of? "Dementia," she said.
My father never saw this coming, none of us did.
Dementia ran on my mother's side. Her mother, my Nana, was senile, the popular diagnosis for older folks back then. My grandfather tried his hardest to take care of her, but she kept escaping their tidy 6th floor apartment to run away. My mother would go over every day to take care of them, but once my grandfather became ill, she took her mother into our apartment. She had two small children, Nana, and her husband in a two-bedroom flat. Nana talked to people under plates, wore tissues on her head, and tried to escape. We were on the first floor, so she could run into traffic if all eyes weren't on her. Soon, it was too much, even for my Wonder Woman mom. Nana was placed in a nursing home and died soon after.
My mother dropped dead on a NYC sidewalk two years after my father started to deteriorate. She was probably going to the store to buy milk and cigarettes. A kind stranger called 911, and a cop came to my parent's door soon after to tell my dad the news. My father cried for death, raged and ranted, then calmed down enough to come back as the dad we remembered for the week of mourning. He even ordered a Manhattan at dinner. His death came exactly a week and an hour after my mother's. He died of a broken heart. My husband cried with all his body after we left the cemetery, weeping, "Poor Buck. Poor Buck." I never saw him cry before.
Now, 18 years later, I sit here with my husband, 59 years old, as he suffers from the same hideous disease.
He is talking to someone I can't see, even laughing with him. He holds a Ph.D. in literature, taught college, had a single handicap golf game, and ate well. We never saw this coming. One day he went to type and jumbled letters came on the screen. He would show up late or early for his classes, wondering what was wrong with the students. He started running red lights. He was graciously counseled to retire, and he did, at 55. His doctor told him it was depression. The second opinion agreed. He was told to do nothing for a year, and he did. He played golf a bit, then one day he couldn't speak or think clearly. I came home from work to find him roaming the neighborhood, eyes ablaze, muttering to himself. I went on family leave. Many tests later we got the working diagnosis, but it meant nothing to him. He never reacted to the words Primary Progressive Aphasia or dementia. I was glad. If he was lucid, I knew what he would talk about doing. He told me after my dad's death that he did not want that life for himself.
I worry I may get it, too. It almost seems inescapable. Dementia has no cure, and the treatments for the symptoms are hit and miss. I thought about getting the full flight of predictive tests, but I know myself, and I scare myself into bracing for the worst. Others scare me, too, when I read their online statements about ending their lives if they learn they have it: I told my children to take me to a state where assisted suicide is legal; it's easy to overdose; I don't want to be a burden on my children. These are caregivers on social media forums. They live with the terror, eyes wide open. We have no children, but who would I burden? My sisters? My brother? Do I stay or do I go? This disease invites pandemonium. Assisted murder-suicides with caregiver spouses of those with dementia don't merit headlines, but their stories are on the sidebars. No thanks. I work on God's timeline.
There are no survivors – yet.
A diagnosis today would paralyze me and create melancholy for all who know me. I would second guess everything, I would read everything, I would cry, I would hardly live. I would be tempted to pick up that first drink after 20 plus years sober. I would even think about ending my life. It would be difficult not to consider. As a high school English teacher, I talk about suicide when I teach Hamlet. I tell the students suicide is a permanent solution to a temporary problem. Dementia isn't temporary. There are no survivors – yet.
I often think what my relatives would have done with an advance diagnosis. My grandmother was a classic worrier. She would have been beyond distraught. My father might have found that gun. My husband would have taken the right number of pills.
An advance diagnosis would paralyze me.
I appreciate the arguments for early diagnosis. Some people are made of sterner stuff. They have the mindset I lack. I admire so many who are contributing to the current conversation about dementia and are active advocates for a cure. They have found a purpose in their fate.
I don't need a test to get my ducks in a row. Loving those with dementia has prompted me to be prepared. I have a different type of bucket list: reset my priorities, slow down, be present, educate others, and make my legal plans. If and when it happens, there will be time for toast and tea and a walk along the shore. There will be time to plan for the inevitable and unenviable end. I am morbid enough to know I will recognize the purple elephant in the room. I don't want the shock and awe now. I can wait. My sisters agree. We will keep our elbows out.
Editor's Note: Consider the other side of the argument here.
Meet Dr. Renee Wegrzyn, the first Director of President Biden's new health agency, ARPA-H
Today's podcast guest, Dr. Renee Wegrzyn, directs ARPA-H, a new agency formed last year to spearhead health innovations. Time will tell if ARPA-H will produce advances on the level of its fellow agency, DARPA.
In today’s podcast episode, I talk with Renee Wegrzyn, appointed by President Biden as the first director of a health agency created last year, the Advanced Research Projects Agency for Health, or ARPA-H. It’s inspired by DARPA, the agency that develops innovations for the Defense department and has been credited with hatching world-changing technologies such as ARPANET, which became the internet.
