The Sickest Babies Are Covered in Wires. New Tech Is Changing That.
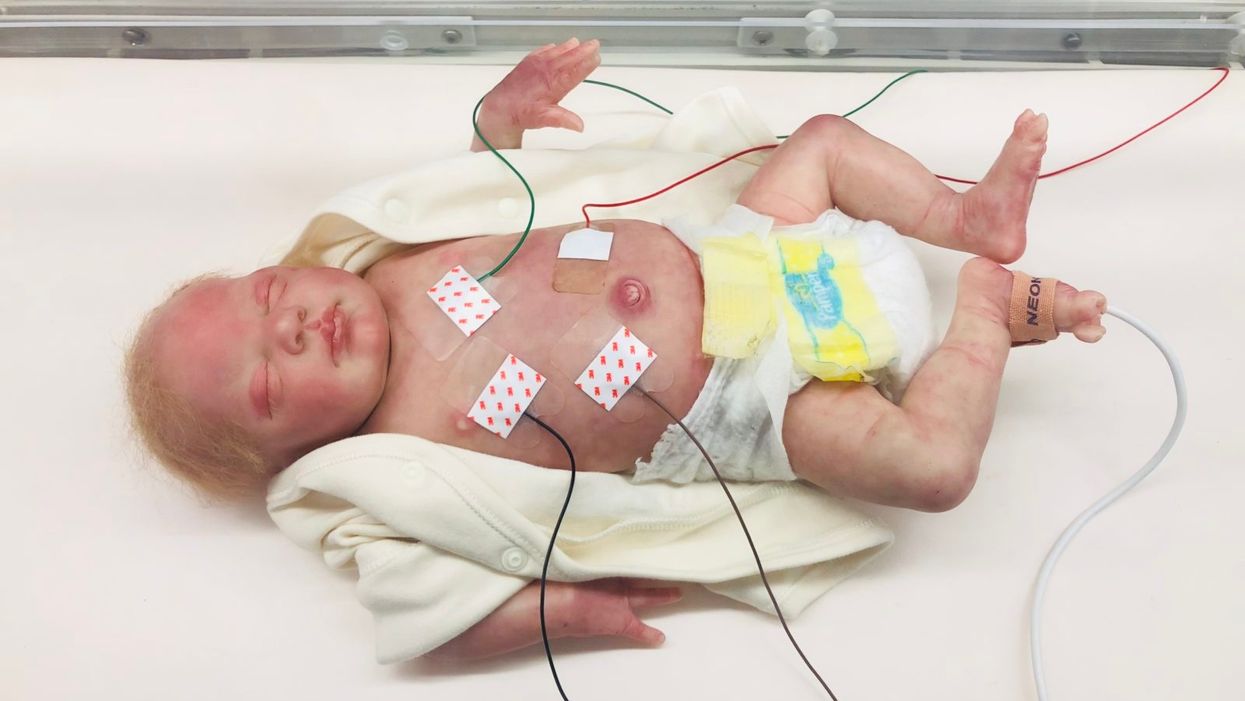
A wired baby in a neonatal intensive care unit.
I'll never forget the experience of having a child in the neonatal intensive care unit (NICU).
Now more than ever, we're working to remove the barriers between new parents and their infants.
It was another layer of uncertainty that filtered into my experience of being a first-time parent. There was so much I didn't know, and the wires attached to my son's small body for the first week of his life were a reminder of that.
I wanted to be the best mother possible. I deeply desired to bring my son home to start our lives. More than anything, I longed for a wireless baby whom I could hold and love freely without limitations.
The wires suggested my baby was fragile and it left me feeling severely unprepared, anxious, and depressed.
In recent years, research has documented the ways that NICU experiences take a toll on parents' mental health. But thankfully, medical technology is rapidly being developed to help reduce the emotional fallout of the NICU. Now more than ever, we're working to remove the barriers between new parents and their infants. The latest example is the first ever wireless monitoring system that was recently developed by a team at Northwestern University.
After listening to the needs of parents and medical staff, Debra Weese-Mayer, M.D., a professor of pediatric autonomic medicine at Feinberg School of Medicine, along with a team of materials scientists, engineers, dermatologists and pediatricians, set out to develop this potentially life-changing technology. Weese-Mayer believes wireless monitoring will have a significant impact for people on all sides of the NICU experience.
"With elimination of the cumbersome wires," she says, "the parents will find their infant more approachable/less intimidating and have improved access to their long-awaited but delivered-too-early infant, allowing them to begin skin-to-skin contact and holding with reduced concern for dislodging wires."
