Scientists redesign bacteria to tackle the antibiotic resistance crisis
Probiotic bacteria can be engineered to fight antibiotic-resistant superbugs by releasing chemicals that kill them.
In 1945, almost two decades after Alexander Fleming discovered penicillin, he warned that as antibiotics use grows, they may lose their efficiency. He was prescient—the first case of penicillin resistance was reported two years later. Back then, not many people paid attention to Fleming’s warning. After all, the “golden era” of the antibiotics age had just began. By the 1950s, three new antibiotics derived from soil bacteria — streptomycin, chloramphenicol, and tetracycline — could cure infectious diseases like tuberculosis, cholera, meningitis and typhoid fever, among others.
Today, these antibiotics and many of their successors developed through the 1980s are gradually losing their effectiveness. The extensive overuse and misuse of antibiotics led to the rise of drug resistance. The livestock sector buys around 80 percent of all antibiotics sold in the U.S. every year. Farmers feed cows and chickens low doses of antibiotics to prevent infections and fatten up the animals, which eventually causes resistant bacterial strains to evolve. If manure from cattle is used on fields, the soil and vegetables can get contaminated with antibiotic-resistant bacteria. Another major factor is doctors overprescribing antibiotics to humans, particularly in low-income countries. Between 2000 to 2018, the global rates of human antibiotic consumption shot up by 46 percent.
In recent years, researchers have been exploring a promising avenue: the use of synthetic biology to engineer new bacteria that may work better than antibiotics. The need continues to grow, as a Lancet study linked antibiotic resistance to over 1.27 million deaths worldwide in 2019, surpassing HIV/AIDS and malaria. The western sub-Saharan Africa region had the highest death rate (27.3 people per 100,000).
Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
To make it worse, our remedy pipelines are drying up. Out of the 18 biggest pharmaceutical companies, 15 abandoned antibiotic development by 2013. According to the AMR Action Fund, venture capital has remained indifferent towards biotech start-ups developing new antibiotics. In 2019, at least two antibiotic start-ups filed for bankruptcy. As of December 2020, there were 43 new antibiotics in clinical development. But because they are based on previously known molecules, scientists say they are inadequate for treating multidrug-resistant bacteria. Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
The rise of synthetic biology
To circumvent this dire future, scientists have been working on alternative solutions using synthetic biology tools, meaning genetically modifying good bacteria to fight the bad ones.
From the time life evolved on earth around 3.8 billion years ago, bacteria have engaged in biological warfare. They constantly strategize new methods to combat each other by synthesizing toxic proteins that kill competition.
For example, Escherichia coli produces bacteriocins or toxins to kill other strains of E.coli that attempt to colonize the same habitat. Microbes like E.coli (which are not all pathogenic) are also naturally present in the human microbiome. The human microbiome harbors up to 100 trillion symbiotic microbial cells. The majority of them are beneficial organisms residing in the gut at different compositions.
The chemicals that these “good bacteria” produce do not pose any health risks to us, but can be toxic to other bacteria, particularly to human pathogens. For the last three decades, scientists have been manipulating bacteria’s biological warfare tactics to our collective advantage.
In the late 1990s, researchers drew inspiration from electrical and computing engineering principles that involve constructing digital circuits to control devices. In certain ways, every cell in living organisms works like a tiny computer. The cell receives messages in the form of biochemical molecules that cling on to its surface. Those messages get processed within the cells through a series of complex molecular interactions.
Synthetic biologists can harness these living cells’ information processing skills and use them to construct genetic circuits that perform specific instructions—for example, secrete a toxin that kills pathogenic bacteria. “Any synthetic genetic circuit is merely a piece of information that hangs around in the bacteria’s cytoplasm,” explains José Rubén Morones-Ramírez, a professor at the Autonomous University of Nuevo León, Mexico. Then the ribosome, which synthesizes proteins in the cell, processes that new information, making the compounds scientists want bacteria to make. “The genetic circuit remains separated from the living cell’s DNA,” Morones-Ramírez explains. When the engineered bacteria replicates, the genetic circuit doesn’t become part of its genome.
Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut.
