The Sickest Babies Are Covered in Wires. New Tech Is Changing That.
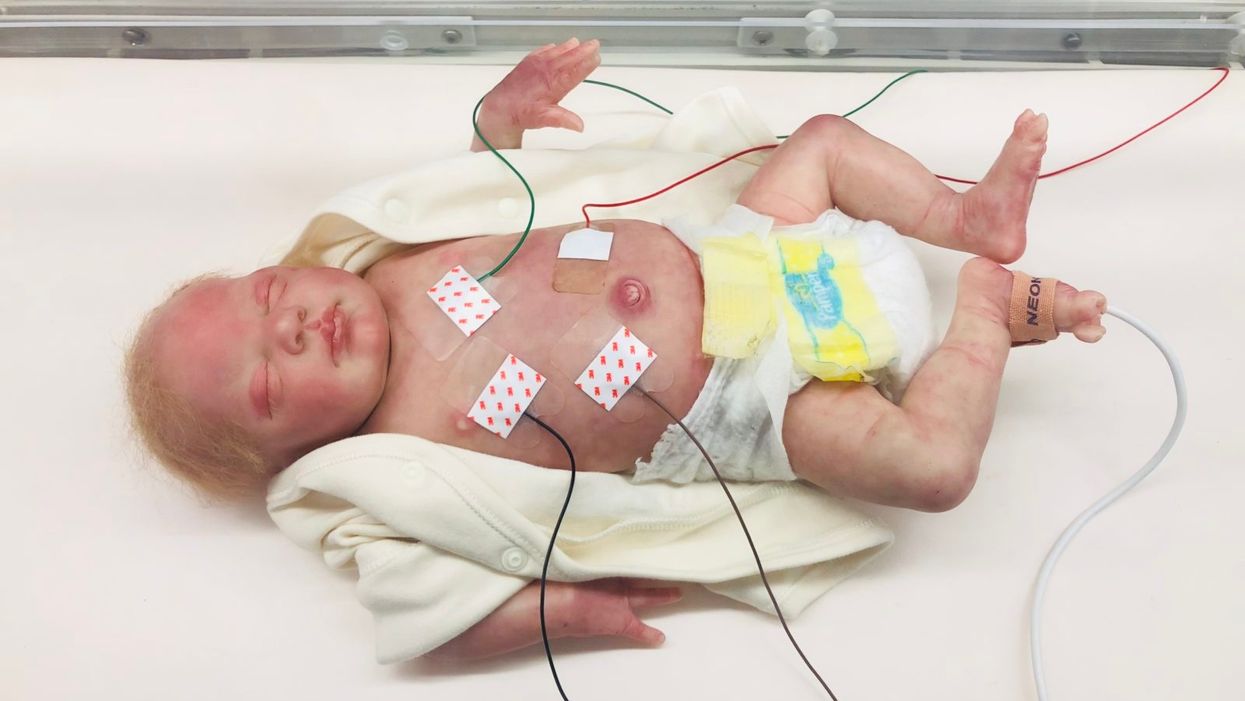
A wired baby in a neonatal intensive care unit.
I'll never forget the experience of having a child in the neonatal intensive care unit (NICU).
Now more than ever, we're working to remove the barriers between new parents and their infants.
It was another layer of uncertainty that filtered into my experience of being a first-time parent. There was so much I didn't know, and the wires attached to my son's small body for the first week of his life were a reminder of that.
I wanted to be the best mother possible. I deeply desired to bring my son home to start our lives. More than anything, I longed for a wireless baby whom I could hold and love freely without limitations.
The wires suggested my baby was fragile and it left me feeling severely unprepared, anxious, and depressed.
In recent years, research has documented the ways that NICU experiences take a toll on parents' mental health. But thankfully, medical technology is rapidly being developed to help reduce the emotional fallout of the NICU. Now more than ever, we're working to remove the barriers between new parents and their infants. The latest example is the first ever wireless monitoring system that was recently developed by a team at Northwestern University.
After listening to the needs of parents and medical staff, Debra Weese-Mayer, M.D., a professor of pediatric autonomic medicine at Feinberg School of Medicine, along with a team of materials scientists, engineers, dermatologists and pediatricians, set out to develop this potentially life-changing technology. Weese-Mayer believes wireless monitoring will have a significant impact for people on all sides of the NICU experience.
