Life is Emerging: Review of Siddhartha Mukherjee’s Song of the Cell
A new book by Pulitzer-winning physician-scientist Siddhartha Mukherjee will be released from Simon & Schuster on October 25, 2022.
The DNA double helix is often the image spiraling at the center of 21st century advances in biomedicine and the growing bioeconomy. And yet, DNA is molecularly inert. DNA, the code for genes, is not alive and is not strictly necessary for life. Ought life be at the center of our communication of living systems? Is not the Cell a superior symbol of life and our manipulation of living systems?
A code for life isn’t a code without the life that instantiates it. A code for life must be translated. The cell is the basic unit of that translation. The cell is the minimal viable package of life as we know it. Therefore, cell biology is at the center of biomedicine’s greatest transformations, suggests Pulitzer-winning physician-scientist Siddhartha Mukherjee in his latest book, The Song of the Cell: The Exploration of Medicine and the New Human.
The Song of the Cell begins with the discovery of cells and of germ theory, featuring characters such as Louis Pasteur and Robert Koch, who brought the cell “into intimate contact with pathology and medicine.” This intercourse would transform biomedicine, leading to the insight that we can treat disease by thinking at the cellular level. The slightest rearrangement of sick cells might be the path toward alleviating suffering for the organism: eroding the cell walls of a bacterium while sparing our human cells; inventing a medium that coaxes sperm and egg to dance into cellular union for in vitro fertilization (IVF); designing molecular missiles that home to the receptors decorating the exterior of cancer cells; teaching adult skin cells to remember their embryonic state for regenerative medicines.
Mukherjee uses the bulk of the book to elucidate key cell types in the human body, along with their “connective relationships” that enable key organs and organ systems to function. This includes the immune system, the heart, the brain, and so on. Mukherjee’s distinctive style features compelling anecdotes and human stories that animate the scientific (and unscientific) processes that have led to our current state of understanding. In his chapter on neurons and the brain, for example, he integrates Santiago Ramon y Cajal’s meticulous black ink sketches of neurons into Mukherjee’s own personal encounter with clinical depression. In one lucid section, he interviews Dr. Helen Mayberg, a pioneering neurologist who takes seriously the descriptive power of her patients’ metaphors, as they suffer from “caves,” “holes,” “voids,” and “force fields” that render their lives gray. Dr. Mayberg aims to stimulate patients’ neuronal cells in a manner that brings back the color.

Beyond exposing the insight and inventiveness that has arisen out of cell-based thinking, it seems that Mukherjee’s bigger project is an epistemological one. The early chapters of The Song of the Cell continually hint at the potential for redefining the basic unit of biology as the cell rather than the gene. The choice to center biomedicine around cells is, above all, a conspicuous choice not to center it around genes (the subject of Mukherjee’s previous book, The Gene), because genes dominate popular science communication.
This choice of cells over genes is most welcome. Cells are alive. Genes are not. Letters—such as the As, Cs, Gs, and Ts that represent the nucleotides of DNA, which make up our genes—must be synthesized into a word or poem or song that offers a glimpse into deeper truths. A key idea embedded in this thinking is that of emergence. Whether in ancient myth or modern art, creation tends to be an emergent process, not a linearly coded script. The cell is our current best guess for the basic unit of life’s emergence, turning a finite set of chemical building blocks—nucleic acids, proteins, sugars, fats—into a replicative, evolving system for fighting stasis and entropy. The cell’s song is one for our times, for it is the song of biology’s emergence out of chemistry and physics, into the “frenetically active process” of homeostasis.
Re-centering our view of biology has practical consequences, too, for how we think about diagnosing and treating disease, and for inventing new medicines. Centering cells presents a challenge: which type of cell to place at the center? Rather than default to the apparent simplicity of DNA as a symbol because it represents the one master code for life, the tension in defining the diversity of cells—a mapping process still far from complete in cutting-edge biology laboratories—can help to create a more thoughtful library of cellular metaphors to shape both the practice and communication of biology.