In 2000, Boston-based researchers constructed an E.coli with a genetic switch that toggled between turning genes on and off two. Later, they built some safety checks into their bacteria. “To prevent unintentional or deleterious consequences, in 2009, we built a safety switch in the engineered bacteria’s genetic circuit that gets triggered after it gets exposed to a pathogen," says James Collins, a professor of biological engineering at MIT and faculty member at Harvard University’s Wyss Institute. “After getting rid of the pathogen, the engineered bacteria is designed to switch off and leave the patient's body.”
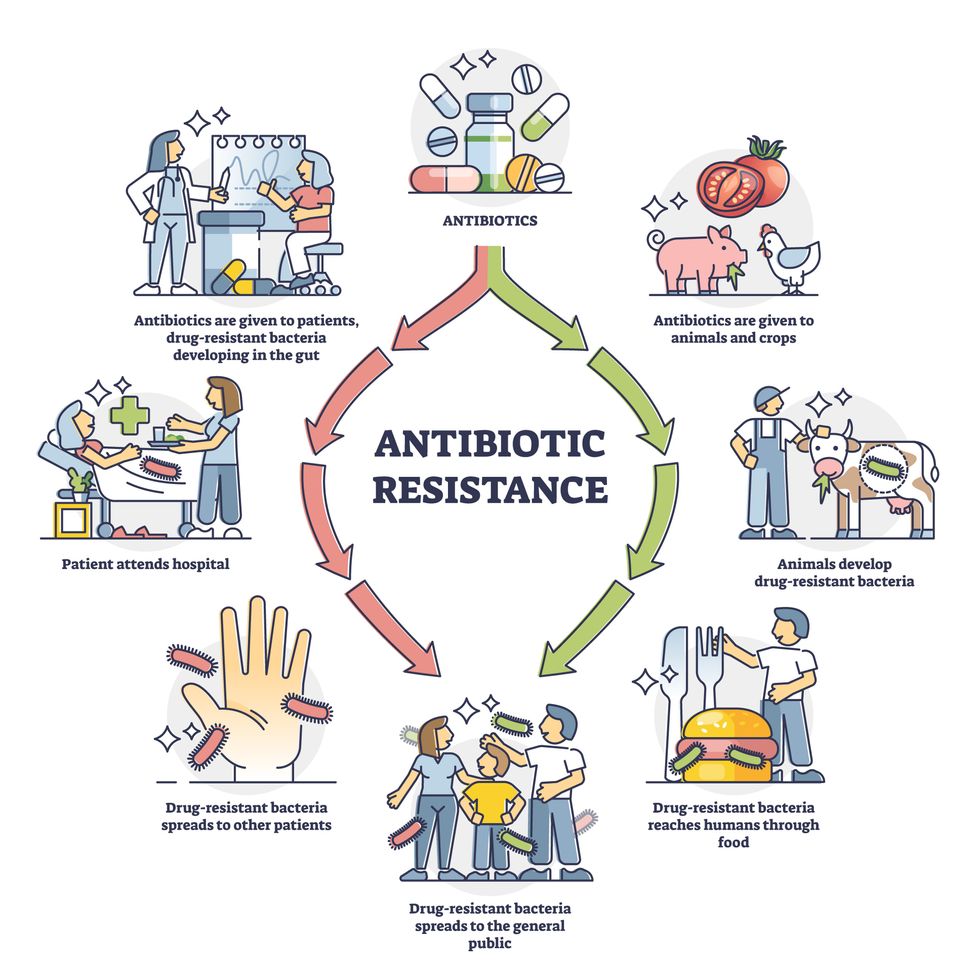
Overuse and misuse of antibiotics causes resistant strains to evolve
Adobe Stock
Seek and destroy
As the field of synthetic biology developed, scientists began using engineered bacteria to tackle superbugs. They first focused on Vibrio cholerae, which in the 19th and 20th century caused cholera pandemics in India, China, the Middle East, Europe, and Americas. Like many other bacteria, V. cholerae communicate with each other via quorum sensing, a process in which the microorganisms release different signaling molecules, to convey messages to its brethren. Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut. When untreated, cholera has a mortality rate of 25 to 50 percent and outbreaks frequently occur in developing countries, especially during floods and droughts.
Sometimes, however, V. cholerae makes mistakes. In 2008, researchers at Cornell University observed that when quorum sensing V. cholerae accidentally released high concentrations of a signaling molecule called CAI-1, it had a counterproductive effect—the pathogen couldn’t colonize the gut.
