The Sickest Babies Are Covered in Wires. New Tech Is Changing That.
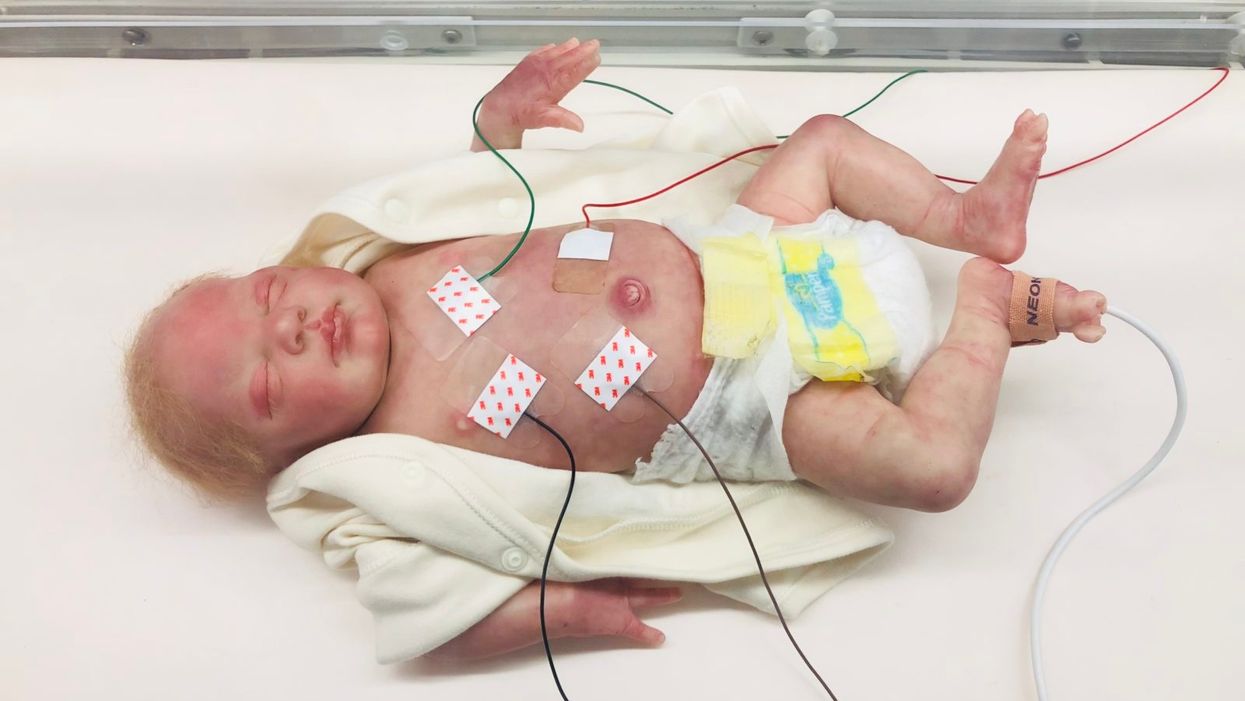
A wired baby in a neonatal intensive care unit.
I'll never forget the experience of having a child in the neonatal intensive care unit (NICU).
Now more than ever, we're working to remove the barriers between new parents and their infants.
It was another layer of uncertainty that filtered into my experience of being a first-time parent. There was so much I didn't know, and the wires attached to my son's small body for the first week of his life were a reminder of that.
I wanted to be the best mother possible. I deeply desired to bring my son home to start our lives. More than anything, I longed for a wireless baby whom I could hold and love freely without limitations.
The wires suggested my baby was fragile and it left me feeling severely unprepared, anxious, and depressed.
In recent years, research has documented the ways that NICU experiences take a toll on parents' mental health. But thankfully, medical technology is rapidly being developed to help reduce the emotional fallout of the NICU. Now more than ever, we're working to remove the barriers between new parents and their infants. The latest example is the first ever wireless monitoring system that was recently developed by a team at Northwestern University.
After listening to the needs of parents and medical staff, Debra Weese-Mayer, M.D., a professor of pediatric autonomic medicine at Feinberg School of Medicine, along with a team of materials scientists, engineers, dermatologists and pediatricians, set out to develop this potentially life-changing technology. Weese-Mayer believes wireless monitoring will have a significant impact for people on all sides of the NICU experience.
"With elimination of the cumbersome wires," she says, "the parents will find their infant more approachable/less intimidating and have improved access to their long-awaited but delivered-too-early infant, allowing them to begin skin-to-skin contact and holding with reduced concern for dislodging wires."
So how does the new system work?
Very thin "skin like" patches made of silicon rubber are placed on the surface of the skin to monitor vitals like heart rate, respiration rate, and body temperature. One patch is placed on the chest or back and the other is placed on the foot.