Further, effective problem solving is often about operating at the right level, or the right scale. The cell feels like appropriate level at which to interrogate many of the diseases that ail us, because the senses that guide our own perceptions of sickness and health—the smoldering pain of inflammation, the tunnel vision of a migraine, the dizziness of a fluttering heart—are emergent.
This, unfortunately, is sort of where Mukherjee leaves the reader, under-exploring the consequences of a biology of emergence. Many practical and profound questions have to do with the ways that each scale of life feeds back on the others. In a tome on Cells and “the future human” I wished that Mukherjee had created more space for seeking the ways that cells will shape and be shaped by the future, of humanity and otherwise.
We are entering a phase of real-world bioengineering that features the modularization of cellular parts within cells, of cells within organs, of organs within bodies, and of bodies within ecosystems. In this reality, we would be unwise to assume that any whole is the mere sum of its parts.
For example, when discussing the regenerative power of pluripotent stem cells, Mukherjee raises the philosophical thought experiment of the Delphic boat, also known as the Ship of Theseus. The boat is made of many pieces of wood, each of which is replaced for repairs over the years, with the boat’s structure unchanged. Eventually none of the boat’s original wood remains: Is it the same boat?
Mukherjee raises the Delphic boat in one paragraph at the end of the chapter on stem cells, as a metaphor related to the possibility of stem cell-enabled regeneration in perpetuity. He does not follow any of the threads of potential answers. Given the current state of cellular engineering, about which Mukherjee is a world expert from his work as a physician-scientist, this book could have used an entire section dedicated to probing this question and, importantly, the ways this thought experiment falls apart.
We are entering a phase of real-world bioengineering that features the modularization of cellular parts within cells, of cells within organs, of organs within bodies, and of bodies within ecosystems. In this reality, we would be unwise to assume that any whole is the mere sum of its parts. Wholeness at any one of these scales of life—organelle, cell, organ, body, ecosystem—is what is at stake if we allow biological reductionism to assume away the relation between those scales.
In other words, Mukherjee succeeds in providing a masterful and compelling narrative of the lives of many of the cells that emerge to enliven us. Like his previous books, it is a worthwhile read for anyone curious about the role of cells in disease and in health. And yet, he fails to offer the broader context of The Song of the Cell.
As leading agronomist and essayist Wes Jackson has written, “The sequence of amino acids that is at home in the human cell, when produced inside the bacterial cell, does not fold quite right. Something about the E. coli internal environment affects the tertiary structure of the protein and makes it inactive. The whole in this case, the E. coli cell, affects the part—the newly made protein. Where is the priority of part now?” [1]
Beyond the ways that different kingdoms of life translate the same genetic code, the practical situation for humanity today relates to the ways that the different disciplines of modern life use values and culture to influence our genes, cells, bodies, and environment. It may be that humans will soon become a bit like the Delphic boat, infused with the buzz of fresh cells to repopulate different niches within our bodies, for healthier, longer lives. But in biology, as in writing, a mixed metaphor can cause something of a cacophony. For we are not boats with parts to be replaced piecemeal. And nor are whales, nor alpine forests, nor topsoil. Life isn’t a sum of parts, and neither is a song that rings true.
[1] Wes Jackson, "Visions and Assumptions," in Nature as Measure (p. 52-53).
A movie still from the 1966 film "Fantastic Voyage"
In the 1966 movie "Fantastic Voyage," actress Raquel Welch and her submarine were shrunk to the size of a cell in order to eliminate a blood clot in a scientist's brain. Now, 55 years later, the scenario is becoming closer to reality.
California-based startup Bionaut Labs has developed a nanobot about the size of a grain of rice that's designed to transport medication to the exact location in the body where it's needed. If you think about it, the conventional way to deliver medicine makes little sense: A painkiller affects the entire body instead of just the arm that's hurting, and chemotherapy is flushed through all the veins instead of precisely targeting the tumor.
"Chemotherapy is delivered systemically," Bionaut-founder and CEO Michael Shpigelmacher says. "Often only a small percentage arrives at the location where it is actually needed."
