9 Tips for Online Mental Health Therapy

Research shows that, for most patients, online therapy offers the same benefits as in-person therapy, yet many people still resist it. A behavioral scientist explains how you can use it to improve mental health.
Telehealth offers a vast improvement in access and convenience to all sorts of medical services, and online therapy for mental health is one of the most promising case studies for telehealth. With many online therapy options available, you can choose whatever works best for you. Yet many people are hesitant about using online therapy. Even if they do give it a try, they often don’t know how to make the most effective use of this treatment modality.
Why do so many feel uncertain about online therapy? A major reason stems from its novelty. Humans are creatures of habit, prone to falling for what behavioral scientists like myself call the status quo bias, a predisposition to stick to traditional practices and behaviors. Many people reject innovative solutions even when they would be helpful. Thus, while teletherapy was available long before the pandemic, and might have fit the needs of many potential clients, relatively few took advantage of this option.
Even when we do try new methodologies, we often don’t do so effectively, because we cling to the same approaches that worked in previous situations. Scientists call this behavior functional fixedness. It’s kind of like the saying about the hammer-nail syndrome: “when you have a hammer, everything looks like a nail.”
These two mental blindspots, the status quo bias and functional fixedness, impact decision making in many areas of life. Fortunately, recent research has shown effective and pragmatic strategies to defeat these dangerous errors in judgment. The nine tips below will help you make the best decisions to get effective online therapy, based on the latest research.
Trust the science of online therapy
Extensive research shows that, for most patients, online therapy offers the same benefits as in-person therapy.
For instance, a 2014 study in the Journal of Affective Disorders reported that online treatment proved just as effective as face-to-face treatment for depression. A 2018 study, published in Journal of Psychological Disorders, found that online cognitive behavioral therapy, or CBT, was just as effective as face-to-face treatment for major depression, panic disorder, social anxiety disorder, and generalized anxiety disorder. And a 2014 study in Behaviour Research and Therapy discovered that online CBT proved effective in treating anxiety disorders, and helped lower costs of treatment.
During the forced teletherapy of COVID, therapists worried that those with serious mental health conditions would be less likely to convert to teletherapy. Yet research published in Counselling Psychology Quarterly has helped to alleviate that concern. It found that those with schizophrenia, bipolar disorder, severe depression, PTSD, and even suicidality converted to teletherapy at about the same rate as those with less severe mental health challenges.
Yet teletherapy may not be for everyone. For example, adolescents had the most varied response to teletherapy, according to a 2020 study in Family Process. Some adapted quickly and easily, while others found it awkward and anxiety-inducing. On the whole, children with trauma respond worse to online therapy, per a 2020 study in Child Abuse & Neglect. The treatment of mental health issues can sometimes require in-person interactions, such as the use of eye movement desensitization and reprocessing to treat post-traumatic stress disorder. And according to a 2020 study from the Journal of Humanistic Psychology, online therapy may not be as effective for those suffering from loneliness.
Leverage the strengths of online therapy
Online therapy is much more accessible than in-person therapy for those with a decent internet connection, webcam, mic, and digital skills. You don’t have to commute to your therapist’s office, wasting money and time. You can take much less medical leave from work, saving you money and hassle with your boss. If you live in a sparsely populated area, online therapy could allow you to access many specialized kinds of therapy that isn’t accessible locally.
Online options are much quicker compared to the long waiting lines for in-person therapy. You also have much more convenient scheduling options. And you won’t have to worry about running into someone you know in the waiting room. Online therapy is easier to conceal from others and reduces stigma. Many patients may feel more comfortable and open to sharing in the privacy and comfort of their own home.
You can use a variety of communication tools suited to your needs at any given time. Video can be used to start a relationship with a therapist and have more intense and nuanced discussions, but can be draining, especially for those with social anxiety. Voice-only may work well for less intense discussions. Email offers a useful option for long-form, well-thought-out messages. Texting is useful for quick, real-time questions, answers, and reinforcement.
Plus, online therapy is often cheaper than in-person therapy. In the midst of COVID, many insurance providers have decided to cover online therapy.