These patches are safer on the skin than previously used adhesives, reducing the cuts and infections associated with past methods. Finally, an antenna continuously delivers power, often from under the mattress.
The data collected from the patches stream from the body to a tablet or computer.

New wireless sensor technology is being studied to replace wired monitoring in NICUs in the coming years.
(Northwestern University)
Weese-Mayer hopes that wireless systems will be standard soon, but first they must undergo more thorough testing. "I would hope that in the next five years, wireless monitoring will be the standard in NICUs, but there are many essential validation steps before this technology will be embraced nationally," she says.
Until the new systems are ready, parents will be left struggling with the obstacles that wired monitoring presents.
Physical intimacy, for example, appears to have pain-reducing qualities -- something that is particularly important for babies who are battling serious illness. But wires make those cuddles more challenging.
There's also been minimal discussion about how wired monitoring can be particularly limiting for parents with disabilities and mobility aids, or even C-sections.
"When he was first born and I was recovering from my c-section, I couldn't deal with keeping the wires untangled while trying to sit down without hurting myself," says Rhiannon Giles, a writer from North Carolina, who delivered her son at just over 31 weeks after suffering from severe preeclampsia.
"The wires were awful," she remembers. "They fell off constantly when I shifted positions or he kicked a leg, which meant the monitors would alarm. It felt like an intrusion into the quiet little world I was trying to mentally create for us."
Over the last few years, researchers have begun to dive deeper into the literal and metaphorical challenges of wired monitoring.
For many parents, the wires prompt anxiety that worsens an already tense and vulnerable time.
I'll never forget the first time I got to hold my son without wires. It was the first time that motherhood felt manageable.
"Seeing my five-pound-babies covered in wires from head to toe rendered me completely overwhelmed," recalls Caila Smith, a mom of five from Indiana, whose NICU experience began when her twins were born pre-term. "The nurses seemed to handle them perfectly, but I was scared to touch them while they appeared so medically frail."
During the nine days it took for both twins to come home, the limited access she had to her babies started to impact her mental health. "If we would've had wireless sensors and monitors, it would've given us a much greater sense of freedom and confidence when snuggling our newborns," Smith says.
Besides enabling more natural interactions, wireless monitoring would make basic caregiving tasks much easier, like putting on a onesie.
"One thing I noticed is that many preemie outfits are made with zippers," points out Giles, "which just don't work well when your baby has wires coming off of them, head to toe."
Wired systems can pose issues for medical staff as well as parents.
"The main concern regarding wired systems is that they restrict access to the baby and often get tangled with other equipment, like IV lines," says Lamia Soghier, Medical Director of the Neonatal Intensive Care Unit at Children's National in Washington, D.C , who was also a NICU parent herself. "The nurses have to untangle the wires, which takes time, before handing the baby to the family."
I'll never forget the first time I got to hold my son without wires. It was the first time that motherhood felt manageable, and I couldn't stop myself from crying. Suddenly, anything felt possible and all the limitations from that first week of life seemed to fade away. The rise of wired-free monitoring will make some of the stressors that accompany NICU stays a thing of the past.
On left, the fungus (the whitish, fluffy material) of the Fungi Mutarium growing within the agar cups and degrading the plastic (the black/gray material in the center).
Between the ever-growing Great Pacific Garbage Patch, the news that over 90% of plastic isn't recycled, and the likely state of your personal trash can, it's clear that the world has a plastic problem.
Scientists around the world have continued to discover different types of fungus that can degrade specific types of plastic.
We now have 150 million tons of plastic in our oceans, according to estimates; by 2050, there could be more plastic than fish. And every new batch of trash compounds the issue: Plastic is notorious for its longevity and resistance to natural degradation.
The Lowdown
Enter the humble mushroom. In 2011, Yale students made headlines with the discovery of a fungus in Ecuador, Pestalotiopsis microspora, that has the ability to digest and break down polyurethane plastic, even in an air-free (anaerobic) environment—which might even make it effective at the bottom of landfills. Although the professor who led the research trip cautioned for moderate expectations, there's an undeniable appeal to the idea of a speedier, cleaner, side effect-free, and natural method of disposing of plastic.
A few years later, this particular application for fungus got a jolt of publicity from designer Katharina Unger, of LIVIN Studio, when she collaborated with the microbiology faculty at Utrecht University to create a project called the Fungi Mutarium. They used the mycelium—which is the threadlike, vegetative part of a mushroom—of two very common types of edible mushrooms, Pleurotus ostreatus (Oyster mushrooms) and Schizophyllum commune (Split gill mushrooms). Over the course of a few months, the fungi fully degraded small pieces of plastic while growing around pods of edible agar. The result? In place of plastic, a small mycelium snack.