So how does the new system work?
Very thin "skin like" patches made of silicon rubber are placed on the surface of the skin to monitor vitals like heart rate, respiration rate, and body temperature. One patch is placed on the chest or back and the other is placed on the foot.
These patches are safer on the skin than previously used adhesives, reducing the cuts and infections associated with past methods. Finally, an antenna continuously delivers power, often from under the mattress.
The data collected from the patches stream from the body to a tablet or computer.

New wireless sensor technology is being studied to replace wired monitoring in NICUs in the coming years.
(Northwestern University)
Weese-Mayer hopes that wireless systems will be standard soon, but first they must undergo more thorough testing. "I would hope that in the next five years, wireless monitoring will be the standard in NICUs, but there are many essential validation steps before this technology will be embraced nationally," she says.
Until the new systems are ready, parents will be left struggling with the obstacles that wired monitoring presents.
Physical intimacy, for example, appears to have pain-reducing qualities -- something that is particularly important for babies who are battling serious illness. But wires make those cuddles more challenging.
There's also been minimal discussion about how wired monitoring can be particularly limiting for parents with disabilities and mobility aids, or even C-sections.
"When he was first born and I was recovering from my c-section, I couldn't deal with keeping the wires untangled while trying to sit down without hurting myself," says Rhiannon Giles, a writer from North Carolina, who delivered her son at just over 31 weeks after suffering from severe preeclampsia.
"The wires were awful," she remembers. "They fell off constantly when I shifted positions or he kicked a leg, which meant the monitors would alarm. It felt like an intrusion into the quiet little world I was trying to mentally create for us."
Over the last few years, researchers have begun to dive deeper into the literal and metaphorical challenges of wired monitoring.
For many parents, the wires prompt anxiety that worsens an already tense and vulnerable time.
I'll never forget the first time I got to hold my son without wires. It was the first time that motherhood felt manageable.
"Seeing my five-pound-babies covered in wires from head to toe rendered me completely overwhelmed," recalls Caila Smith, a mom of five from Indiana, whose NICU experience began when her twins were born pre-term. "The nurses seemed to handle them perfectly, but I was scared to touch them while they appeared so medically frail."
During the nine days it took for both twins to come home, the limited access she had to her babies started to impact her mental health. "If we would've had wireless sensors and monitors, it would've given us a much greater sense of freedom and confidence when snuggling our newborns," Smith says.
Besides enabling more natural interactions, wireless monitoring would make basic caregiving tasks much easier, like putting on a onesie.
"One thing I noticed is that many preemie outfits are made with zippers," points out Giles, "which just don't work well when your baby has wires coming off of them, head to toe."
Wired systems can pose issues for medical staff as well as parents.
"The main concern regarding wired systems is that they restrict access to the baby and often get tangled with other equipment, like IV lines," says Lamia Soghier, Medical Director of the Neonatal Intensive Care Unit at Children's National in Washington, D.C , who was also a NICU parent herself. "The nurses have to untangle the wires, which takes time, before handing the baby to the family."
I'll never forget the first time I got to hold my son without wires. It was the first time that motherhood felt manageable, and I couldn't stop myself from crying. Suddenly, anything felt possible and all the limitations from that first week of life seemed to fade away. The rise of wired-free monitoring will make some of the stressors that accompany NICU stays a thing of the past.
Entomologist Jessica Ware is using new technologies to identify insect species in a changing climate. She shares her suggestions for how we can live harmoniously with creeper crawlers everywhere.
Jessica Ware is obsessed with bugs.
My guest today is a leading researcher on insects, the president of the Entomological Society of America and a curator at the American Museum of Natural History. Learn more about her here.
You may not think that insects and human health go hand-in-hand, but as Jessica makes clear, they’re closely related. A lot of people care about their health, and the health of other creatures on the planet, and the health of the planet itself, but researchers like Jessica are studying another thing we should be focusing on even more: how these seemingly separate areas are deeply entwined. (This is the theme of an upcoming event hosted by Leaps.org and the Aspen Institute.)
Listen to the Episode
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google

Entomologist Jessica Ware
D. Finnin / AMNH
Maybe it feels like a core human instinct to demonize bugs as gross. We seem to try to eradicate them in every way possible, whether that’s with poison, or getting out our blood thirst by stomping them whenever they creep and crawl into sight.