"With elimination of the cumbersome wires," she says, "the parents will find their infant more approachable/less intimidating and have improved access to their long-awaited but delivered-too-early infant, allowing them to begin skin-to-skin contact and holding with reduced concern for dislodging wires."
So how does the new system work?
Very thin "skin like" patches made of silicon rubber are placed on the surface of the skin to monitor vitals like heart rate, respiration rate, and body temperature. One patch is placed on the chest or back and the other is placed on the foot.
These patches are safer on the skin than previously used adhesives, reducing the cuts and infections associated with past methods. Finally, an antenna continuously delivers power, often from under the mattress.
The data collected from the patches stream from the body to a tablet or computer.

New wireless sensor technology is being studied to replace wired monitoring in NICUs in the coming years.
(Northwestern University)
Weese-Mayer hopes that wireless systems will be standard soon, but first they must undergo more thorough testing. "I would hope that in the next five years, wireless monitoring will be the standard in NICUs, but there are many essential validation steps before this technology will be embraced nationally," she says.
Until the new systems are ready, parents will be left struggling with the obstacles that wired monitoring presents.
Physical intimacy, for example, appears to have pain-reducing qualities -- something that is particularly important for babies who are battling serious illness. But wires make those cuddles more challenging.
There's also been minimal discussion about how wired monitoring can be particularly limiting for parents with disabilities and mobility aids, or even C-sections.
"When he was first born and I was recovering from my c-section, I couldn't deal with keeping the wires untangled while trying to sit down without hurting myself," says Rhiannon Giles, a writer from North Carolina, who delivered her son at just over 31 weeks after suffering from severe preeclampsia.
"The wires were awful," she remembers. "They fell off constantly when I shifted positions or he kicked a leg, which meant the monitors would alarm. It felt like an intrusion into the quiet little world I was trying to mentally create for us."
Over the last few years, researchers have begun to dive deeper into the literal and metaphorical challenges of wired monitoring.
For many parents, the wires prompt anxiety that worsens an already tense and vulnerable time.
I'll never forget the first time I got to hold my son without wires. It was the first time that motherhood felt manageable.
"Seeing my five-pound-babies covered in wires from head to toe rendered me completely overwhelmed," recalls Caila Smith, a mom of five from Indiana, whose NICU experience began when her twins were born pre-term. "The nurses seemed to handle them perfectly, but I was scared to touch them while they appeared so medically frail."
During the nine days it took for both twins to come home, the limited access she had to her babies started to impact her mental health. "If we would've had wireless sensors and monitors, it would've given us a much greater sense of freedom and confidence when snuggling our newborns," Smith says.
Besides enabling more natural interactions, wireless monitoring would make basic caregiving tasks much easier, like putting on a onesie.
"One thing I noticed is that many preemie outfits are made with zippers," points out Giles, "which just don't work well when your baby has wires coming off of them, head to toe."
Wired systems can pose issues for medical staff as well as parents.
"The main concern regarding wired systems is that they restrict access to the baby and often get tangled with other equipment, like IV lines," says Lamia Soghier, Medical Director of the Neonatal Intensive Care Unit at Children's National in Washington, D.C , who was also a NICU parent herself. "The nurses have to untangle the wires, which takes time, before handing the baby to the family."
I'll never forget the first time I got to hold my son without wires. It was the first time that motherhood felt manageable, and I couldn't stop myself from crying. Suddenly, anything felt possible and all the limitations from that first week of life seemed to fade away. The rise of wired-free monitoring will make some of the stressors that accompany NICU stays a thing of the past.
Antibody Testing Alone is Not the Key to Re-Opening Society
Immunity tests have too many unknowns right now to make them very useful in determining protective antibody status.
[Editor's Note: We asked experts from different specialties to weigh in on a timely Big Question: "How should immunity testing play a role in re-opening society?" Below, a virologist offers her perspective.]
With the advent of serology testing and increased emphasis on "re-opening" America, public health officials have begun considering whether or not people who have recovered from COVID-19 can safely re-enter the workplace.
"Immunity certificates cannot certify what is not known."
Conventional wisdom holds that people who have developed antibodies in response to infection with SARS-CoV-2, the coronavirus that causes COVID-19, are likely to be immune to reinfection.
