Americans Fell for a Theranos-Style Scam 100 Years Ago. Will We Ever Learn?

Medical scams like Theranos are as American as America itself.
The huckster understands what people want – an easy route to good health -- and figures out just how to provide it as long as no one asks too many questions.
"Americans are very much prone to this sort of thinking: Give me a pill or give me a magical bean that can make me lose weight!"
The keys to success: Hoopla, fancy technology, and gullibility. And oh yes, one more thing: a blood sample. Well, lots and lots of blood samples. Every testing fee counts.
Sound familiar? It could be the story of the preternaturally persuasive Elizabeth Holmes, the disgraced founder of Theranos who stands accused of perpetrating a massive blood-testing fraud. But this is a different story from a different time, one that dates back 100 years but sounds almost like it could unfold on the front page of The Wall Street Journal today.
The main difference: Back then, watchdogs thought they'd be able to vanquish fake medicine and scam science. Fat chance, it turned out. It seems like we're more likely to lose-weight-quick than make much of a dent into quackery and health fraud.
Why? Have we learned anything at all over the past century? As we sweep into a new decade, experts says we're not as advanced as we'd like to think. But the fight against fraud and fakery continues.
Quackery: As American As America Itself
In the 17th century, British healers of questionable reputation got a new name -- "quack," from the Dutch word "quacksalver," which originally referred to someone who treats others with home remedies but developed a new meaning along the lines of "charlatan." And these quacks got a new place to sell their wares: the American colonies.
By 1692, a Boston newspaper advertised a patent medicine that promised to cure "the Griping of the Guts, and the Wind Cholick" and – for good measure – "preventeth that woeful Distemper of the Dry Belly Ach." A couple centuries later, the most famous woman in the United States wasn't a first lady or feminist but a hawker of nostrums named Lydia Estes Pinkham whose "vegetable compound" promised to banish "female complaints." One advertisement suggested that the "sure cure" would have saved the life of a Connecticut clergyman whose wife killed him after suffering from feminine maladies for 16 years.
By the early 20th century, Americans were fascinated by electricity and radiation, and both healers and hucksters embraced the new high-tech era. Men with flagging libidos, for example, could irradiate their private parts with the radioactive Radiendocrinator or buy battery-powered electric belts equipped with dangling bits to supercharge their, um, dangling bits.
The Rise of the Radio Wave 'Cure'
Enter radionics, the (supposed) science of better health via radio waves. The idea was that "healthy people radiate healthy energy," and sickness could be reversed through diagnosis and re-tuning, write Dr. Lydia Kang and Nate Pedersen in their 2017 book "Quackery: A Brief History of the Worst Ways to Cure Everything."
Detecting illness and fixing it required machinery -- Dynamizers, Radioclasts and Oscillocasts – that could cost hundreds of dollars each. Thousands of physicians bought them. Fortunately, they could work remotely, for a fee. The worried-and-potentially-unwell just needed to send a blood sample and, of course, a personal check.
Sting operations revealed radionics to be bogus. A skeptic sent a blood sample to one radionics practitioner in Albuquerque who reported back with news of an infected fallopian tube. In fact, the blood sample came from a male guinea pig. As an American Medical Association leader reported, the guinea pig "had shown no female characteristics up to that time, and a postmortem examination yielded no evidence of ladylike attributes."
When Quackery Refused to Yield
The rise of bogus medical technology in the early 20th century spawned a watchdog industry as organizations like the American Medical Association swept into action, said medical historian Eric Boyle, author of 2012's "Quack Medicine: A History of Combating Health Fraud in Twentieth-Century America."
"When quackery was recognized as a major problem, the people who campaigned for its demise were confident that they could get rid of it," he said. "A lot of people believed that increased education, the truths of science, and laws designed to protect consumers would ultimately drive quackery from the marketplace. And then throughout the century, as modern medicine developed, and more effectively treated one disease after another, many observers remained confident in that prediction."
There's a bid to "flood the information highway with truth to turn the storm of fake promotional stuff into a trickle."
But fake medicine persisted as Americans continued their quest to get- healthy-quick… or get-rich-quick by promising to help others to get- healthy-quick. Even radionics refused to die. It's still around in various forms. And, as the Theranos scandal reveals, we're still hoping our blood can offer the keys to longevity and good health.
