How Excessive Regulation Helped Ignite COVID-19's Rampant Spread
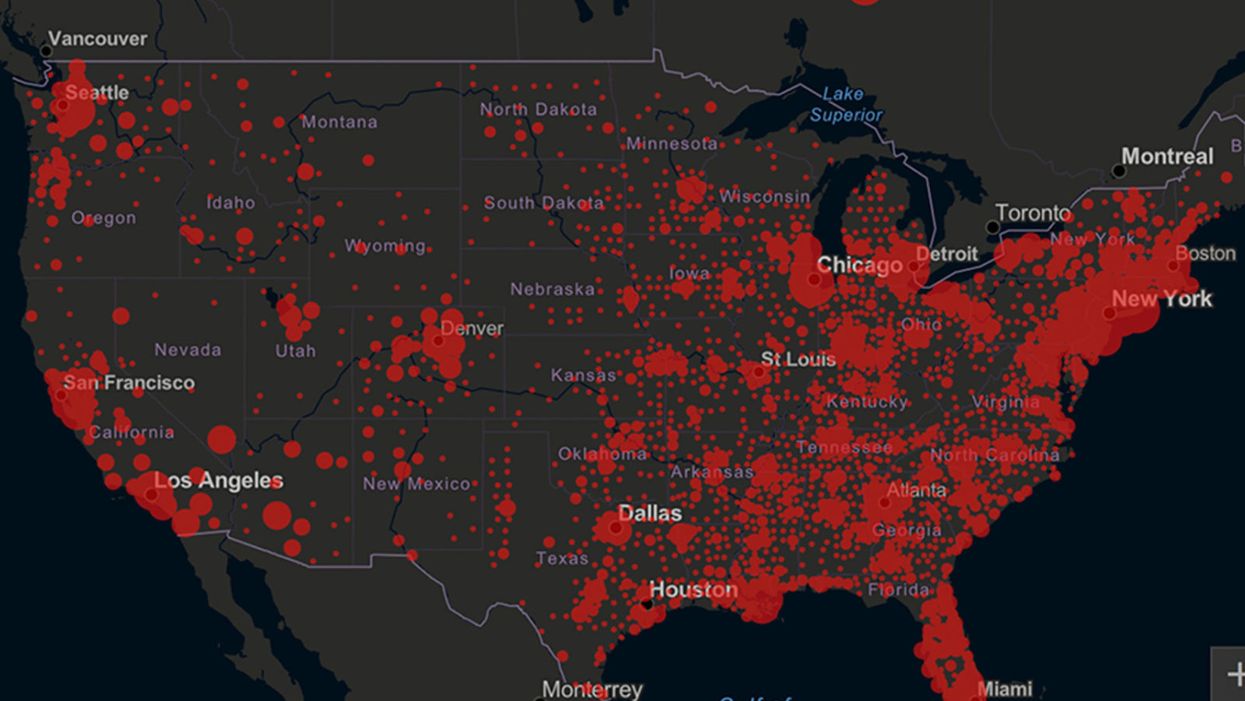
Screenshot of an interactive map of coronavirus cases across the United States, current as of 1:45 p.m. Pacific time on Tuesday, March 31st. Full map accessible at https://coronavirus.jhu.edu/map.html
When historians of the future look back at the 2020 pandemic, the heroic work of Helen Y. Chu, a flu researcher at the University of Washington, will be worthy of recognition.
Chu's team bravely defied the order and conducted the testing anyway.
In late January, Chu was testing nasal swabs for the Seattle Flu Study to monitor influenza spread when she learned of the first case of COVID-19 in Washington state. She deemed it a pressing public health matter to document if and how the illness was spreading locally, so that early containment efforts could succeed. So she sought regulatory approval to adapt the Flu Study to test for the coronavirus, but the federal government denied the request because the original project was funded to study only influenza.
Aware of the urgency, Chu's team bravely defied the order and conducted the testing anyway. Soon they identified a local case in a teenager without any travel history, followed by others. Still, the government tried to shutter their efforts until the outbreak grew dangerous enough to command attention.
Needless testing delays, prompted by excessive regulatory interference, eliminated any chances of curbing the pandemic at its initial stages. Even after Chu went out on a limb to sound alarms, a heavy-handed bureaucracy crushed the nation's ability to roll out early and widespread testing across the country. The Centers for Disease Control and Prevention infamously blundered its own test, while also impeding state and private labs from coming on board, fueling a massive shortage.
The long holdup created "a backlog of testing that needed to be done," says Amesh Adalja, an infectious disease specialist who is a senior scholar at the Johns Hopkins University Center for Health Security.