So the group, led by John March, professor of biological and environmental engineering, developed a novel strategy to combat V. cholerae. They genetically engineered E.coli to eavesdrop on V. cholerae communication networks and equipped it with the ability to release the CAI-1 molecules. That interfered with V. cholerae progress. Two years later, the Cornell team showed that V. cholerae-infected mice treated with engineered E.coli had a 92 percent survival rate.
These findings inspired researchers to sic the good bacteria present in foods like yogurt and kimchi onto the drug-resistant ones.
Three years later in 2011, Singapore-based scientists engineered E.coli to detect and destroy Pseudomonas aeruginosa, an often drug-resistant pathogen that causes pneumonia, urinary tract infections, and sepsis. Once the genetically engineered E.coli found its target through its quorum sensing molecules, it then released a peptide, that could eradicate 99 percent of P. aeruginosa cells in a test-tube experiment. The team outlined their work in a Molecular Systems Biology study.
“At the time, we knew that we were entering new, uncharted territory,” says lead author Matthew Chang, an associate professor and synthetic biologist at the National University of Singapore and lead author of the study. “To date, we are still in the process of trying to understand how long these microbes stay in our bodies and how they might continue to evolve.”
More teams followed the same path. In a 2013 study, MIT researchers also genetically engineered E.coli to detect P. aeruginosa via the pathogen’s quorum-sensing molecules. It then destroyed the pathogen by secreting a lab-made toxin.
Probiotics that fight
A year later in 2014, a Nature study found that the abundance of Ruminococcus obeum, a probiotic bacteria naturally occurring in the human microbiome, interrupts and reduces V.cholerae’s colonization— by detecting the pathogen’s quorum sensing molecules. The natural accumulation of R. obeum in Bangladeshi adults helped them recover from cholera despite living in an area with frequent outbreaks.
The findings from 2008 to 2014 inspired Collins and his team to delve into how good bacteria present in foods like yogurt and kimchi can attack drug-resistant bacteria. In 2018, Collins and his team developed the engineered probiotic strategy. They tweaked a bacteria commonly found in yogurt called Lactococcus lactis to treat cholera.
Engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut. --José Rubén Morones-Ramírez.
More scientists followed with more experiments. So far, researchers have engineered various probiotic organisms to fight pathogenic bacteria like Staphylococcus aureus (leading cause of skin, tissue, bone, joint and blood infections) and Clostridium perfringens (which causes watery diarrhea) in test-tube and animal experiments. In 2020, Russian scientists engineered a probiotic called Pichia pastoris to produce an enzyme called lysostaphin that eradicated S. aureus in vitro. Another 2020 study from China used an engineered probiotic bacteria Lactobacilli casei as a vaccine to prevent C. perfringens infection in rabbits.
In a study last year, Ramírez’s group at the Autonomous University of Nuevo León, engineered E. coli to detect quorum-sensing molecules from Methicillin-resistant Staphylococcus aureus or MRSA, a notorious superbug. The E. coli then releases a bacteriocin that kills MRSA. “An antibiotic is just a molecule that is not intelligent,” says Ramírez. “On the other hand, engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut.”
Collins and Timothy Lu, an associate professor of biological engineering at MIT, found that engineered E. coli can help treat other conditions—such as phenylketonuria, a rare metabolic disorder, that causes the build-up of an amino acid phenylalanine. Their start-up Synlogic aims to commercialize the technology, and has completed a phase 2 clinical trial.
Circumventing the challenges
The bacteria-engineering technique is not without pitfalls. One major challenge is that beneficial gut bacteria produce their own quorum-sensing molecules that can be similar to those that pathogens secrete. If an engineered bacteria’s biosensor is not specific enough, it will be ineffective.
Another concern is whether engineered bacteria might mutate after entering the gut. “As with any technology, there are risks where bad actors could have the capability to engineer a microbe to act quite nastily,” says Collins of MIT. But Collins and Ramírez both insist that the chances of the engineered bacteria mutating on its own are virtually non-existent. “It is extremely unlikely for the engineered bacteria to mutate,” Ramírez says. “Coaxing a living cell to do anything on command is immensely challenging. Usually, the greater risk is that the engineered bacteria entirely lose its functionality.”