Why Do We Still Fall for Scams?
In our own era, the Theranos company rose to prominence when founder and CEO Elizabeth Holmes convinced journalists and investors that she'd found a way to cheaply test drops of blood for hundreds of conditions. Then it all fell apart, famously, when the world learned that the technology didn't work. The company has folded, and Holmes faces a federal trial on fraud charges this year.
"There were a lot of prominent, very smart people who bought into the myth of Elizabeth Holmes," a former employee told "60 Minutes," even though the blood tests never actually worked as advertised.
Shouldn't "prominent, very smart people" know better? "People are gullible," said Dr. Stephen Barrett, a psychiatrist and leading quack-buster who runs the QuackWatch website. But there's more to the story. According to him, we're uniquely vulnerable as individuals to bogus medicine.
Scam artists specifically pinpoint their target audiences, such as "smart people," desperate people and alienated people, he said.
Smart people, for example, might be overconfident about their ability to detect fraud and fall for bogus medicine. Alienated people may distrust the establishment, whether it's the medical field or government watchdogs, and be more receptive to alternative sources of information.
Dr. Barrett also points a finger at magical thinking, which comes in different forms. It could mean a New Age-style belief that our minds can control the world around us. Or, as professional quack-buster Alex Berezow said, it could refer to "our cultural obsession with quick fixes."
"Americans are very much prone to this sort of thinking: Give me a pill or give me a magical bean that can make me lose weight! But complex problems need complex solutions," said Berezow, a microbiologist who debunks junk science in his job as a spokesman for the American Council on Science & Health.
American mistrust of expertise makes matters worse, he said. "When I tell people they need to get vaccinated, I'm called a shill for the pharmaceutical industry," he said. "If I say dietary supplements generally don't work, I'm a shill for doctors who want to keep people sick."
What can ordinary citizens do to protect themselves from fake medicine? "You have to have a healthy skepticism of everything," Berezow said. "When you come across something new, is someone trying to take advantage of you? It's a horrible way to think about the world, but there's some truth to it."
"Like any chronic disease, we will have to live with it while we do our best to fight it."
The government and experts have their own roles to play via regulation and education, respectively. For all the criticism it gets, the Food & Drug Administration does serve as a bulwark against fakery in prescription medicine. And while celebrities like Gwyneth "Goop" Paltrow hawk countless questionable medical products on the Internet, scientists and physicians are fighting back by using social media as a tool to promote the truth. There's a bid to "flood the information highway with truth to turn the storm of fake promotional stuff into a trickle," said Dr. Randi Hutter Epstein, a writer in residence at Yale School of Medicine and author of 2018's "Aroused: The History of Hormones and How They Control Just About Everything."
What's next? Like death, taxes and Cher, charlatans are likely to always be with us. Boyle quoted the late William Jarvis, a pioneering quack-buster in the late 20th century who believed health fraud would never be eradicated: "Like any chronic disease, we will have to live with it while we do our best to fight it."
Three Big Biotech Ideas to Watch in 2020—And Beyond
Body-on-a-chip, prime editing, and gut microbes all are poised to make a big impact in 2020.
1. Happening Now: Body-on-a-Chip Technology Is Enabling Safer Drug Trials and Better Cancer Research
Researchers have increasingly used the technology known as "lab-on-a-chip" or "organ-on-a-chip" to test the effects of pharmaceuticals, toxins, and chemicals on humans. Rather than testing on animals, which raises ethical concerns and can sometimes be inaccurate, and human-based clinical trials, which can be expensive and difficult to iterate, scientists turn to tiny, micro-engineered chips—about the size of a thumb drive.
It's possible that doctors could one day take individual cell samples and create personalized treatments, testing out any medications on the chip.
The chips are lined with living samples of human cells, which mimic the physiology and mechanical forces experienced by cells inside the human body, down to blood flow and breathing motions; the functions of organs ranging from kidneys and lungs to skin, eyes, and the blood-brain barrier.