But where did our fear of bugs really come from? Jessica makes a compelling case that a lot of it is cultural, rather than in-born, and we should be following the lead of other cultures that have learned to live with and appreciate bugs.
The truth is that a healthy planet depends on insects. You may feel stung by that news if you hate bugs. Reality bites.
Jessica and I talk about whether learning to live with insects should include eating them and gene editing them so they don’t transmit viruses. She also tells me about her important research into using genomic tools to track bugs in the wild to figure out why and how we’ve lost 50 percent of the insect population since 1970 according to some estimates – bad news because the ecosystems that make up the planet heavily depend on insects. Jessica is leading the way to better understand what’s causing these declines in order to start reversing these trends to save the insects and to save ourselves.
They received retinal implants to restore their vision. Then the company turned its back on them.
A company called Second Sight made an implant that partially restored vision to people who'd been blind for decades. But when Second Sight pivoted, it stopped servicing its product, leaving many in the dark.
The first thing Jeroen Perk saw after he partially regained his sight nearly a decade ago was the outline of his guide dog Pedro.
“There was a white floor, and the dog was black,” recalls Perk, a 43-year-old investigator for the Dutch customs service. “I was crying. It was a very nice moment.”
Perk was diagnosed with retinitis pigmentosa as a child and had been blind since early adulthood. He has been able to use the implant placed into his retina in 2013 to help identify street crossings, and even ski and pursue archery. A video posted by the company that designed and manufactured the device indicates he’s a good shot.
Less black-and-white has been the journey Perk and others have been on after they were implanted with the Argus II, a second-generation device created by a Los Angeles-based company called Second Sight Medical Devices.
The Argus II uses the implant and a video camera embedded in a special pair of glasses to provide limited vision to those with retinitis pigmentosa, a genetic disease that causes cells in the retina to deteriorate. The camera feeds information to the implant, which sends electrical impulses into the retina to recapitulate what the camera sees. The impulses appear in the Argus II as a 60-pixel grid of blacks, grays and whites in the user’s eye that can render rough outlines of objects and their motion.
Smartphone and computer manufacturers typically stop issuing software upgrades to their devices after two or three years, eventually rendering them bricks. But is the smartphone approach acceptable for a device that helps restore the most crucial sense a human being possesses?
Ross Doerr, a retired disability rights attorney in Maine who received an Argus II in 2019, describes the field of vision as the equivalent of an index card held at arm’s length. Perk often brings objects close to his face to decipher them. Moreover, users must swivel their heads to take in visual data; moving their eyeballs does not work.
Despite its limitations, the Argus II beats the alternative. Perk no longer relies on his guide dog. Doerr was uplifted when he was able to see the outlines of Christmas trees at a holiday show.
“The fairy godmother department sort of reaches out and taps you on the shoulder once in a while,” Doerr says of his implant, which came about purely by chance. A surgeon treating his cataracts was partnered with the son of another surgeon who was implanting the devices, and he was referred.
Doerr had no reason to believe the shower of fairy dust wouldn’t continue. Second Sight held out promises that the Argus II recipients’ vision would gradually improve through upgrades to much higher pixel densities. The ability to recognize individual faces was even touted as a possibility. In the winter of 2020, Doerr was preparing to travel across the U.S. to Second Sight’s headquarters to receive an upgrade. But then COVID-19 descended, and the trip was canceled.
The pandemic also hit Second Sight’s bottom line. Doerr found out about its tribulations only from one of the company’s vision therapists, who told him the entire department was being laid off. Second Sight cut nearly 80% of its workforce in March 2020 and announced it would wind down operations.

Ross Doerr has mostly stopped using his Argus II, the result of combination of fear of losing its assistance from wear and tear and disdain for the company that brought it to market.
Jan Doerr
Second Sight’s implosion left some 350 Argus recipients in the metaphorical dark about what to do if their implants failed. Skeleton staff seem to have rarely responded to queries from their customers, at least based on the experiences of Perk and Doerr. And some recipients have unfortunately returned to the actual dark as well, as reports have surfaced of Argus II failures due to aging or worn-down parts.
Product support for complex products is remarkably uneven. Although the iconic Ford Mustang ceased production in the late 1960s, its parts market is so robust that it’s theoretically possible to assemble a new vehicle from recently crafted components. Conversely, smartphone and computer manufacturers typically stop issuing software upgrades to their devices after two or three years, eventually rendering them bricks. Consumers have accepted both extremes.
But is the smartphone approach acceptable for a device that helps restore the most crucial sense a human being possesses?