For most acute viral infections, this is generally true. However, SARS-CoV-2 is a new pathogen, and there are currently many unanswered questions about immunity. Can recovered patients be reinfected or transmit the virus? Does symptom severity determine how protective responses will be after recovery? How long will protection last? Understanding these basic features is essential to phased re-opening of the government and economy for people who have recovered from COVID-19.
One mechanism that has been considered is issuing "immunity certificates" to individuals with antibodies against SARS-CoV-2. These certificates would verify that individuals have already recovered from COVID-19, and thus have antibodies in their blood that will protect them against reinfection, enabling them to safely return to work and participate in society. Although this sounds reasonable in theory, there are many practical reasons why this is not a wise policy decision to ease off restrictive stay-home orders and distancing practices.
Too Many Scientific Unknowns
Serology tests measure antibodies in the serum—the liquid component of blood, which is where the antibodies are located. In this case, serology tests measure antibodies that specifically bind to SARS-CoV-2 virus particles. Usually when a person is infected with a virus, they develop antibodies that can "recognize" that virus, so the presence of SARS-CoV-2 antibodies indicates that a person has been previously exposed to the virus. Broad serology testing is critical to knowing how many people have been infected with SARS-CoV-2, since testing capacity for the virus itself has been so low.
Tests for the virus measure amounts of SARS-CoV-2 RNA—the virus's genetic material—directly, and thus will not detect the virus once a person has recovered. Thus, the majority of people who were not severely ill and did not require hospitalization, or did not have direct contact with a confirmed case, will not test positive for the virus weeks after they have recovered and can only determine if they had COVID-19 by testing for antibodies.
In most cases, for most pathogens, antibodies are also neutralizing, meaning they bind to the virus and render it incapable of infecting cells, and this protects against future infections. Immunity certificates are based on the assumption that people with antibodies specific for SARS-CoV-2 will be protected against reinfection. The problem is that we've only known that SARS-CoV-2 existed for a little over four months. Although studies so far indicate that most (but not all) patients with confirmed COVID-19 cases develop antibodies, we don't know the extent to which antibodies are protective against reinfection, or how long that protection will last. Immunity certificates cannot certify what is not known.
The limited data so far is encouraging with regard to protective immunity. Most of the patient sera tested for antibodies show reasonable titers of IgG, the type of antibodies most likely to be neutralizing. Furthermore, studies have shown that these IgG antibodies are capable of neutralizing surrogate viruses as well as infectious SARS-CoV-2 in laboratory tests. In addition, rhesus monkeys that were experimentally infected with SARS-CoV-2 and allowed to recover were protected from reinfection after a subsequent experimental challenge. These data tentatively suggest that most people are likely to develop neutralizing IgG, and protective immunity, after being infected by SARS-CoV-2.
However, not all COVID-19 patients do produce high levels of antibodies specific for SARS-CoV-2. A small number of patients in one study had no detectable neutralizing IgG. There have also been reports of patients in South Korea testing PCR positive after a prior negative test, indicating reinfection or reactivation. These cases may be explained by the sensitivity of the PCR test, and no data have been produced to indicate that these cases are genuine reinfection or recurrence of viral infection.
Complicating matters further, not all serology tests measure antibody titers. Some rapid serology tests are designed to be binary—the test can either detect antibodies or not, but does not give information about the amount of antibodies circulating. Based on our current knowledge, we cannot be certain that merely having any level of detectable antibodies alone guarantees protection from reinfection, or from a subclinical reinfection that might not cause a second case of COVID-19, but could still result in transmission to others. These unknowns remain problematic even with tests that accurately detect the presence of antibodies—which is not a given today, as many of the newly available tests are reportedly unreliable.
A Logistical and Ethical Quagmire
While most people are eager to cast off the isolation of physical distancing and resume their normal lives, mere desire to return to normality is not an indicator of whether those antibodies actually work, and no certificate can confer immune protection. Furthermore, immunity certificates could lead to some complicated logistical and ethical issues. If antibodies do not guarantee protective immunity, certifying that they do could give antibody-positive people a false sense of security, causing them to relax infection control practices such as distancing and hand hygiene.
"We should not, however, place our faith in assumptions and make return to normality contingent on an arbitrary and uninformative piece of paper."