Address the weaknesses
One weakness is the requirement for appropriate technology and skills to engage in online therapy. Another is the difficulty of forming a close therapeutic relationship with your therapist. You won’t be able to communicate non-verbals as fully and the therapist will not be able to read you as well, requiring you to be more deliberate in how you express yourself.
Another important issue is that online therapy is subject to less government oversight compared to the in-person approach, which is regulated in each state, providing a baseline of quality control. As a result, you have to do more research on the providers that offer online therapy to make sure they’re reputable, use only licensed therapists, and have a clear and transparent pay structure.
Be intentional about advocating for yourself
Figure out what kind of goals you want to achieve. Consider how, within the context of your goals, you can leverage the benefits of online therapy while addressing the weaknesses. Write down and commit to achieving your goals. Remember, you need to be your own advocate, especially in the less regulated space of online therapy, so focus on being proactive in achieving your goals.
Develop your Hero’s Journey
Because online therapy can occur at various times of day through videos calls, emails and text, it might feel more open-ended and less organized, which can have advantages and disadvantages. One way you can give it more structure is to ground these interactions in the story of your self-improvement. Our minds perceive the world through narratives. Create a story of how you’ll get from where you are to where you want to go, meaning your goals.
A good template to use is the Hero’s Journey. Start the narrative with where you are, and what caused you to seek therapy. Write about the obstacles you will need to overcome, and the kind of help from a therapist that you’ll need in the process. Then, describe the final end state: how will you be better off after this journey, including what you will have learned.
Especially in online therapy, you need to be on top of things. Too many people let the therapist manage the treatment plan. As you pursue your hero’s journey, another way to organize for success is to take notes on your progress, and reevaluate how you’re doing every month with your therapist.
Identify your ideal mentor
Since it’s more difficult to be confident about the quality of service providers in an online setting, you should identify in advance the traits of your desired therapist. Every Hero’s Journey involves a mentor figure who guides the protagonist through this journey. So who’s your ideal mentor? Write out their top 10 characteristics, from most to least important.
For example, you might want someone who is:
- Empathetic
- Caring
- Good listener
- Logical
- Direct
- Questioning
- Non-judgmental
- Organized
- Curious
- Flexible
That’s my list. Depending on what challenge you’re facing and your personality and preferences, you should make your own. Then, when you are matched with a therapist, evaluate how well they fit your ideal list.
Fail fast
When you first match with a therapist, try to fail fast. That means, instead of focusing on getting treatment, focus on figuring out if the therapist is a good match based on the traits you identified above. That will enable you to move on quickly if they’re not, and it’s very much worth it to figure that out early.
Tell them your goals, your story, and your vision of your ideal mentor. Ask them whether they think they are a match, and what kind of a treatment plan they would suggest based on the information you provided. And observe them yourself in your initial interactions, focusing on whether they’re a good match. Often, you’ll find that your initial vision of your ideal mentor is incomplete, and you’ll learn through doing therapy what kind of a therapist is the best fit for you.
Choose a small but meaningful subgoal to work on first
This small subgoal should be sufficient to be meaningful and impactful for improving your mental health, but not a big stretch for you to achieve. This subgoal should be a tool for you to use to evaluate whether the therapist is indeed a good fit for you. It will also help you evaluate whether the treatment plan makes sense, or whether it needs to be revised.
Know when to wrap things up
As you approach the end of your planned work and you see you’re reaching your goals, talk to the therapist about how to wrap up rather than letting things drag on for too long. You don’t want to become dependent on therapy: it’s meant to be a temporary intervention. Some less scrupulous therapists will insist that therapy should never end and we should all stay in therapy forever, and you want to avoid falling for this line. When you reach your goals, end your therapy, unless you discover a serious new reason to continue it. Still, it may be wise to set up occasional check-ins once every three to six months to make sure you’re staying on the right track.
Want to Motivate Vaccinations? Message Optimism, Not Doom
There is a lot to be optimistic about regarding the new safe and highly effective vaccines, which are moving society closer toward the goal of close human contact once again.