In a public health crisis, "the ideal situation" would allow the government's test to be "supplanted by private laboratories" without such "a lag in that transition," Adalja says. Only after the eventual release of CDC's test could private industry "begin in earnest" to develop its own versions under the Food and Drug Administration's emergency use authorization.
In a statement, CDC acknowledged that "this process has not gone as smoothly as we would have liked, but there is currently no backlog for testing at CDC."
Now, universities and corporations are in a race against time, playing catch up as the virus continues its relentless spread, also afflicting many health care workers on the front lines.
"Home-testing accessibility is key to preventing further spread of the COVID-19 pandemic."
Hospitals are attempting to add the novel coronavirus to the testing panel of their existent diagnostic machines, which would reduce the results processing time from 48 hours to as little as four hours. Meanwhile, at least four companies announced plans to deliver at-home collection tests to help meet the demand – before a startling injunction by the FDA halted their plans.
Everlywell, an Austin, Texas-based digital health company, had been set to launch online sales of at-home collection kits directly to consumers last week. Scaling up in a matter of days to an initial supply of 30,000 tests, Everlywell collaborated with multiple laboratories where consumers could ship their nasal swab samples overnight, projecting capacity to screen a quarter-million individuals on a weekly basis, says Frank Ong, chief medical and scientific officer.
Secure digital results would have been available online within 48 hours of a sample's arrival at the lab, as well as a telehealth consultation with an independent, board-certified doctor if someone tested positive, for an inclusive $135 cost. The test has a less than 3 percent false-negative rate, Ong says, and in the event of an inadequate self-swab, the lab would not report a conclusive finding. "Home-testing accessibility," he says, "is key to preventing further spread of the COVID-19 pandemic."
But on March 20, the FDA announced restrictions on home collection tests due to concerns about accuracy. The agency did note "the public health value in expanding the availability of COVID-19 testing through safe and accurate tests that may include home collection," while adding that "we are actively working with test developers in this space."
After the restrictions were announced, Everlywell decided to allocate its initial supply of COVID-19 collection kits to hospitals, clinics, nursing homes, and other qualifying health care companies that can commit to no-cost screening of frontline workers and high-risk symptomatic patients. For now, no consumers can order a home-collection test.
"Losing two months is close to disastrous, and that's what we did."
Currently, the U.S. has ramped up to testing an estimated 100,000 people a day, according to Stat News. But 150,000 or more Americans should be tested every day, says Ashish Jha, professor and director of the Harvard Global Health Institute. Due to the dearth of tests, many sick people who suspect they are infected still cannot get confirmation unless they need to be hospitalized.
To give a concrete sense of how far behind we are in testing, consider Palm Beach County, Fla. The state's only drive-thru test center just opened there, requiring an appointment. The center aims to test 750 people per day, but more than 330,000 people have already called to try to book a slot.
"This is such a rapidly moving infection that losing a few days is bad, and losing a couple of weeks is terrible," says Jha, a practicing general internist. "Losing two months is close to disastrous, and that's what we did."
At this point, it will take a long time to fully ramp up. "We are blindfolded," he adds, "and I'd like to take the blindfolds off so we can fight this battle with our eyes wide open."
Better late than never: Yesterday, FDA Commissioner Stephen Hahn said in a statement that the agency has worked with more than 230 test developers and has approved 20 tests since January. An especially notable one was authorized last Friday – 67 days since the country's first known case in Washington state. It's a rapid point-of-care test from medical-device firm Abbott that provides positive results in five minutes and negative results in 13 minutes. Abbott will send 50,000 tests a day to urgent care settings. The first tests are expected to ship tomorrow.
A company has slashed the cost of assessing a person's genome to just $100. With lower costs - and as other genetic tools mature and evolve - a wave of new therapies could be coming in the near future.
In May 2022, Californian biotech Ultima Genomics announced that its UG 100 platform was capable of sequencing an entire human genome for just $100, a landmark moment in the history of the field. The announcement was particularly remarkable because few had previously heard of the company, a relative unknown in an industry long dominated by global giant Illumina which controls about 80 percent of the world’s sequencing market.
Ultima’s secret was to completely revamp many technical aspects of the way Illumina have traditionally deciphered DNA. The process usually involves first splitting the double helix DNA structure into single strands, then breaking these strands into short fragments which are laid out on a glass surface called a flow cell. When this flow cell is loaded into the sequencing machine, color-coded tags are attached to each individual base letter. A laser scans the bases individually while a camera simultaneously records the color associated with them, a process which is repeated until every single fragment has been sequenced.
Instead, Ultima has found a series of shortcuts to slash the cost and boost efficiency. “Ultima Genomics has developed a fundamentally new sequencing architecture designed to scale beyond conventional approaches,” says Josh Lauer, Ultima’s chief commercial officer.