Time will tell if ARPA-H will lead to similar achievements in the realm of health. That’s what President Biden and Congress expect in return for funding ARPA-H at 2.5 billion dollars over three years.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
How will the agency figure out which projects to take on, especially with so many patient advocates for different diseases demanding moonshot funding for rapid progress?
I talked with Dr. Wegrzyn about the opportunities and challenges, what lessons ARPA-H is borrowing from Operation Warp Speed, how she decided on the first ARPA-H project that was announced recently, why a separate agency was needed instead of reforming HHS and the National Institutes of Health to be better at innovation, and how ARPA-H will make progress on disease prevention in addition to treatments for cancer, Alzheimer’s and diabetes, among many other health priorities.
Dr. Wegrzyn’s resume leaves no doubt of her suitability for this role. She was a program manager at DARPA where she focused on applying gene editing and synthetic biology to the goal of improving biosecurity. For her work there, she received the Superior Public Service Medal and, in case that wasn’t enough ARPA experience, she also worked at another ARPA that leads advanced projects in intelligence, called I-ARPA. Before that, she ran technical teams in the private sector working on gene therapies and disease diagnostics, among other areas. She has been a vice president of business development at Gingko Bioworks and headed innovation at Concentric by Gingko. Her training and education includes a PhD and undergraduate degree in applied biology from the Georgia Institute of Technology and she did her postdoc as an Alexander von Humboldt Fellow in Heidelberg, Germany.
Dr. Wegrzyn told me that she’s “in the hot seat.” The pressure is on for ARPA-H especially after the need and potential for health innovation was spot lit by the pandemic and the unprecedented speed of vaccine development. We'll soon find out if ARPA-H can produce gamechangers in health that are equivalent to DARPA’s creation of the internet.
Show links:
ARPA-H - https://arpa-h.gov/
Dr. Wegrzyn profile - https://arpa-h.gov/people/renee-wegrzyn/
Dr. Wegrzyn Twitter - https://twitter.com/rwegrzyn?lang=en
President Biden Announces Dr. Wegrzyn's appointment - https://www.whitehouse.gov/briefing-room/statement...
Leaps.org coverage of ARPA-H - https://leaps.org/arpa/
ARPA-H program for joints to heal themselves - https://arpa-h.gov/news/nitro/ -
ARPA-H virtual talent search - https://arpa-h.gov/news/aco-talent-search/

Dr. Renee Wegrzyn was appointed director of ARPA-H last October.
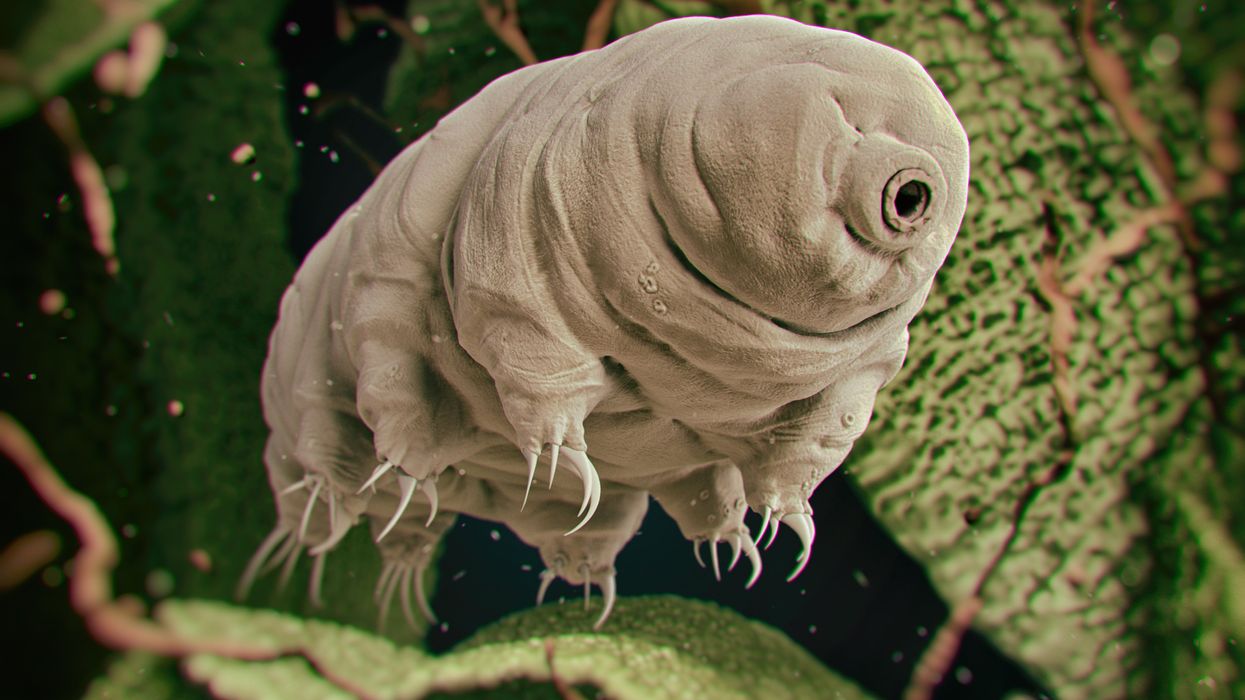
Tiny, tough “water bears” may help bring new vaccines and medicines to sub-Saharan Africa
Tardigrades can completely dehydrate and later rehydrate themselves, a survival trick that scientists are harnessing to preserve medicines in hot temperatures.