But what if it was possible to send a tiny robot through the body to attack a tumor or deliver a drug at exactly the right location?
Several startups and academic institutes worldwide are working to develop such a solution but Bionaut Labs seems the furthest along in advancing its invention. "You can think of the Bionaut as a tiny screw that moves through the veins as if steered by an invisible screwdriver until it arrives at the tumor," Shpigelmacher explains. Via Zoom, he shares the screen of an X-ray machine in his Culver City lab to demonstrate how the half-transparent, yellowish device winds its way along the spine in the body. The nanobot contains a tiny but powerful magnet. The "invisible screwdriver" is an external magnetic field that rotates that magnet inside the device and gets it to move and change directions.
The current model has a diameter of less than a millimeter. Shpigelmacher's engineers could build the miniature vehicle even smaller but the current size has the advantage of being big enough to see with bare eyes. It can also deliver more medicine than a tinier version. In the Zoom demonstration, the micorobot is injected into the spine, not unlike an epidural, and pulled along the spine through an outside magnet until the Bionaut reaches the brainstem. Depending which organ it needs to reach, it could be inserted elsewhere, for instance through a catheter.
"The hope is that we can develop a vehicle to transport medication deep into the body," says Max Planck scientist Tian Qiu.
Imagine moving a screw through a steak with a magnet — that's essentially how the device works. But of course, the Bionaut is considerably different from an ordinary screw: "At the right location, we give a magnetic signal, and it unloads its medicine package," Shpigelmacher says.
To start, Bionaut Labs wants to use its device to treat Parkinson's disease and brain stem gliomas, a type of cancer that largely affects children and teenagers. About 300 to 400 young people a year are diagnosed with this type of tumor. Radiation and brain surgery risk damaging sensitive brain tissue, and chemotherapy often doesn't work. Most children with these tumors live less than 18 months. A nanobot delivering targeted chemotherapy could be a gamechanger. "These patients really don't have any other hope," Shpigelmacher says.
Of course, the main challenge of the developing such a device is guaranteeing that it's safe. Because tissue is so sensitive, any mistake could risk disastrous results. In recent years, Bionaut has tested its technology in dozens of healthy sheep and pigs with no major adverse effects. Sheep make a good stand-in for humans because their brains and spines are similar to ours.

The Bionaut device is about the size of a grain of rice.
Bionaut Labs
"As the Bionaut moves through brain tissue, it creates a transient track that heals within a few weeks," Shpigelmacher says. The company is hoping to be the first to test a nanobot in humans. In December 2022, it announced that a recent round of funding drew $43.2 million, for a total of 63.2 million, enabling more research and, if all goes smoothly, human clinical trials by early next year.
Once the technique has been perfected, further applications could include addressing other kinds of brain disorders that are considered incurable now, such as Alzheimer's or Huntington's disease. "Microrobots could serve as a bridgehead, opening the gateway to the brain and facilitating precise access of deep brain structure – either to deliver medication, take cell samples or stimulate specific brain regions," Shpigelmacher says.
Robot-assisted hybrid surgery with artificial intelligence is already used in state-of-the-art surgery centers, and many medical experts believe that nanorobotics will be the instrument of the future. In 2016, three scientists were awarded the Nobel Prize in Chemistry for their development of "the world's smallest machines," nano "elevators" and minuscule motors. Since then, the scientific experiments have progressed to the point where applicable devices are moving closer to actually being implemented.
Bionaut's technology was initially developed by a research team lead by Peer Fischer, head of the independent Micro Nano and Molecular Systems Lab at the Max Planck Institute for Intelligent Systems in Stuttgart, Germany. Fischer is considered a pioneer in the research of nano systems, which he began at Harvard University more than a decade ago. He and his team are advising Bionaut Labs and have licensed their technology to the company.
"The hope is that we can develop a vehicle to transport medication deep into the body," says Max Planck scientist Tian Qiu, who leads the cooperation with Bionaut Labs. He agrees with Shpigelmacher that the Bionaut's size is perfect for transporting medication loads and is researching potential applications for even smaller nanorobots, especially in the eye, where the tissue is extremely sensitive. "Nanorobots can sneak through very fine tissue without causing damage."