This ‘new architecture’ is a series of subtle but highly impactful tweaks to the sequencing process ranging from replacing the costly flow cell with a silicon wafer which is both cheaper and allows more DNA to be read at once, to utilizing machine learning to convert optical data into usable information.
To put $100 genome in perspective, back in 2012 the cost of sequencing a single genome was around $10,000, a price tag which dropped to $1,000 a few years later. Before Ultima’s announcement, the cost of sequencing an individual genome was around $600.
Several studies have found that nearly 12 percent of healthy people who have their genome sequenced, then discover they have a variant pointing to a heightened risk of developing a disease that can be monitored, treated or prevented.
While Ultima’s new machine is not widely available yet, Illumina’s response has been rapid. Last month the company unveiled the NovaSeq X series, which it describes as its fastest most cost-efficient sequencing platform yet, capable of sequencing genomes at $200, with further price cuts likely to follow.
But what will the rapidly tumbling cost of sequencing actually mean for medicine? “Well to start with, obviously it’s going to mean more people getting their genome sequenced,” says Michael Snyder, professor of genetics at Stanford University. “It'll be a lot more accessible to people.”
At the moment sequencing is mainly limited to certain cancer patients where it is used to inform treatment options, and individuals with undiagnosed illnesses. In the past, initiatives such as SeqFirst have attempted further widen access to genome sequencing based on growing amounts of research illustrating the potential benefits of the technology in healthcare. Several studies have found that nearly 12 percent of healthy people who have their genome sequenced, then discover they have a variant pointing to a heightened risk of developing a disease that can be monitored, treated or prevented.
“While whole genome sequencing is not yet widely used in the U.S., it has started to come into pediatric critical care settings such as newborn intensive care units,” says Professor Michael Bamshad, who heads the genetic medicine division in the University of Washington’s pediatrics department. “It is also being used more often in outpatient clinical genetics services, particularly when conventional testing fails to identify explanatory variants.”
But the cost of sequencing itself is only one part of the price tag. The subsequent clinical interpretation and genetic counselling services often come to several thousand dollars, a cost which insurers are not always willing to pay.
As a result, while Bamshad and others hope that the arrival of the $100 genome will create new opportunities to use genetic testing in innovative ways, the most immediate benefits are likely to come in the realm of research.
Bigger Data
There are numerous ways in which cheaper sequencing is likely to advance scientific research, for example the ability to collect data on much larger patient groups. This will be a major boon to scientists working on complex heterogeneous diseases such as schizophrenia or depression where there are many genes involved which all exert subtle effects, as well as substantial variance across the patient population. Bigger studies could help scientists identify subgroups of patients where the disease appears to be driven by similar gene variants, who can then be more precisely targeted with specific drugs.
If insurers can figure out the economics, Snyder even foresees a future where at a certain age, all of us can qualify for annual sequencing of our blood cells to search for early signs of cancer or the potential onset of other diseases like type 2 diabetes.
David Curtis, a genetics professor at University College London, says that scientists studying these illnesses have previously been forced to rely on genome-wide association studies which are limited because they only identify common gene variants. “We might see a significant increase in the number of large association studies using sequence data,” he says. “It would be far preferable to use this because it provides information about rare, potentially functional variants.”
Cheaper sequencing will also aid researchers working on diseases which have traditionally been underfunded. Bamshad cites cystic fibrosis, a condition which affects around 40,000 children and adults in the U.S., as one particularly pertinent example.
“Funds for gene discovery for rare diseases are very limited,” he says. “We’re one of three sites that did whole genome sequencing on 5,500 people with cystic fibrosis, but our statistical power is limited. A $100 genome would make it much more feasible to sequence everyone in the U.S. with cystic fibrosis and make it more likely that we discover novel risk factors and pathways influencing clinical outcomes.”
For progressive diseases that are more common like cancer and type 2 diabetes, as well as neurodegenerative conditions like multiple sclerosis and ALS, geneticists will be able to go even further and afford to sequence individual tumor cells or neurons at different time points. This will enable them to analyze how individual DNA modifications like methylation, change as the disease develops.
In the case of cancer, this could help scientists understand how tumors evolve to evade treatments. Within in a clinical setting, the ability to sequence not just one, but many different cells across a patient’s tumor could point to the combination of treatments which offer the best chance of eradicating the entire cancer.
“What happens at the moment with a solid tumor is you treat with one drug, and maybe 80 percent of that tumor is susceptible to that drug,” says Neil Ward, vice president and general manager in the EMEA region for genomics company PacBio. “But the other 20 percent of the tumor has already got mutations that make it resistant, which is probably why a lot of modern therapies extend life for sadly only a matter of months rather than curing, because they treat a big percentage of the tumor, but not the whole thing. So going forwards, I think that we will see genomics play a huge role in cancer treatments, through using multiple modalities to treat someone's cancer.”