Microscopic tardigrades, widely considered to be some of the toughest animals on earth, can survive for decades without oxygen or water and are thought to have lived through a crash-landing on the moon. Also known as water bears, they survive by fully dehydrating and later rehydrating themselves – a feat only a few animals can accomplish. Now scientists are harnessing tardigrades’ talents to make medicines that can be dried and stored at ambient temperatures and later rehydrated for use—instead of being kept refrigerated or frozen.
Many biologics—pharmaceutical products made by using living cells or synthesized from biological sources—require refrigeration, which isn’t always available in many remote locales or places with unreliable electricity. These products include mRNA and other vaccines, monoclonal antibodies and immuno-therapies for cancer, rheumatoid arthritis and other conditions. Cooling is also needed for medicines for blood clotting disorders like hemophilia and for trauma patients.
Formulating biologics to withstand drying and hot temperatures has been the holy grail for pharmaceutical researchers for decades. It’s a hard feat to manage. “Biologic pharmaceuticals are highly efficacious, but many are inherently unstable,” says Thomas Boothby, assistant professor of molecular biology at University of Wyoming. Therefore, during storage and shipping, they must be refrigerated at 2 to 8 degrees Celsius (35 to 46 degrees Fahrenheit). Some must be frozen, typically at -20 degrees Celsius, but sometimes as low -90 degrees Celsius as was the case with the Pfizer Covid vaccine.
For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
The costly cold chain
The logistics network that ensures those temperature requirements are met from production to administration is called the cold chain. This cold chain network is often unreliable or entirely lacking in remote, rural areas in developing nations that have malfunctioning electrical grids. “Almost all routine vaccines require a cold chain,” says Christopher Fox, senior vice president of formulations at the Access to Advanced Health Institute. But when the power goes out, so does refrigeration, putting refrigerated or frozen medical products at risk. Consequently, the mRNA vaccines developed for Covid-19 and other conditions, as well as more traditional vaccines for cholera, tetanus and other diseases, often can’t be delivered to the most remote parts of the world.
To understand the scope of the challenge, consider this: In the U.S., more than 984 million doses of Covid-19 vaccine have been distributed so far. Each one needed refrigeration that, even in the U.S., proved challenging. Now extrapolate to all vaccines and the entire world. For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
Globally, the cold chain packaging market is valued at over $15 billion and is expected to exceed $60 billion by 2033.
Adobe Stock
Freeze-drying, also called lyophilization, which is common for many vaccines, isn’t always an option. Many freeze-dried vaccines still need refrigeration, and even medicines approved for storage at ambient temperatures break down in the heat of sub-Saharan Africa. “Even in a freeze-dried state, biologics often will undergo partial rehydration and dehydration, which can be extremely damaging,” Boothby explains.
The cold chain is also very expensive to maintain. The global pharmaceutical cold chain packaging market is valued at more than $15 billion, and is expected to exceed $60 billion by 2033, according to a report by Future Market Insights. This cost is only expected to grow. According to the consulting company Accenture, the number of medicines that require the cold chain are expected to grow by 48 percent, compared to only 21 percent for non-cold-chain therapies.
Tardigrades to the rescue
Tardigrades are only about a millimeter long – with four legs and claws, and they lumber around like bears, thus their nickname – but could provide a big solution. “Tardigrades are unique in the animal kingdom, in that they’re able to survive a vast array of environmental insults,” says Boothby, the Wyoming professor. “They can be dried out, frozen, heated past the boiling point of water and irradiated at levels that are thousands of times more than you or I could survive.” So, his team is gradually unlocking tardigrades’ survival secrets and applying them to biologic pharmaceuticals to make them withstand both extreme heat and desiccation without losing efficacy.