In "Fantastic Voyage," Raquel Welch's adventures inside the body of a dissident scientist let her swim through his veins into his brain, but her shrunken miniature submarine is attacked by antibodies; she has to flee through the nerves into the scientist's eye where she escapes into freedom on a tear drop. In reality, the exit in the lab is much more mundane. The Bionaut simply leaves the body through the same port where it entered. But apart from the dramatization, the "Fantastic Voyage" was almost prophetic, or, as Shpigelmacher says, "Science fiction becomes science reality."
This article was first published by Leaps.org on April 12, 2021.
How the Human Brain Project Built a Mind of its Own
In 2013, the Human Brain Project set out to build a realistic computer model of the brain over ten years. Now, experts are reflecting on HBP's achievements with an eye toward the future.
In 2009, neuroscientist Henry Markram gave an ambitious TED talk. “Our mission is to build a detailed, realistic computer model of the human brain,” he said, naming three reasons for this unmatched feat of engineering. One was because understanding the human brain was essential to get along in society. Another was because experimenting on animal brains could only get scientists so far in understanding the human ones. Third, medicines for mental disorders weren’t good enough. “There are two billion people on the planet that are affected by mental disorders, and the drugs that are used today are largely empirical,” Markram said. “I think that we can come up with very concrete solutions on how to treat disorders.”
Markram's arguments were very persuasive. In 2013, the European Commission launched the Human Brain Project, or HBP, as part of its Future and Emerging Technologies program. Viewed as Europe’s chance to try to win the “brain race” between the U.S., China, Japan, and other countries, the project received about a billion euros in funding with the goal to simulate the entire human brain on a supercomputer, or in silico, by 2023.
Now, after 10 years of dedicated neuroscience research, the HBP is coming to an end. As its many critics warned, it did not manage to build an entire human brain in silico. Instead, it achieved a multifaceted array of different goals, some of them unexpected.
Scholars have found that the project did help advance neuroscience more than some detractors initially expected, specifically in the area of brain simulations and virtual models. Using an interdisciplinary approach of combining technology, such as AI and digital simulations, with neuroscience, the HBP worked to gain a deeper understanding of the human brain’s complicated structure and functions, which in some cases led to novel treatments for brain disorders. Lastly, through online platforms, the HBP spearheaded a previously unmatched level of global neuroscience collaborations.
Simulating a human brain stirs up controversy
Right from the start, the project was plagued with controversy and condemnation. One of its prominent critics was Yves Fregnac, a professor in cognitive science at the Polytechnic Institute of Paris and research director at the French National Centre for Scientific Research. Fregnac argued in numerous articles that the HBP was overfunded based on proposals with unrealistic goals. “This new way of over-selling scientific targets, deeply aligned with what modern society expects from mega-sciences in the broad sense (big investment, big return), has been observed on several occasions in different scientific sub-fields,” he wrote in one of his articles, “before invading the field of brain sciences and neuromarketing.”
"A human brain model can simulate an experiment a million times for many different conditions, but the actual human experiment can be performed only once or a few times," said Viktor Jirsa, a professor at Aix-Marseille University.
Responding to such critiques, the HBP worked to restructure the effort in its early days with new leadership, organization, and goals that were more flexible and attainable. “The HBP got a more versatile, pluralistic approach,” said Viktor Jirsa, a professor at Aix-Marseille University and one of the HBP lead scientists. He believes that these changes fixed at least some of HBP’s issues. “The project has been on a very productive and scientifically fruitful course since then.”
After restructuring, the HBP became a European hub on brain research, with hundreds of scientists joining its growing network. The HBP created projects focused on various brain topics, from consciousness to neurodegenerative diseases. HBP scientists worked on complex subjects, such as mapping out the brain, combining neuroscience and robotics, and experimenting with neuromorphic computing, a computational technique inspired by the human brain structure and function—to name just a few.