After COVID-19 was declared a worldwide pandemic by the World Health Organization on March 11, 2020, life as we knew it altered dramatically and millions went into lockdown. Since then, most of the world has had to contend with masks, distancing, ventilation and cycles of lockdowns as surges flare up. Deaths from COVID-19 infection, along with economic and mental health effects from the shutdowns, have been devastating. The need for an ultimate solution -- safe and effective vaccines -- has been paramount.
On November 9, 2020 (just 8 months after the pandemic announcement), the press release for the first effective COVID-19 vaccine from Pfizer/BioNTech was issued, followed by positive announcements regarding the safety and efficacy of five other vaccines from Moderna, University of Oxford/AztraZeneca, Novavax, Johnson and Johnson and Sputnik V. The Moderna and Pfizer vaccines have earned emergency use authorization through the FDA in the United States and are being distributed. We -- after many long months -- are seeing control of the devastating COVID-19 pandemic glimmering into sight.
To be clear, these vaccine candidates for COVID-19, both authorized and not yet authorized, are highly effective and safe. In fact, across all trials and sites, all six vaccines were 100% effective in preventing hospitalizations and death from COVID-19.
All Vaccines' Phase 3 Clinical Data
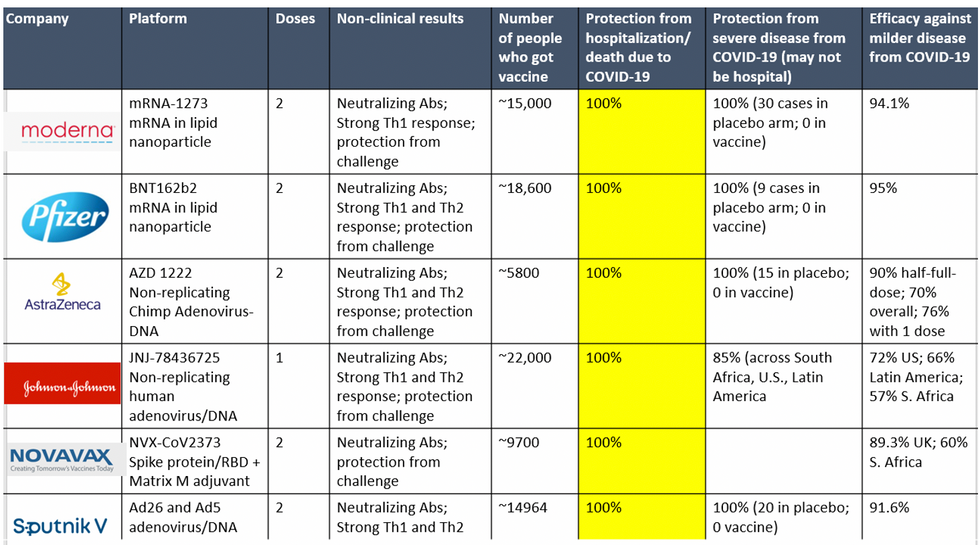
Complete protection against hospitalization and death from COVID-19 exhibited by all vaccines with phase 3 clinical trial data.
This astounding level of protection from SARS-CoV-2 from all vaccine candidates across multiple regions is likely due to robust T cell response from vaccination and will "defang" the virus from the concerns that led to COVID-19 restrictions initially: the ability of the virus to cause severe illness. This is a time of hope and optimism. After the devastating third surge of COVID-19 infections and deaths over the winter, we finally have an opportunity to stem the crisis – if only people readily accept the vaccines.
Amidst these incredible scientific advancements, however, public health officials and politicians have been pushing downright discouraging messaging. The ubiquitous talk of ongoing masks and distancing restrictions without any clear end in sight threatens to dampen uptake of the vaccines. It's imperative that we break down each concern and see if we can revitalize our public health messaging accordingly.
The first concern: we currently do not know if the vaccines block asymptomatic infection as well as symptomatic disease, since none of the phase 3 vaccine trials were set up to answer this question. However, there is biological plausibility that the antibodies and T-cell responses blocking symptomatic disease will also block asymptomatic infection in the nasal passages. IgG immunoglobulins (generated and measured by the vaccine trials) enter the nasal mucosa and systemic vaccinations generate IgA antibodies at mucosal surfaces. Monoclonal antibodies given to outpatients with COVID-19 hasten viral clearance from the airways.