Other researchers have continued to tackle the subject. In 2017, scientist Sehroon Khan and his research team at the World Agroforestry Centre in Kunming, China discovered another biodegrading fungus in a landfill in Islamabad, Pakistan: Aspergillus tubingensis, which turns out to be capable of colonizing polyester polyurethane (PU) and breaking it down it into smaller pieces within the span of two months. (PU often shows up in the form of packing foam—the kind of thing you might find cushioning a microwave or a new TV.)
Next Up
Utrecht University has continued its research, and scientists around the world have continued to discover different types of fungus that can degrade different, specific types of plastic. Khan and his team alone have discovered around 50 more species since 2017. They are currently working on finding the optimal conditions of temperature and environment for each strain of fungus to do its work.
Their biggest problem is perhaps the most common obstacle in innovative scientific research: Cash. "We are developing these things for large-scale," Khan says. "But [it] needs a lot of funding to get to the real application of plastic waste." They plan to apply for a patent soon and to publish three new articles about their most recent research, which might help boost interest and secure more grants.
Is there a way to get the fungi to work faster and to process bigger batches?
Khan's team is working on the breakdown process at this point, but researchers who want to continue in Unger's model of an edible end product also need to figure out how to efficiently and properly prepare the plastic input. "The fungi is sensitive to infection from bacteria," Unger says—which could turn it into a destructive mold. "This is a challenge for industrialization—[the] sterilization of the materials, and making the fungi resistant, strong, and faster-growing, to allow for a commercial process."
Open Questions
Whether it's Khan's polyurethane-chomping fungus or the edible agar pods from the Fungi Mutarium, the biggest question is still about scale. Both projects took several months to fully degrade a small amount of plastic. That's much shorter than plastic's normal lifespan, but still won't be enough to keep up with the global production of plastic. Is there a way to get the fungi to work faster and to process bigger batches?
We'd also need to figure out where these plastic recyclers would live. Could individuals keep a small compost-like heap, feeding in their own plastic and harvesting the mushrooms? Or could this be a replacement for local recycling centers?
There are still only these few small experiments for reference. But taken together, they suggest a fascinating future for waste disposal: An army of mycelium chewing quietly and methodically through our plastic bags and foam coffee cups—and potentially even creating a new food source along the way. We could have our trash and eat it, too.
Electricity is emerging as a powerful treatment for chronic ailments.
Kelly, a case manager for an insurance company, spent years battling both migraines and Crohn's, a disease in which the immune system attacks the intestines.
For many people, like Kelly, a stronger electric boost to the vagus nerve could be life-changing.
After she had her large intestine removed, her body couldn't absorb migraine medication. Last year, about twice a month, she endured migraines so bad she couldn't function. "It would go up to a ten, and I would rock, wait it out," she said. The pain might last for three days.
Then her neurologist showed her a new device, gammaCore, that tames migraines by stimulating a nerve—not medication. "I don't have to put a chemical in my body," she said. "I was thrilled."
At first, Kelly used the device at the onset of a migraine, applying electricity to her pulse at the front of her neck for six minutes. The pain peaked at about half the usual intensity--low enough, she said, that she could go to work. Four months ago, she began using the device for two minutes each night as prevention, and she hasn't had a serious migraine since.
The Department of Defense and Veterans Administration now offer gammaCore to patients, but it hasn't yet been approved by Medicare, Medicaid, or most insurers. A month of therapy costs $600 before insurance or a generous financial assistance program kicks in.

A patient uses gammaCore, a non invasive vagal nerve stimulator device that was FDA approved in November 2018, to treat her migraine.
(Photo captured from a patient video at gammacore.com)
If the poet Walt Whitman wrote "I Sing The Body Electric" today, he might get specific and point to the vagus nerve, a bundle of fibers that run from the brainstem down the neck to the heart and gut. Singing stimulates it—and for many people, like Kelly, a stronger electric boost to the nerve could be life-changing.
The mind-body connection isn't just an idea — the vagus nerve literally carries signals from the mind to the body and back. It may explain the link between childhood trauma and illnesses such as chronic pain and headaches in adults. "How is it possible that a psychological event causes pain years later?" asked Peter Staats, co-founder of electroCore, which has won approval for its new device from the Food and Drug Administration (FDA) for both migraine and cluster headaches. "There has to be a mind-body interface, and that is the vagus nerve," he said.
Scientists knew that this nerve controlled your heart rate and blood pressure, but in the past decade it has been linked to both pain and the immune system.
"Everything is gated through the vagus -- problems with the gut, the heart, and the lungs," said Chris Wilson, a researcher at Loma Linda University, in California. Wilson is studying how vagus nerve stimulation (VNS) could help pre-term babies who develop lung infections. "Nearly every one of our chronic diseases, including cancer, Alzheimer's, Parkinson's, chronic arthritis and rheumatoid arthritis, and depression and chronic pain…could benefit from an appropriate stimulator," he said.