Certificates could be forged, putting susceptible people at higher exposure risk. It's not clear who would issue them, what they would entitle the bearer to do or not do, or how certification would be verified or enforced. There are many ways in which such certificates could be used as a pretext to discriminate against people based on health status, in addition to disability, race, and socioeconomic status. Tracking people based on immune status raises further concerns about privacy and civil rights.
Rather than issuing documents confirming immune status, we should instead "re-open" society cautiously, with widespread virus and serology testing to accurately identify and isolate infected cases rapidly, with immediate contact tracing to safely quarantine and monitor those at exposure risk. Broad serosurveillance must be coupled with functional assays for neutralization activity to begin assessing how protective antibodies might actually be against SARS-CoV-2 infection. To understand how long immunity lasts, we should study antibodies, as well as the functional capabilities of other components of the larger immune system, such as T cells, in recovered COVID-19 patients over time.
We should not, however, place our faith in assumptions and make return to normality contingent on an arbitrary and uninformative piece of paper. Re-opening society, the government, and the economy depends not only on accurately determining how many people have antibodies to SARS-CoV-2, but on a deeper understanding of how those antibodies work to provide protection.
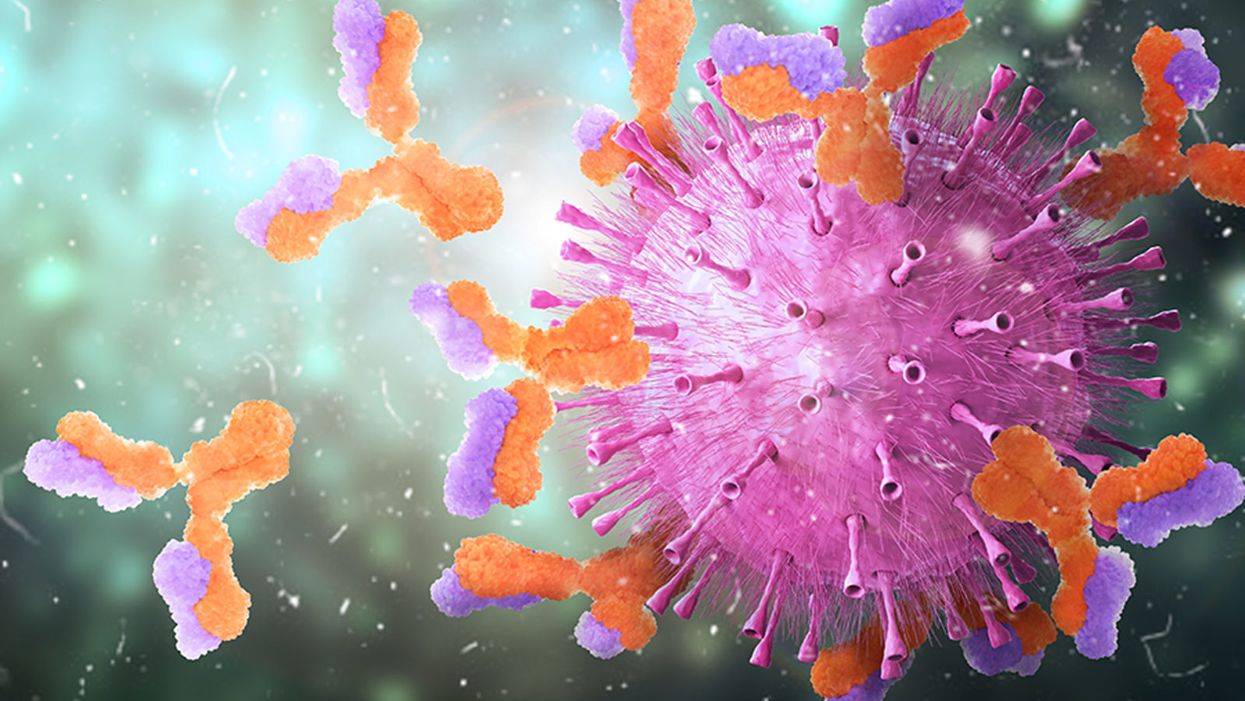
Harvard Researchers Are Using a Breakthrough Tool to Find the Antibodies That Best Knock Out the Coronavirus
Knowing which antibodies bind best to the coronavirus can help guide new medicines and vaccine development.
To find a cure for a deadly infectious disease in the 1995 medical thriller Outbreak, scientists extract the virus's antibodies from its original host—an African monkey.