Simulations advance knowledge and treatment options
In 2013, it seemed that bringing neuroscience into a digital age would be farfetched, but research within the HBP has made this achievable. The virtual maps and simulations various HBP teams create through brain imaging data make it easier for neuroscientists to understand brain developments and functions. The teams publish these models on the HBP’s EBRAINS online platform—one of the first to offer access to such data to neuroscientists worldwide via an open-source online site. “This digital infrastructure is backed by high-performance computers, with large datasets and various computational tools,” said Lucy Xiaolu Wang, an assistant professor in the Resource Economics Department at the University of Massachusetts Amherst, who studies the economics of the HBP. That means it can be used in place of many different types of human experimentation.
Jirsa’s team is one of many within the project that works on virtual brain models and brain simulations. Compiling patient data, Jirsa and his team can create digital simulations of different brain activities—and repeat these experiments many times, which isn’t often possible in surgeries on real brains. “A human brain model can simulate an experiment a million times for many different conditions,” Jirsa explained, “but the actual human experiment can be performed only once or a few times.” Using simulations also saves scientists and doctors time and money when looking at ways to diagnose and treat patients with brain disorders.

Compiling patient data, scientists can create digital simulations of different brain activities—and repeat these experiments many times.
The Human Brain Project
Simulations can help scientists get a full picture that otherwise is unattainable. “Another benefit is data completion,” added Jirsa, “in which incomplete data can be complemented by the model. In clinical settings, we can often measure only certain brain areas, but when linked to the brain model, we can enlarge the range of accessible brain regions and make better diagnostic predictions.”
With time, Jirsa’s team was able to move into patient-specific simulations. “We advanced from generic brain models to the ability to use a specific patient’s brain data, from measurements like MRI and others, to create individualized predictive models and simulations,” Jirsa explained. He and his team are working on this personalization technique to treat patients with epilepsy. According to the World Health Organization, about 50 million people worldwide suffer from epilepsy, a disorder that causes recurring seizures. While some epilepsy causes are known others remain an enigma, and many are hard to treat. For some patients whose epilepsy doesn’t respond to medications, removing part of the brain where seizures occur may be the only option. Understanding where in the patients’ brains seizures arise can give scientists a better idea of how to treat them and whether to use surgery versus medications.
“We apply such personalized models…to precisely identify where in a patient’s brain seizures emerge,” Jirsa explained. “This guides individual surgery decisions for patients for which surgery is the only treatment option.” He credits the HBP for the opportunity to develop this novel approach. “The personalization of our epilepsy models was only made possible by the Human Brain Project, in which all the necessary tools have been developed. Without the HBP, the technology would not be in clinical trials today.”
Personalized simulations can significantly advance treatments, predict the outcome of specific medical procedures and optimize them before actually treating patients. Jirsa is watching this happen firsthand in his ongoing research. “Our technology for creating personalized brain models is now used in a large clinical trial for epilepsy, funded by the French state, where we collaborate with clinicians in hospitals,” he explained. “We have also founded a spinoff company called VB Tech (Virtual Brain Technologies) to commercialize our personalized brain model technology and make it available to all patients.”
The Human Brain Project created a level of interconnectedness within the neuroscience research community that never existed before—a network not unlike the brain’s own.
Other experts believe it’s too soon to tell whether brain simulations could change epilepsy treatments. “The life cycle of developing treatments applicable to patients often runs over a decade,” Wang stated. “It is still too early to draw a clear link between HBP’s various project areas with patient care.” However, she admits that some studies built on the HBP-collected knowledge are already showing promise. “Researchers have used neuroscientific atlases and computational tools to develop activity-specific stimulation programs that enabled paraplegic patients to move again in a small-size clinical trial,” Wang said. Another intriguing study looked at simulations of Alzheimer’s in the brain to understand how it evolves over time.