It's unfortunate that Kelly got her device only after her large intestine was gone. SetPoint Medical, a privately held California company founded to develop electronic treatments for chronic autoimmune diseases, has announced early positive results with VNS for both Crohn's and rheumatoid arthritis.
As SetPoint's chief medical officer, David Chernoff, put it, "We're hacking into the nervous system to activate a system that is already there," an approach that, he said, could work "on many diseases that are pain- and inflammation-based." Inflammation plays a role in much modern illness, including depression and obesity. The FDA already has approved VNS for both, using surgically implanted devices similar to pacemakers. (GammaCore is external.)
The history of VNS implants goes back to 1997, when the FDA approved one for treating epilepsy and researchers noticed that it rapidly lifted depression in epileptic patients. By 2005, the agency had approved an implant for treatment-resistant depression. (Insurance companies declined to reimburse the approach and it didn't take off, but that might change: in February, the Center for Medicare and Medicaid Services asked for more data to evaluate coverage.) In 2015, the FDA approved an implant in the abdomen to regulate appetite signals and help obese people lose weight.
The link to inflammation had emerged a decade earlier, when researchers at the Feinstein Institute for Medical Research, in Manhasset, New York, demonstrated that stimulating the nerve with electricity in rats suppressed the production of cytokines, a signaling protein important in the immune system. The researchers developed a concept of a hard-wired pathway, through the vagus nerve, between the immune and nervous system. That pathway, they argued, regulates inflammation. While other researchers argue that VNS is helpful by other routes, there is clear evidence that, one way or another, it does affect immunity.
At the same time, investors are seeking alternatives to drugs.
The Feinstein rat research concluded that it took only a minute a day of stimulation and tiny amounts of energy to activate an anti-inflammatory reflex. This means you can use devices "the size of a coffee bean," said Chernoff, much less clunky than current pacemakers—and advances in electronic technology are making them possible.
At the same time, investors are seeking alternatives to drugs. "There's been a push back on drug pricing," noted Lisa Rhoads, a managing director at Easton Capital Investment Group, in New York, which supported electroCore, "and so many unintended consequences."
In 2016, the U.S. National Institutes of Health began pumping money into relevant research, in a program called "Stimulating Peripheral Activity to Relieve Conditions," which focuses on "understanding peripheral nerves — nerves that connect the brain and spinal cord to the rest of the body — and how their electrical signals control internal organ function."
GlaxoSmithKline formed Galvani Bioelectronics with Google to study miniature implants. It had already invested in Action Potential Venture Capital, in Cambridge, Massachusetts, which holds SetPoint and seven other companies "that are all targeting a nerve to treat a chronic disease," noted partner Imran Eba. "I see a future in which bioelectronics medicine is competing directly with drugs," he said.
Treating the body with electricity could bring more ease and lower costs. Many people with serious auto-immune disease, for example, have to inject themselves with drugs that cost $60,000 a year. SetPoint's implant would cost less and only need charging once a week, using a charger worn around the neck, Chernoff said. The company receives notices remotely and can monitor compliance.
Implants also allow the treatment to target a nerve precisely, which could be important with Parkinson's, chronic pain, and depression, observed James Cavuoto, editor and publisher of Neurotech Reports. They may also allow for more fine-turning. "In general, the industry is looking for signals, biomarkers that indicate when is the right time to turn on and turn off the stimulation. It could dramatically increase the effectiveness of the therapy and conserve battery life," he said.
Eventually, external devices could receive data from biomarkers as well. "It could be something you wear on your wrist," Cavuoto noted. Bluetooth-enabled devices could communicate with phones or laptops for data capture. External devices don't require surgery and put the patient in charge. "In the future you'll see more customer specification: Give the patient a tablet or phone app that lets them track and modify their parameters, within a range. With digital devices we have an enormous capability to customize therapies and collect data and get feedback that can be fed back to the clinician," Cavuoto said.
Slow deep breathing, the traditional mind-body intervention, is "like watching Little League. What we're doing is Major League."
It's even possible to stimulate the vagus through the ear, where one branch of the bundle of fibers begins. In a fetus, the tissue that becomes the ear is also part of the vagus nerve, and that one bit remains. "It's the same point as the acupuncture point," explained Mark George, a psychiatrist and pioneer researcher in depression at Medical University of South Carolina in Charleston. "Acupuncture figured out years ago by trial and error what we're just learning about now."
Slow deep breathing, the traditional mind-body intervention, also affects the vagus nerve in positive ways, but gently. "That's like watching Little League," Staats, the co-founder of electroCore, said. "What we're doing is Major League."
In ten years, researcher Wilson suggested, you could be wearing "a little ear cuff" that monitors your basic autonomic tone, a heart-attack risk measure governed in part by the vagus nerve. If your tone looked iffy, the stimulator would intervene, he said, "and improve your mood, cognition, and health."
In the meantime, we can take some long slow breaths, read Whitman, and sing.