However, the biggest challenge is bringing the curative bacteria to consumers. Pharmaceutical companies aren’t interested in antibiotics or their alternatives because it’s less profitable than developing new medicines for non-infectious diseases. Unlike the more chronic conditions like diabetes or cancer that require long-term medications, infectious diseases are usually treated much quicker. Running clinical trials are expensive and antibiotic-alternatives aren’t lucrative enough.
“Unfortunately, new medications for antibiotic resistant infections have been pushed to the bottom of the field,” says Lu of MIT. “It's not because the technology does not work. This is more of a market issue. Because clinical trials cost hundreds of millions of dollars, the only solution is that governments will need to fund them.” Lu stresses that societies must lobby to change how the modern healthcare industry works. “The whole world needs better treatments for antibiotic resistance.”
Patients voice hope and relief as FDA gives third-ever drug approval for ALS
On Sept. 29, the FDA approved Relyvrio, a new drug for ALS, even though a study of 137 ALS patients did not result in “substantial evidence” that Relyvrio was effective.
At age 52, Glen Rouse suffered from arm weakness and a lot of muscle twitches. “I first thought something was wrong when I could not throw a 50-pound bag of dog food over the tailgate of my truck—something I use to do effortlessly,” said the 54-year-old resident of Anderson, California, about three hours north of San Francisco.
In August, Rouse retired as a forester for a private timber company, a job he had held for 31 years. The impetus: amyotrophic lateral sclerosis, or ALS, a progressive neuromuscular disease that is commonly known as Lou Gehrig’s disease, named after the New York Yankees’ first baseman who succumbed to it less than a month shy of his 38th birthday in 1941. ALS eventually robs an individual of the ability to talk, walk, chew, swallow and breathe.
Rouse is now dependent on ventilation through a nasal mask and uses a powerchair to get around. “I can no longer walk or use my arms very well,” he said. “I can still move my wrists and fingers. I can also transfer from my chair to the toilet if I have two of my friends help me.”
It’s “shocking” that modern medicine has very little to offer to people with this devastating condition, Rouse said. But there is hope on the horizon. Yesterday, the U.S. Food and Drug Administration approved Relyvrio, a drug made up of two parts, sodium phenylbutyrate and taurursodiol, to treat patients with ALS.
“This approval provides another important treatment option for ALS, a life-threatening disease that currently has no cure,” said Billy Dunn, director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research, in a statement. “The FDA remains committed to facilitating the development of additional ALS treatments.”
Until this point, the FDA had approved only two other medications—Riluzole (rilutek) in 1995 and Radicava (edaravone) in 2017—to extend life in patients with ALS, which typically kills within two to five years after diagnosis. That’s why earlier this week, Rouse was optimistic about the FDA’s likely approval of a controversial new drug for ALS.
When Relyvrio is taken in addition to Riluzole, it appears to slow functional decline by an additional 25 percent and extend life by another 6 to 10 months, said Richard Bedlak, director of the Duke ALS Clinic. “It is not a cure, but it is definitely a step forward.”
“The whole ALS community is extremely excited about it,” he said the day before Relyvrio’s expected approval. “We are very hopeful. We’re on pins and needles.”
A study of 137 ALS patients did not result in “substantial evidence” that Relyvrio was effective, the agency’s Peripheral and Central Nervous System Drugs Advisory Committee concluded in March. However, after some persuasion from FDA officials, patients and their families, the committee met again and decided to recommend approving the drug.
In January 2019, following an ALS diagnosis at age 58 in October the previous year, Jeff Sarnacki, of Chester, Maryland, was accepted into a trial for Relyvrio. “Because of the trial, we did experience hope and a greater sense of help than had we not had that opportunity,” said Juliet Taylor, his wife and caregiver. They both believed the drug “worked for him in giving him more time.”
In June 2019, Sarnacki chose an open-label extension, offered to patients by drug researchers after a study ends, and took the active drug until he died peacefully at home under hospice care in May 2020, five days after his 60th birthday. A retired agent with the federal Bureau of Alcohol, Tobacco, Firearms and Explosives who later worked as a security consultant, Sarnacki lived about 19 months after diagnosis, which is shorter than the typical prognosis.
His symptoms began with leg cramps in fall 2017 and foot drop in early 2018. A feeding tube was placed in 2019, as it became necessary early in his illness, Taylor said. He also took Radicava and Riluzole, the two previously approved drugs, for his ALS. “We were both incredulous that, so many years after Lou Gehrig’s own diagnosis, there were so few treatments available,” she said.