Some challenges remain hard to overcome even with computer simulations. “The major challenge has always been the parameter explosion, which means that many different model parameters can lead to the same result,” Jirsa explained. An example of this parameter explosion could be two different types of neurodegenerative conditions, such as Parkinson’s and Huntington’s diseases. Both afflict the same area of the brain, the basal ganglia, which can affect movement, but are caused by two different underlying mechanisms. “We face the same situation in the living brain, in which a large range of diverse mechanisms can produce the same behavior,” Jirsa said. The simulations still have to overcome the same challenge.
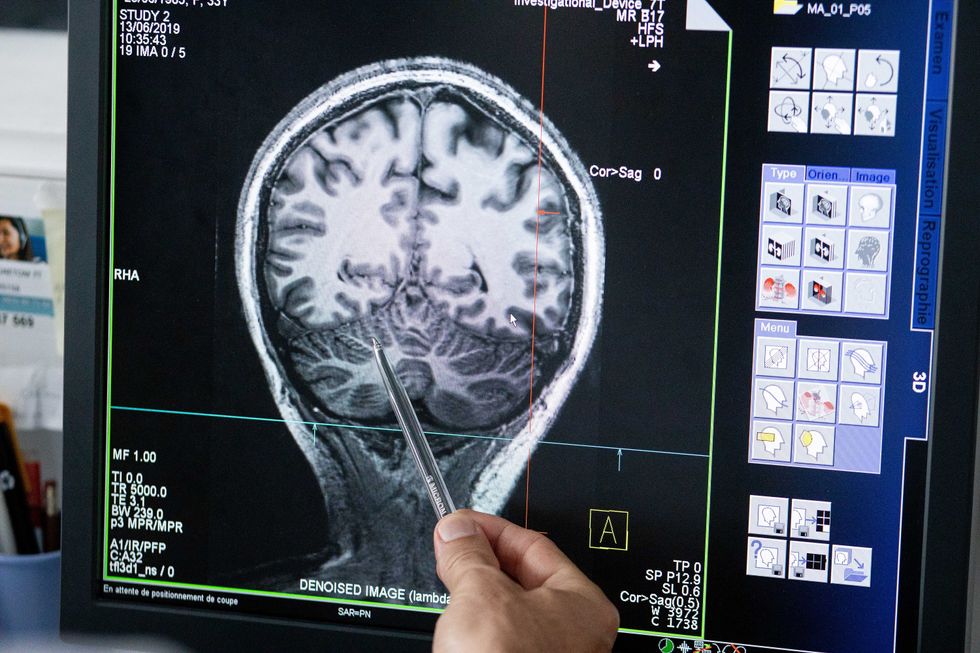
Understanding where in the patients’ brains seizures arise can give scientists a better idea of how to treat them and whether to use surgery versus medications.
The Human Brain Project
A network not unlike the brain’s own
Though the HBP will be closing this year, its legacy continues in various studies, spin-off companies, and its online platform, EBRAINS. “The HBP is one of the earliest brain initiatives in the world, and the 10-year long-term goal has united many researchers to collaborate on brain sciences with advanced computational tools,” Wang said. “Beyond the many research articles and projects collaborated on during the HBP, the online neuroscience research infrastructure EBRAINS will be left as a legacy even after the project ends.”
Those who worked within the HBP see the end of this project as the next step in neuroscience research. “Neuroscience has come closer to very meaningful applications through the systematic link with new digital technologies and collaborative work,” Jirsa stated. “In that way, the project really had a pioneering role.” It also created a level of interconnectedness within the neuroscience research community that never existed before—a network not unlike the brain’s own. “Interconnectedness is an important advance and prerequisite for progress,” Jirsa said. “The neuroscience community has in the past been rather fragmented and this has dramatically changed in recent years thanks to the Human Brain Project.”
According to its website, by 2023 HBP’s network counted over 500 scientists from over 123 institutions and 16 different countries, creating one of the largest multi-national research groups in the world. Even though the project hasn’t produced the in-silico brain as Markram envisioned it, the HBP created a communal mind with immense potential. “It has challenged us to think beyond the boundaries of our own laboratories,” Jirsa said, “and enabled us to go much further together than we could have ever conceived going by ourselves.”