If insurers can figure out the economics, Snyder even foresees a future where at a certain age, all of us can qualify for annual sequencing of our blood cells to search for early signs of cancer or the potential onset of other diseases like type 2 diabetes.
“There are companies already working on looking for cancer signatures in methylated DNA,” he says. “If it was determined that you had early stage cancer, pre-symptomatically, that could then be validated with targeted MRI, followed by surgery or chemotherapy. It makes a big difference catching cancer early. If there were signs of type 2 diabetes, you could start taking steps to mitigate your glucose rise, and possibly prevent it or at least delay the onset.”
This would already revolutionize the way we seek to prevent a whole range of illnesses, but others feel that the $100 genome could also usher in even more powerful and controversial preventative medicine schemes.
Newborn screening
In the eyes of Kári Stefánsson, the Icelandic neurologist who been a visionary for so many advances in the field of human genetics over the last 25 years, the falling cost of sequencing means it will be feasible to sequence the genomes of every baby born.
“We have recently done an analysis of genomes in Iceland and the UK Biobank, and in 4 percent of people you find mutations that lead to serious disease, that can be prevented or dealt with,” says Stefansson, CEO of deCODE genetics, a subsidiary of the pharmaceutical company Amgen. “This could transform our healthcare systems.”
As well as identifying newborns with rare diseases, this kind of genomic information could be used to compute a person’s risk score for developing chronic illnesses later in life. If for example, they have a higher than average risk of colon or breast cancer, they could be pre-emptively scheduled for annual colonoscopies or mammograms as soon as they hit adulthood.
To a limited extent, this is already happening. In the UK, Genomics England has launched the Newborn Genomes Programme, which plans to undertake whole-genome sequencing of up to 200,000 newborn babies, with the aim of enabling the early identification of rare genetic diseases.
"I have not had my own genome sequenced and I would not have wanted my parents to have agreed to this," Curtis says. "I don’t see that sequencing children for the sake of some vague, ill-defined benefits could ever be justifiable.”
However, some scientists feel that it is tricky to justify sequencing the genomes of apparently healthy babies, given the data privacy issues involved. They point out that we still know too little about the links which can be drawn between genetic information at birth, and risk of chronic illness later in life.
“I think there are very difficult ethical issues involved in sequencing children if there are no clear and immediate clinical benefits,” says Curtis. “They cannot consent to this process. I have not had my own genome sequenced and I would not have wanted my parents to have agreed to this. I don’t see that sequencing children for the sake of some vague, ill-defined benefits could ever be justifiable.”
Curtis points out that there are many inherent risks about this data being available. It may fall into the hands of insurance companies, and it could even be used by governments for surveillance purposes.
“Genetic sequence data is very useful indeed for forensic purposes. Its full potential has yet to be realized but identifying rare variants could provide a quick and easy way to find relatives of a perpetrator,” he says. “If large numbers of people had been sequenced in a healthcare system then it could be difficult for a future government to resist the temptation to use this as a resource to investigate serious crimes.”
While sequencing becoming more widely available will present difficult ethical and moral challenges, it will offer many benefits for society as a whole. Cheaper sequencing will help boost the diversity of genomic datasets which have traditionally been skewed towards individuals of white, European descent, meaning that much of the actionable medical information which has come out of these studies is not relevant to people of other ethnicities.
Ward predicts that in the coming years, the growing amount of genetic information will ultimately change the outcomes for many with rare, previously incurable illnesses.
“If you're the parent of a child that has a susceptible or a suspected rare genetic disease, their genome will get sequenced, and while sadly that doesn’t always lead to treatments, it’s building up a knowledge base so companies can spring up and target that niche of a disease,” he says. “As a result there’s a whole tidal wave of new therapies that are going to come to market over the next five years, as the genetic tools we have, mature and evolve.”
In this week's Friday Five, an old diabetes drug finds an exciting new purpose. Plus, how to make the cities of the future less toxic, making old mice younger with EVs, a new reason for mysterious stillbirths - and much more.
The Friday Five covers five stories in research that you may have missed this week. There are plenty of controversies and troubling ethical issues in science – and we get into many of them in our online magazine – but this news roundup focuses on scientific creativity and progress to give you a therapeutic dose of inspiration headed into the weekend.
Here is the promising research covered in this week's Friday Five:
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
- How to make cities of the future less noisy
- An old diabetes drug could have a new purpose: treating an irregular heartbeat
- A new reason for mysterious stillbirths
- Making old mice younger with EVs
- No pain - or mucus - no gain
And an honorable mention this week: How treatments for depression can change the structure of the brain