A more recent—and potentially even more useful—development takes organ-on-a-chip technology to the next level by integrating several chips into a "body-on-a-chip." Since human organs don't work in isolation, seeing how they all react—and interact—once a foreign element has been introduced can be crucial to understanding how a certain treatment will or won't perform. Dr. Shyni Varghese, a MEDx investigator at the Duke University School of Medicine, is one of the researchers working with these systems in order to gain a more nuanced understanding of how multiple different organs react to the same stimuli.
Her lab is working on "tumor-on-a-chip" models, which can not only show the progression and treatment of cancer, but also model how other organs would react to immunotherapy and other drugs. "The effect of drugs on different organs can be tested to identify potential side effects," Varghese says. In addition, these models can help the researchers figure out how cancers grow and spread, as well as how to effectively encourage immune cells to move in and attack a tumor.
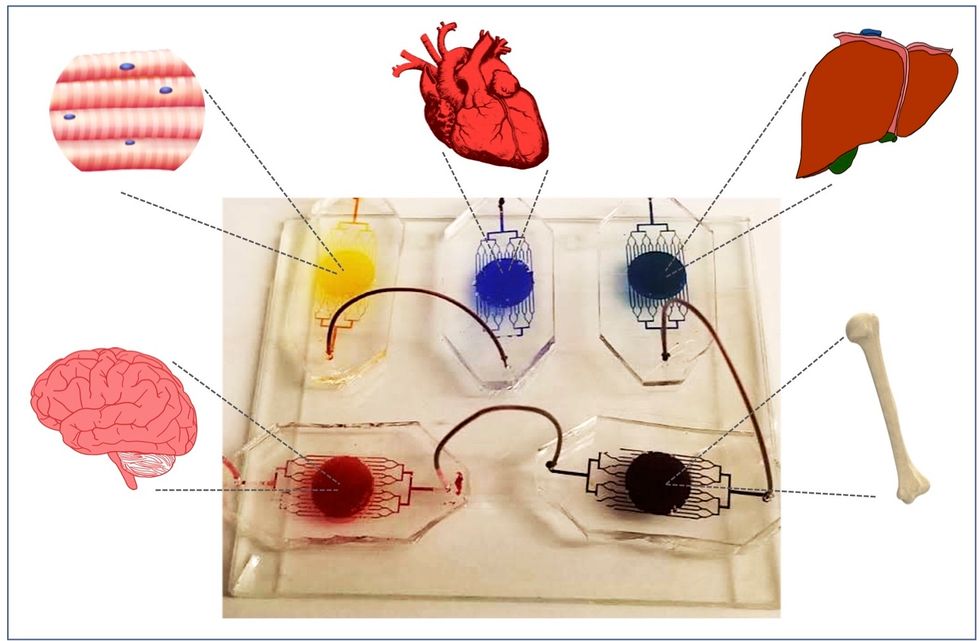
One body-on-a-chip used by Dr. Varghese's lab tracks the interactions of five organs—brain, heart, liver, muscle, and bone.
As their research progresses, Varghese and her team are looking for ways to maintain the long-term function of the engineered organs. In addition, she notes that this kind of research is not just useful for generalized testing; "organ-on-chip technologies allow patient-specific analyses, which can be used towards a fundamental understanding of disease progression," Varghese says. It's possible that doctors could one day take individual cell samples and create personalized treatments, testing out any medications on the chip for safety, efficacy, and potential side effects before writing a prescription.
2. Happening Soon: Prime Editing Will Have the Power to "Find and Replace" Disease-Causing Genes
Biochemist David Liu made industry-wide news last fall when he and his lab at MIT's Broad Institute, led by Andrew Anzalone, published a paper on prime editing: a new, more focused technology for editing genes. Prime editing is a descendant of the CRISPR-Cas9 system that researchers have been working with for years, and a cousin to Liu's previous innovation—base editing, which can make a limited number of changes to a single DNA letter at a time.
By contrast, prime editing has the potential to make much larger insertions and deletions; it also doesn't require the tweaked cells to divide in order to write the changes into the DNA, which could make it especially suitable for central nervous system diseases, like Parkinson's.
Crucially, the prime editing technique has a much higher efficiency rate than the older CRISPR system, and a much lower incidence of accidental insertions or deletions, which can make dangerous changes for a patient.
It also has a very broad potential range: according to Liu, 89% of the pathogenic mutations that have been collected in ClinVar (a public archive of human variations) could, in principle, be treated with prime editing—although he is careful to note that correcting a single genetic mutation may not be sufficient to fully treat a genetic disease.