Although it is prudent for those who are vaccinated to wear masks around the unvaccinated in case a slight risk of transmission remains, two fully vaccinated people can comfortably abandon masking around each other.
Moreover, data from the AztraZeneca trial (including in the phase 3 trial final results manuscript), where weekly self-swabbing was done by participants, and data from the Moderna trial, where a nasal swab was performed prior to the second dose, both showed risk reductions in asymptomatic infection with even a single dose. Finally, real-world data from a large Pfizer-based vaccine campaign in Israel shows a 50% reduction in infections (asymptomatic or symptomatic) after just the first dose.
Therefore, the likelihood of these vaccines blocking asymptomatic carriage, as well as symptomatic disease, is high. Although it is prudent for those who are vaccinated to wear masks around the unvaccinated in case a slight risk of transmission remains, two fully vaccinated people can comfortably abandon masking around each other. Moreover, as the percentage of vaccinated people increases, it will be increasingly untenable to impose restrictions on this group. Once herd immunity is reached, these restrictions can and should be abandoned altogether.
The second concern translating to "doom and gloom" messaging lately is around the identification of troubling new variants due to enhanced surveillance via viral sequencing. Four major variants circulating at this point (with others described in the past) are the B.1.1.7 variant ("UK variant"), B.1.351 ("South Africa variant), P.1. ("Brazil variant"), and the L452R variant identified in California. Although the UK variant is likely to be more transmissible, as is the South Africa variant, we have no reason to believe that masks, distancing and ventilation are ineffective against these variants.
Moreover, neutralizing antibody titers with the Pfizer and Moderna vaccines do not seem to be significantly reduced against the variants. Finally, although the Novavax 2-dose and Johnson and Johnson (J&J) 1-dose vaccines had lower rates of efficacy against moderate COVID-19 disease in South Africa, their efficacy against severe disease was impressively high. In fact J&J's vaccine still prevented 100% of hospitalizations and death from COVID-19. When combining both hospitalizations/deaths and severe symptoms managed at home, the J&J 1-dose vaccine was 85% protective across all three sites of the trial: the U.S., Latin America (including Brazil), and South Africa.
In South Africa, nearly all cases of COVID-19 (95%) were due to infection with the B.1.351 SARS-CoV-2 variant. Finally, since herd immunity does not rely on maximal immune responses among all individuals in a society, the Moderna/Pfizer/J&J vaccines are all likely to achieve that goal against variants. And thankfully, all of these vaccines can be easily modified to boost specifically against a new variant if needed (indeed, Moderna and Pfizer are already working on boosters against the prominent variants).
The third concern of some public health officials is that people will abandon all restrictions once vaccinated unless overly cautious messages are drilled into them. Indeed, the false idea that if you "give people an inch, they will take a mile" has been misinforming our messaging about mitigation since the beginning of the pandemic. For example, the very phrase "stay at home" with all of its non-applicability for essential workers and single individuals is stigmatizing and unrealistic for many. Instead, the message should have focused on how people can additively reduce their risks under different circumstances.
The public will be more inclined to trust health officials if those officials communicate with nuanced messages backed up by evidence, rather than with broad brushstrokes that shame. Therefore, we should be saying that "vaccinated people can be together with other vaccinated individuals without restrictions but must protect the unvaccinated with masks and distancing." And we can say "unvaccinated individuals should adhere to all current restrictions until vaccinated" without fear of misunderstandings. Indeed, this kind of layered advice has been communicated to people living with HIV and those without HIV for a long time (if you have HIV but partner does not, take these precautions; if both have HIV, you can do this, etc.).
Our heady progress in vaccine development, along with the incredible efficacy results of all of them, is unprecedented. However, we are at risk of undermining such progress if people balk at the vaccine because they don't believe it will make enough of a difference. One of the most critical messages we can deliver right now is that these vaccines will eventually free us from the restrictions of this pandemic. Let's use tiered messaging and clear communication to boost vaccine optimism and uptake, and get us to the goal of close human contact once again.