The dearth of successful treatments for ALS is “certainly not for lack of trying,” said Karen Raley Steffens, a registered nurse and ALS support services coordinator at the Les Turner ALS Foundation in Skokie, Ill. “There are thousands of researchers and scientists all over the world working tirelessly to try to develop treatments for ALS.”
Unfortunately, she added, research takes time and exorbitant amounts of funding, while bureaucratic challenges persist. The rare disease also manifests and progresses in many different ways, so many treatments are needed.
As of 2017, the Centers for Disease Control and Prevention estimated that more than 31,000 people in the U.S. live with ALS, and an average of 5,000 people are newly diagnosed every year. It is slightly more common in men than women. Most people are diagnosed between the ages of 55 and 75.
Most cases of ALS are sporadic, meaning that doctors don’t know the cause. There is about a one-year interval between symptom onset and an ALS diagnosis for most patients, so many motor neurons are lost by the time individuals can enroll in a clinical trial, said Richard Bedlack, professor of neurology and director of the Duke ALS Clinic in Durham, North Carolina.
Bedlack found the new drug, Relyvrio, to be “very promising,” which is why he testified to the FDA in favor of approval. (He’s a consultant and disease state speaker for multiple companies including Amylyx, manufacturer of Relyvrio.)
The “drug has different mechanisms of action than the currently approved treatments,” Bedlack said. He added that, when Relyvrio is taken in addition to Riluzole, it appears to slow functional decline by an additional 25 percent and extend life by another 6 to 10 months. “It is not a cure, but it is definitely a step forward.”
T. Scott Diesing, a neurohospitalist and director of general neurology at the University of Nebraska Medical Center in Omaha, said he hopes the drug is “as good as people anticipated it should be, because there are not too many options for these patients.”
"FDA went out on a limb in approving Relyvrio based on limited results from a small trial while a larger study remains in progress," said Florian P. Thomas, co-director of the ALS Center at Hackensack University Medical Center and Hackensack Meridian School of Medicine in New Jersey. "While it is definitely promising, clearly, the last word on this drug has not been spoken."
So far, Rouse's voice is holding up, but he knows the day will come when ALS will steal that and much more from him.
ALS is 100 percent fatal, with some patients dying as soon as a year after diagnosis. A few have lasted as long as 15 years, but those are the exceptions, Diesing said.
“If this drug can provide even months of additional life, or would maintain quality of life, that’s a big deal,” he noted, adding that “the patients are saying, ‘I know it’s not proven conclusively, but what do we have to lose?’ So, they would like to try it while additional studies are ongoing.” The drug has already been conditionally approved in Canada.
As his disease progresses, Rouse hopes to get a speech-to-text voice-generating computer that he can control with his eyes. So far, his voice is holding up, but he knows the day will come when ALS will steal that and much more from him. He works at I AM ALS, a patient-led community, and six of his friends have already died of the disease.
“Every time I lose a friend to ALS, I grieve and am sad but I resolve myself to keep working harder for them, myself and others,” Rouse said. “People living with ALS find great purpose in life advocating and trying to make a difference.”
Friday Five Podcast: New drug may slow the rate of Alzheimer's disease
On September 27, pharmaceuticals Biogen and Eisai announced that their drug, lecanemab, can slow the rate of Alzheimer's disease, according to a clinical trial. Today's Friday Five episode covers this story and other health research over the month of September.
The Friday Five covers important stories in health and science research that you may have missed - usually over the previous week, but today's episode is a lookback on important studies over the month of September.
Most recently, on September 27, pharmaceuticals Biogen and Eisai announced that a clinical trial showed their drug, lecanemab, can slow the rate of Alzheimer's disease. There are plenty of controversies and troubling ethical issues in science – and we get into many of them in our online magazine – but this news roundup focuses on scientific creativity and progress to give you a therapeutic dose of inspiration headed into the weekend and the new month.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
This Friday Five episode covers the following studies published and announced over the past month:
- A new drug is shown to slow the rate of Alzheimer's disease
- The need for speed if you want to reduce your risk of dementia
- How to refreeze the north and south poles
- Ancient wisdom about Neti pots could pay off for Covid
- Two women, one man and a baby