Figuring out just how prime editing can be used most effectively and safely will be a long process, but it's already underway. The same day that Liu and his team posted their paper, they also made the basic prime editing constructs available for researchers around the world through Addgene, a plasmid repository, so that others in the scientific community can test out the technique for themselves. It might be years before human patients will see the results, and in the meantime, significant bioethical questions remain about the limits and sociological effects of such a powerful gene-editing tool. But in the long fight against genetic diseases, it's a huge step forward.
3. Happening When We Fund It: Focusing on Microbiome Health Could Help Us Tackle Social Inequality—And Vice Versa
The past decade has seen a growing awareness of the major role that the microbiome, the microbes present in our digestive tract, play in human health. Having a less-healthy microbiome is correlated with health risks like diabetes and depression, and interventions that target gut health, ranging from kombucha to fecal transplants, have cropped up with increasing frequency.
New research from the University of Maine's Dr. Suzanne Ishaq takes an even broader view, arguing that low-income and disadvantaged populations are less likely to have healthy, diverse gut bacteria, and that increasing access to beneficial microorganisms is an important juncture of social justice and public health.
"Basically, allowing people to lead healthy lives allows them to access and recruit microbes."
"Typically, having a more diverse bacterial community is associated with health, and having fewer different species is associated with illness and may leave you open to infection from bacteria that are good at exploiting opportunities," Ishaq says.
Having a healthy biome doesn't mean meeting one fixed ratio of gut bacteria, since different combinations of microbes can generate roughly similar results when they work in concert. Generally, "good" microbes are the ones that break down fiber and create the byproducts that we use for energy, or ones like lactic acid bacteria that work to make microbials and keep other bacteria in check. The microbial universe in your gut is chaotic, Ishaq says. "Microbes in your gut interact with each other, with you, with your food, or maybe they don't interact at all and pass right through you." Overall, it's tricky to name specific microbial communities that will make or break someone's health.
There are important corollaries between environment and biome health, though, which Ishaq points out: Living in urban environments reduces microbial exposure, and losing the microorganisms that humans typically source from soil and plants can reduce our adaptive immunity and ability to fight off conditions like allergies and asthma. Access to green space within cities can counteract those effects, but in the U.S. that access varies along income, education, and racial lines. Likewise, lower-income communities are more likely to live in food deserts or areas where the cheapest, most convenient food options are monotonous and low in fiber, further reducing microbial diversity.
Ishaq also suggests other areas that would benefit from further study, like the correlation between paid family leave, breastfeeding, and gut microbiota. There are technical and ethical challenges to direct experimentation with human populations—but that's not what Ishaq sees as the main impediment to future research.
"The biggest roadblock is money, and the solution is also money," she says. "Basically, allowing people to lead healthy lives allows them to access and recruit microbes."
That means investment in things we already understand to improve public health, like better education and healthcare, green space, and nutritious food. It also means funding ambitious, interdisciplinary research that will investigate the connections between urban infrastructure, housing policy, social equity, and the millions of microbes keeping us company day in and day out.
Scientists Just Started Testing a New Class of Drugs to Slow--and Even Reverse--Aging
Eliminating "zombie-like" cells, called senescent cells, may hold the key to slowing aging and its chronic diseases.
Imagine reversing the processes of aging. It's an age-old quest, and now a study from the Mayo Clinic may be the first ray of light in the dawn of that new era.
The immune system can handle a certain amount of senescence, but that capacity declines with age.
The small preliminary report, just nine patients, primarily looked at the safety and tolerability of the compounds used. But it also showed that a new class of small molecules called senolytics, which has proven to reverse markers of aging in animal studies, can work in humans.
Aging is a relentless assault of chronic diseases including Alzheimer's, cardiovascular disease, diabetes, and frailty. Developing one chronic condition strongly predicts the rapid onset of another. They pile on top of each other and impede the body's ability to respond to the next challenge.
"Potentially, by targeting fundamental aging processes, it may be possible to delay or prevent or alleviate multiple age-related conditions and many diseases as a group, instead of one at a time," says James Kirkland, the Mayo Clinic physician who led the study and is a top researcher in the growing field of geroscience, the biology of aging.