"When a person is infected, the immune system makes antibodies kind of blindly."
The antibodies prevent the monkeys from getting sick, so doctors use these antibodies to make the therapeutic serum for humans. With SARS-CoV-2, the original hosts might be bats or pangolins, but scientists don't have access to either, so they are turning to the humans who beat the virus.
Patients who recovered from COVID-19 are valuable reservoirs of viral antibodies and may help scientists develop efficient therapeutics, says Stephen J. Elledge, professor of genetics and medicine at Harvard Medical School in Boston. Studying the structure of the antibodies floating in their blood can help understand what their immune systems did right to kill the pathogen.
When viruses invade the body, the immune system builds antibodies against them. The antibodies work like Velcro strips—they use special spots on their surface called paratopes to cling to the specific spots on the viral shell called epitopes. Once the antibodies circulating in the blood find their "match," they cling on to the virus and deactivate it.
But that process is far from simple. The epitopes and paratopes are built of various peptides that have complex shapes, are folded in specific ways, and may carry an electrical charge that repels certain molecules. Only when all of these parameters match, an antibody can get close enough to a viral particle—and shut it out.
So the immune system forges many different antibodies with varied parameters in hopes that some will work. "When a person is infected, the immune system makes antibodies kind of blindly," Elledge says. "It's doing a shotgun approach. It's not sure which ones will work, but it knows once it's made a good one that works."
Elledge and his team want to take the guessing out of the process. They are using their home-built tool VirScan to comb through the blood samples of the recovered COVID-19 patients to see what parameters the efficient antibodies should have. First developed in 2015, the VirScan has a library of epitopes found on the shells of viruses known to afflict humans, akin to a database of criminals' mug shots maintained by the police.
Originally, VirScan was meant to reveal which pathogens a person overcame throughout a lifetime, and could identify over 1,000 different strains of viruses and bacteria. When the team ran blood samples against the VirScan's library, the tool would pick out all the "usual suspects." And unlike traditional blood tests called ELISA, which can only detect one pathogen at a time, VirScan can detect all of them at once. Now, the team has updated VirScan with the SARS-CoV-2 "mug shot" and is beginning to test which antibodies from the recovered patients' blood will bind to them.
Knowing which antibodies bind best can also help fine-tune vaccines.
Obtaining blood samples was a challenge that caused some delays. "So far most of the recovered patients have been in China and those samples are hard to get," Elledge says. It also takes a person five to 10 days to develop antibodies, so the blood must be drawn at the right time during the illness. If a person is asymptomatic, it's hard to pinpoint the right moment. "We just got a couple of blood samples so we are testing now," he said. The team hopes to get some results very soon.
Elucidating the structure of efficient antibodies can help create therapeutics for COVID-19. "VirScan is a powerful technology to study antibody responses," says Harvard Medical School professor Dan Barouch, who also directs the Center for Virology and Vaccine Research. "A detailed understanding of the antibody responses to COVID-19 will help guide the design of next-generation vaccines and therapeutics."
For example, scientists can synthesize antibodies to specs and give them to patients as medicine. Once vaccines are designed, medics can use VirScan to see if those vaccinated again COVID-19 generate the necessary antibodies.
Knowing which antibodies bind best can also help fine-tune vaccines. Sometimes, viruses cause the immune system to generate antibodies that don't deactivate it. "We think the virus is trying to confuse the immune system; it is its business plan," Elledge says—so those unhelpful antibodies shouldn't be included in vaccines.
More importantly, VirScan can also tell which people have developed immunity to SARS-CoV-2 and can return to their workplaces and businesses, which is crucial to restoring the economy. Knowing one's immunity status is especially important for doctors working on the frontlines, Elledge notes. "The resistant ones can intubate the sick."
Lina Zeldovich has written about science, medicine and technology for Popular Science, Smithsonian, National Geographic, Scientific American, Reader’s Digest, the New York Times and other major national and international publications. A Columbia J-School alumna, she has won several awards for her stories, including the ASJA Crisis Coverage Award for Covid reporting, and has been a contributing editor at Nautilus Magazine. In 2021, Zeldovich released her first book, The Other Dark Matter, published by the University of Chicago Press, about the science and business of turning waste into wealth and health. You can find her on http://linazeldovich.com/ and @linazeldovich.