Margaret McLean, a senior fellow at the Markkula Center for Applied Ethics at Santa Clara University in California, notes companies like Second Sight have a greater obligation for product support than other consumer product ventures.
“In this particular case, you have a great deal of risk that is involved in using this device, the implant, and the after care of this device,” she says. “You cannot, like with your car, decide that ‘I don’t like my Mustang anymore,’ and go out and buy a Corvette.”
And, whether the Argus II implant works or not, its physical presence can impact critical medical decisions. Doerr’s doctor wanted him to undergo an MRI to assist in diagnosing attacks of vertigo. But the physician was concerned his implant might interfere. With the latest available manufacturer advisories on his implant nearly a decade old, the procedure was held up. Doerr spent months importuning Second Sight through phone calls, emails and Facebook postings to learn if his implant was contraindicated with MRIs, which he never received. Although the cause of his vertigo was found without an MRI, Doerr was hardly assured.
“Put that into context for a minute. I get into a serious car accident. I end up in the emergency room, and I have a tag saying I have an implanted medical device,” he says. “You can’t do an MRI until you get the proper information from the company. Who’s going to answer the phone?”
Second Sight’s management did answer the call to revamp its business. It netted nearly $78 million through a private stock placement and an initial public offering last year. At the end of 2021, Second Sight had nearly $70 million in cash on hand, according to a recent filing with the Securities and Exchange Commission.
And while the Argus II is still touted at length on Second Sight’s home page, it appears little of its corporate coffers are earmarked toward its support. These days, the company is focused on obtaining federal approvals for Orion, a new implant that would go directly into the recipient’s brain and could be used to remedy blindness from a variety of causes. It obtained a $6.4 million grant from the National Institutes of Health in May 2021 to help develop Orion.
Presented with a list of written questions by email, Second Sight’s spokesperson, Dave Gentry of the investor relations firm Red Chip Companies, copied a subordinate with an abrupt message to “please handle.” That was the only response from a company representative. A call to Second Sight acting chief executive officer Scott Dunbar went unreturned.
Whether or not the Orion succeeds remains to be seen. The company’s SEC filings suggest a viable and FDA-approved device is years away, and that operational losses are expected for the “foreseeable future.” Second Sight reported zero revenue in 2020 or 2021.
Moreover, the experiences of the Argus II recipients could color the reception of future Second Sight products. Doerr notes that his insurer paid nearly $500,000 to implant his device and for training on how to use it.
“What’s the insurance industry going to say the next time this crops up?” Doerr asks, noting that the company’s reputation is “completely shot” with the recipients of its implants.
Perk, who made speeches to praise the Argus II and is still featured in a video on the Second Sight website, says he also no longer supports the company.

Jeroen Perk, an investigator for the Dutch customs service, cried for joy after partially regaining his sight, but he no longer trusts Second Sight, the company that provided his implant.
Nanda Perk
Nevertheless, Perk remains highly reliant on the technology. When he dropped an external component of his device in late 2020 and it broke, Perk briefly debated whether to remain blind or find a way to get his Argus II working again. Three months later, he was able to revive it by crowdsourcing parts, primarily from surgeons with spare components or other Argus II recipients who no longer use their devices. Perk now has several spare parts in reserve in case of future breakdowns.
Despite the frantic efforts to retain what little sight he has, Perk has no regrets about having the device implanted. And while he no longer trusts Second Sight, he is looking forward to possibly obtaining more advanced implants from companies in the Netherlands and Australia working on their own products.
Doerr suggests that biotech firms whose implants are distributed globally be bound to some sort of international treaty requiring them to service their products in perpetuity. Such treaties are still applied to the salvage rights for ships that sunk centuries ago, he notes.
“I think that in a global tech economy, that would be a good thing,” says McLean, the fellow at Santa Clara, “but I am not optimistic about it in the near term. Business incentives push toward return on share to stockholders, not to patients and other stakeholders. We likely need to rely on some combination of corporately responsibility…and [international] government regulation. It’s tough—the Paris Climate Accord implementation at a slow walk comes to mind.”
Unlike Perk, Doerr has mostly stopped using his Argus II, the result of combination of fear of losing its assistance from wear and tear and disdain for the company that brought it to market. At 70, Doerr says he does not have the time or energy to hold the company more accountable. And with Second Sight having gone through a considerable corporate reorganization, Doerr believes a lawsuit to compel it to better serve its Argus recipients would be nothing but an extremely costly longshot.
“It’s corporate America at its best,” he observes.