Boothby’s team is focusing on blood clotting factor VIII, which, as the name implies, causes blood to clot. Currently, Boothby is concentrating on the so-called cytoplasmic abundant heat soluble (CAHS) protein family, which is found only in tardigrades, protecting them when they dry out. “We showed we can desiccate a biologic (blood clotting factor VIII, a key clotting component) in the presence of tardigrade proteins,” he says—without losing any of its effectiveness.
The researchers mixed the tardigrade protein with the blood clotting factor and then dried and rehydrated that substance six times without damaging the latter. This suggests that biologics protected with tardigrade proteins can withstand real-world fluctuations in humidity.
Furthermore, Boothby’s team found that when the blood clotting factor was dried and stabilized with tardigrade proteins, it retained its efficacy at temperatures as high as 95 degrees Celsius. That’s over 200 degrees Fahrenheit, much hotter than the 58 degrees Celsius that the World Meteorological Organization lists as the hottest recorded air temperature on earth. In contrast, without the protein, the blood clotting factor degraded significantly. The team published their findings in the journal Nature in March.
Although tardigrades rarely live more than 2.5 years, they have survived in a desiccated state for up to two decades, according to Animal Diversity Web. This suggests that tardigrades’ CAHS protein can protect biologic pharmaceuticals nearly indefinitely without refrigeration or freezing, which makes it significantly easier to deliver them in locations where refrigeration is unreliable or doesn’t exist.
The tricks of the tardigrades
Besides the CAHS proteins, tardigrades rely on a type of sugar called trehalose and some other protectants. So, rather than drying up, their cells solidify into rigid, glass-like structures. As that happens, viscosity between cells increases, thereby slowing their biological functions so much that they all but stop.
Now Boothby is combining CAHS D, one of the proteins in the CAHS family, with trehalose. He found that CAHS D and trehalose each protected proteins through repeated drying and rehydrating cycles. They also work synergistically, which means that together they might stabilize biologics under a variety of dry storage conditions.
“We’re finding the protective effect is not just additive but actually is synergistic,” he says. “We’re keen to see if something like that also holds true with different protein combinations.” If so, combinations could possibly protect against a variety of conditions.
Commercialization outlook
Before any stabilization technology for biologics can be commercialized, it first must be approved by the appropriate regulators. In the U.S., that’s the U.S. Food and Drug Administration. Developing a new formulation would require clinical testing and vast numbers of participants. So existing vaccines and biologics likely won’t be re-formulated for dry storage. “Many were developed decades ago,” says Fox. “They‘re not going to be reformulated into thermo-stable vaccines overnight,” if ever, he predicts.
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits.
Instead, this technology is most likely to be used for the new products and formulations that are just being created. New and improved vaccines will be the first to benefit. Good candidates include the plethora of mRNA vaccines, as well as biologic pharmaceuticals for neglected diseases that affect parts of the world where reliable cold chain is difficult to maintain, Boothby says. Some examples include new, more effective vaccines for malaria and for pathogenic Escherichia coli, which causes diarrhea.
Tallying up the benefits
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits. For instance, MenAfriVac, a meningitis vaccine (without tardigrade proteins) developed for sub-Saharan Africa, can be stored at up to 40 degrees Celsius for four days before administration. “If you have a few days where you don’t need to maintain the cold chain, it’s easier to transport vaccines to remote areas,” Fox says, where refrigeration does not exist or is not reliable.
Better health is an obvious benefit. MenAfriVac reduced suspected meningitis cases by 57 percent in the overall population and more than 99 percent among vaccinated individuals.
Lower healthcare costs are another benefit. One study done in Togo found that the cold chain-related costs increased the per dose vaccine price up to 11-fold. The ability to ship the vaccines using the usual cold chain, but transporting them at ambient temperatures for the final few days cut the cost in half.
There are environmental benefits, too, such as reducing fuel consumption and greenhouse gas emissions. Cold chain transports consume 20 percent more fuel than non-cold chain shipping, due to refrigeration equipment, according to the International Trade Administration.
A study by researchers at Johns Hopkins University compared the greenhouse gas emissions of the new, oral Vaxart COVID-19 vaccine (which doesn’t require refrigeration) with four intramuscular vaccines (which require refrigeration or freezing). While the Vaxart vaccine is still in clinical trials, the study found that “up to 82.25 million kilograms of CO2 could be averted by using oral vaccines in the U.S. alone.” That is akin to taking 17,700 vehicles out of service for one year.
Although tardigrades’ protective proteins won’t be a component of biologic pharmaceutics for several years, scientists are proving that this approach is viable. They are hopeful that a day will come when vaccines and biologics can be delivered anywhere in the world without needing refrigerators or freezers en route.