Inside Scoop: How a DARPA Scientist Helped Usher in a Game-Changing Covid Treatment
Amy Jenkins is a program manager for the Defense Advanced Research Projects Agency's Biological Technologies Office, which runs a project called the Pandemic Prevention Platform.
Amy Jenkins was in her office at DARPA, a research and development agency within the Department of Defense, when she first heard about a respiratory illness plaguing the Chinese city of Wuhan. Because she's a program manager for DARPA's Biological Technologies Office, her colleagues started stopping by. "It's really unusual, isn't it?" they would say.
At the time, China had a few dozen cases of what we now call COVID-19. "We should maybe keep an eye on that," she thought.
Early in 2020, still just keeping watch, she was visiting researchers working on DARPA's Pandemic Prevention Platform (P3), a project to develop treatments for "any known or previously unknown infectious threat," within 60 days of its appearance. "We looked at each other and said, 'Should we be doing something?'" she says.
For projects like P3, groups of scientists—often at universities and private companies—compete for DARPA contracts, and program managers like Jenkins oversee the work. Those that won the P3 bid included scientists at AbCellera Biologics, Inc., AstraZeneca, Duke University, and Vanderbilt University.
At the time Jenkins was talking to the P3 performers, though, they didn't have evidence of community transmission. "We would have to cross that bar before we considered doing anything," she says.
The world soon leapt far over that bar. By the time Jenkins and her team decided P3 should be doing something—with their real work beginning in late February--it was too late to prevent this pandemic. But she could help P3 dig into the chemical foundations of COVID-19's malfeasance, and cut off its roots. That work represents, in fact, her roots.
In late February 2020, DARPA received a single blood sample from a recovered COVID-19 patient, in which P3 researchers could go fishing for antibodies. The day it arrived, Jenkins's stomach roiled. "We get one shot," she thought.
Fighting the Smallest Enemies
Jenkins, who's in her early 40s, first got into germs the way many 90s kids did: by reading The Hot Zone, a novel about a hemorrhagic fever gone rogue. It wasn't exactly the disintegrating organs that hooked her. It was the idea that "these very pathogens that we can't even see can make us so sick and bring us to our knees," she says. Reading about scientists facing down deadly disease, she wondered, "How do these things make you so sick?"
She chased that question in college, majoring in both biomolecular science and chemistry, and later became an antibody expert. Antibodies are proteins that hook to a pathogen to block it from attaching to your cells, or tag it for destruction by the rest of the immune system. Soon, she jumped on the "monoclonal antibodies" train—developing synthetic versions of these natural defenses, which doctors can give to people to help them battle an early-stage infection, and even to prevent an infection from taking root after an exposure.
Jenkins likens the antibody treatments to the old aphorism about fishing: Vaccines teach your body how to fish, but antibodies simply give your body the pesca-fare. While that, as the saying goes, won't feed you for a lifetime, it will last a few weeks or months. Monoclonal antibodies thus are a promising preventative option in the immediate short-term when a vaccine hasn't yet been given (or hasn't had time to produce an immune response), as well as an important treatment weapon in the current fight. After former president Donald Trump contracted COVID-19, he received a monoclonal antibody treatment from biotech company Regeneron.
As for Jenkins, she started working as a DARPA Biological Technologies Office contractor soon after completing her postdoc. But it was a suit job, not a labcoat job. And suit jobs, at first, left Jenkins conflicted, worried about being bored. She'd give it a year, she thought. But the year expired, and bored she was not. Around five years later, in June 2019, the agency hired her to manage several of the office's programs. A year into that gig, the world was months into a pandemic.
The Pandemic Pivot
At DARPA, Jenkins inherited five programs, including P3. P3 works by taking blood from recovered people, fishing out their antibodies, identifying the most effective ones, and then figuring out how to manufacture them fast. Back then, P3 existed to help with nebulous, future outbreaks: Pandemic X. Not this pandemic. "I did not have a crystal ball," she says, "but I will say that all of us in the infectious diseases and public-health realm knew that the next pandemic was coming."
Three days after a January 2020 meeting with P3 researchers, COVID-19 appeared in Seattle, then began whipping through communities. The time had come for P3 teams to swivel. "We had done this," she says. "We had practiced this before." But would their methods stand up to something unknown, racing through the global population? "The big anxiety was, 'Wow, this was real,'" says Jenkins.