Getting Rid of "Zombie" Cells
One element common to many of the diseases is senescence, a kind of limbo or zombie-like state where cells no longer divide or perform many regular functions, but they don't die. Senescence is thought to be beneficial in that it inhibits the cancerous proliferation of cells. But in aging, the senescent cells still produce molecules that create inflammation both locally and throughout the body. It is a cycle that feeds upon itself, slowly ratcheting down normal body function and health.
Disease and harmful stimuli like radiation to treat cancer can also generate senescence, which is why young cancer patients seem to experience earlier and more rapid aging. The immune system can handle a certain amount of senescence, but that capacity declines with age. There also appears to be a threshold effect, a tipping point where senescence becomes a dominant factor in aging.
Kirkland's team used an artificial intelligence approach called machine learning to look for cell signaling networks that keep senescent cells from dying. To date, researchers have identified at least eight such signaling networks, some of which seem to be unique to a particular type of cell or tissue, but others are shared or overlap.
Then a computer search identified molecules known to disrupt these signaling pathways "and allow cells that are fully senescent to kill themselves," he explains. The process is a bit like looking for the right weapons in a video game to wipe out lingering zombie cells. But instead of swords, guns, and grenades, the list of biological tools so far includes experimental molecules, approved drugs, and natural supplements.
Treatment
"We found early on that targeting single components of those networks will only kill a very small minority of senescent cells or senescent cell types," says Kirkland. "So instead of going after one drug-one target-one disease, we're going after networks with combinations of drugs or drugs that have multiple targets. And we're going after every age-related disease."
The FDA is grappling with guidance for researchers wanting to conduct clinical trials on something as broad as aging rather than a single disease.
The large number of potential senolytic (i.e. zombie-neutralizing) compounds they identified allowed Kirkland to be choosy, "purposefully selecting drugs where the side effects profile was good...and with short elimination half-lives." The hit and run approach meant they didn't have to worry about maintaining a steady state of drugs in the body for an extended period of time. Some of the compounds they selected need only a half hour exposure to trigger the dying process in senescent cells, which can then take several days.
Work in mice has already shown impressive results in reversing diabetes, weight gain, Alzheimer's, cardiovascular disease and other conditions using senolytic agents.
That led to Kirkland's pilot study in humans with diabetes-related kidney disease using a three-day regimen of dasatinib, a kinase inhibitor first approved in 2006 to treat some forms of blood cancer, and quercetin, a flavonoid found in many plants and sold as a food supplement.
The combination was safe and well tolerated; it reduced the number of senescent cells in the belly fat of patients and restored their normal function, according to results published in September in the journal EBioMedicine. This preliminary paper was based on 9 patients in an ongoing study of 30 patients.
Kirkland cautions that these are initial and incomplete findings looking primarily at safety issues, not effectiveness. There is still much to be learned about the use of senolytics, starting with proof that they actually provide clinical benefit, and against what chronic conditions. The drug combinations, doses, duration, and frequency, not to mention potential risks all must be worked out. Additional studies of other diseases are being developed.
What's Next
Ron Kohanski, a senior administrator at the NIH National Institute on Aging (NIA), says the field of senolytics is so new that there isn't even a consensus on how to identify a senescent cell, and the FDA is grappling with guidance for researchers wanting to conduct clinical trials on something as broad as aging rather than a single disease.
Intellectual property concerns may temper the pharmaceutical industry's interest in developing senolytics to treat chronic diseases of aging. It looks like many mix-and-match combinations are possible, and many of the potential molecules identified so far are found in nature or are drugs whose patents have or will soon expire. So the ability to set high prices for such future drugs, and hence the willingness to spend money on expensive clinical trials, may be limited.
Still, Kohanski believes the field can move forward quickly because it often will include products that are already widely used and have a known safety profile. And approaches like Kirkland's hit and run strategy will minimize potential exposure and risk.
He says the NIA is going to support a number of clinical trials using these new approaches. Pharmaceutical companies may feel that they can develop a unique part of a senolytic combination regimen that will justify their investment. And if they don't, countries with socialized medicine may take the lead in supporting such research with the goal of reducing the costs of treating aging patients.