While facing down that realness, Jenkins was also managing other projects. In one called PREPARE, groups develop "medical countermeasures" that modulate a person's genetic code to boost their bodies' responses to threats. Another project, NOW, envisions shipping-container-sized factories that can make thousands of vaccine doses in days. And then there's Prometheus—which means "forethought" in Greek, and is the name of the god who stole fire and gave it to humans. Wrapping up as COVID ramped up, Prometheus aimed to identify people who are contagious—with whatever—before they start coughing, and even if they never do.
All of DARPA's projects focus on developing early-stage technology, passing it off to other agencies or industry to put it into operation. The orientation toward a specific goal appealed to Jenkins, as a contrast to academia. "You go down a rabbit hole for years at a time sometimes, chasing some concept you found interesting in the lab," she says. That's good for the human pursuit of knowledge, and leads to later applications, but DARPA wants a practical prototype—stat.
"Dual-Use" Technologies
That desire, though, and the fact that DARPA is a defense agency, present philosophical complications. "Bioethics in the national-security context turns all the dials up to 10+," says Jonathan Moreno, a medical ethicist at the University of Pennsylvania.
While developing antibody treatments to stem a pandemic seems straightforwardly good, all biological research—especially that backed by military money—requires evaluating potential knock-on applications, even those that might come from outside the entity that did the developing. As Moreno put it, "Albert Einstein wasn't thinking about blowing up Hiroshima." Particularly sensitive are so-called "dual-use" technologies—those tools that could be used for both benign and nefarious purposes, or are of interest to both the civilian and military worlds.
Moreno takes Prometheus itself as an example of "dual-use" technology. "Think about somebody wearing a suicide vest. Instead of a suicide vest, make them extremely contagious with something. The flu plus Ebola," he says. "Send them someplace, a sensitive environment. We would like to be able to defend against that"—not just tell whether Uncle Fred is bringing asymptomatic COVID home for Christmas. Prometheus, Jenkins says, had safety in mind from the get-go, and required contenders to "develop a risk mitigation plan" and "detail their strategy for appropriate control of information."
To look at a different program, if you can modulate genes to help healing, you probably know something (or know someone else could infer something) about how to hinder healing. Those sorts of risks are why PREPARE researchers got their own "ethical, legal, and social implications" panel, which meets quarterly "to ensure that we are performing all research and publications in a safe and ethical manner," says Jenkins.
DARPA as a whole, Moreno says, is institutionally sensitive to bioethics. The agency has ethics panels, and funded a 2014 National Academies assessment of how to address the "ethical, legal, and societal issues" around technology that has military relevance. "In the cases of biotechnologies where some of that research brushes up against what could legitimately be considered dual-use, that in itself justifies our investment," says Jenkins. "DARPA deliberately focuses on safety and countermeasures against potentially dangerous technologies, and we structure our programs to be transparent, safe, and legal."
Going Fishing
In late February 2020, DARPA received a single blood sample from a recovered COVID-19 patient, in which P3 researchers could go fishing for antibodies. The day it arrived, Jenkins's stomach roiled. "We get one shot," she thought.
As scientists from the P3-funded AbCellera went through the processes they'd practiced, Jenkins managed their work, tracking progress and relaying results. Soon, the team had isolated a suitable protein: bamlanivimab. It attaches to and blocks off the infamous spike proteins on SARS-CoV-2—those sticky suction-cups in illustrations. Partnering with Eli Lilly in a manufacturing agreement, the biotech company brought it to clinical trials in May, just a few months after its work on the deadly pathogen began, after much of the planet became a hot zone.
On November 10—Jenkins's favorite day at the (home) office—the FDA provided Eli Lilly emergency use authorization for bamlanivimab. But she's only mutedly screaming (with joy) inside her heart. "This pandemic isn't 'one morning we're going to wake up and it's all over,'" she says. When it is over, she and her colleagues plan to celebrate their promethean work. "I'm hoping to be able to do it in person," she says. "Until then, I have not taken a breath."

